Clear Sky Science · en
Synthesis of sialylated human milk oligosaccharides by automated glycan assembly
Why Better Milk Sugars Matter
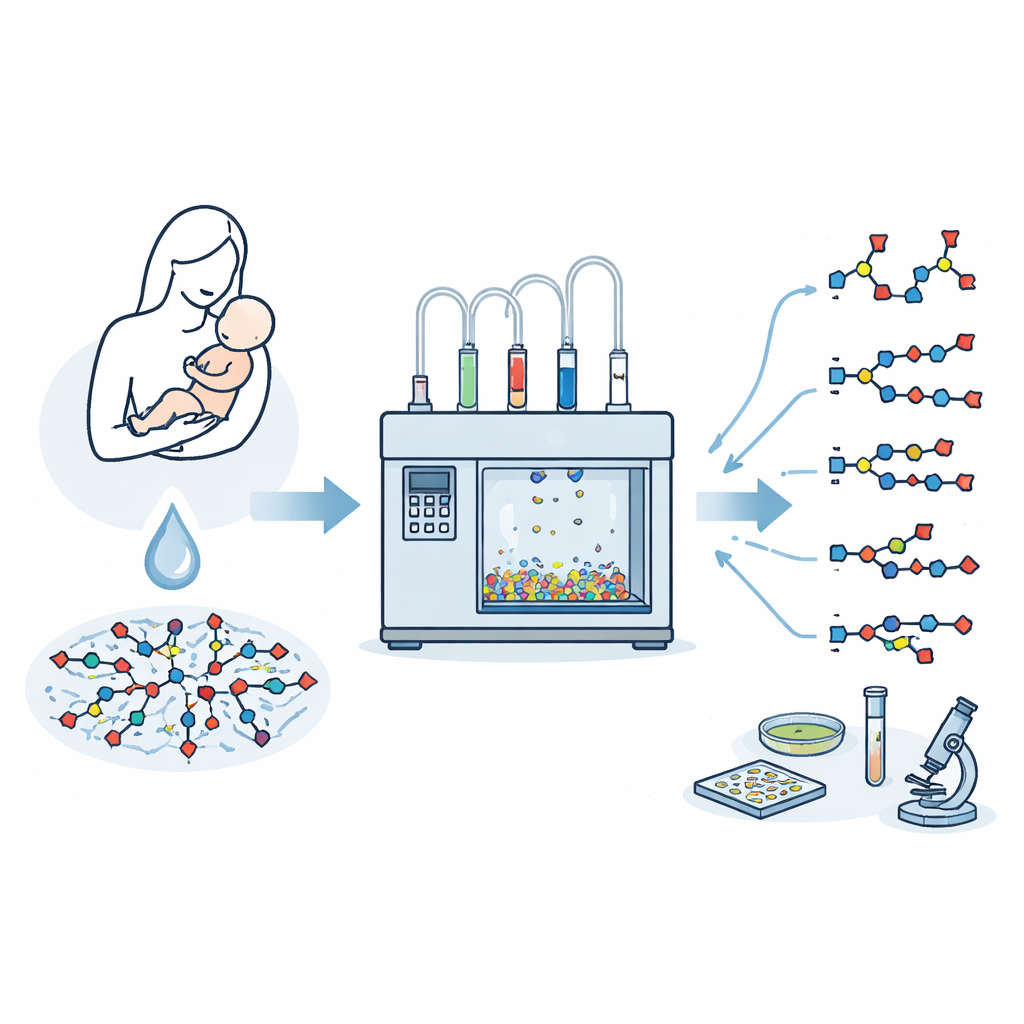
Breast milk contains a rich mix of complex sugars that help shape a baby’s gut, immune system, and resistance to infections. Among the most important of these are sialylated human milk oligosaccharides—branching sugar chains capped with a special sugar called sialic acid. Scientists would like to study each of these molecules in detail and even turn them into medicines or diagnostics, but isolating pure samples from milk is extremely difficult. This study describes a way to build these intricate milk sugars rapidly and precisely using an automated sugar‑building machine.
Special Sugars at the Ends of Chains
Sialic acids sit at the tips of many sugar chains on cell surfaces, where they play key roles in communication between cells, immune defense, and how viruses and bacteria latch onto our tissues. In breast milk, roughly one in five known oligosaccharides carries sialic acid. These sialylated structures are linked to benefits such as protection against gut disease in preterm infants and acting as decoys that trap flu viruses. Yet nature provides them only in tiny amounts mixed into a complex soup, so researchers need efficient synthetic routes to obtain milligram to gram quantities of single, well‑defined structures.
Turning Sugar Assembly into a Machine Process
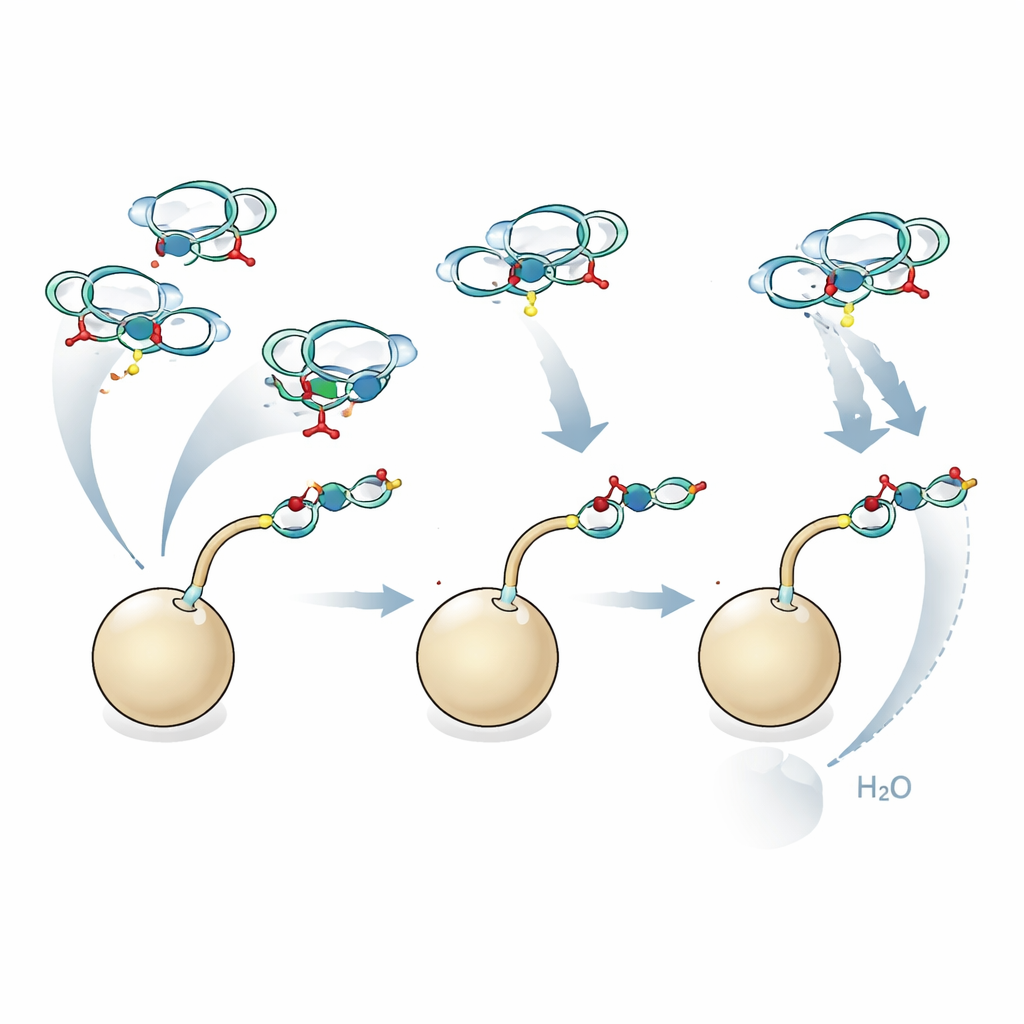
Automated glycan assembly is a technology that strings together simple sugar building blocks on tiny resin beads, step by step, under computer control. It avoids laborious purification after each step and can compress syntheses that once took months into days. However, attaching sialic acid to a growing chain on a solid support has been a long‑standing problem. The shape and charge of sialic acid favor an unwanted breakdown reaction instead of the desired bond formation, and it is difficult to control which three‑dimensional arrangement ("handedness") the new bond takes. Earlier work either relied on pre‑made two‑sugar blocks tailored to each target or worked only for very short chains.
A New Class of “Caged” Sialic Acid Blocks
The authors adopt and adapt a clever concept: macrobicyclic sialic acid donors. In these building blocks, a small chemical tether connects different parts of the sialic acid, forming a rigid cage‑like structure. This constraint steers the incoming sugar chain to attack from the correct side, boosting the desired three‑dimensional outcome while suppressing the breakdown pathway. The team systematically optimized how these donors behave on solid support—tuning solvent mixtures, how much donor is added at once, temperature, and the design of the incoming sugar units. They discovered that moderately reactive donors perform better on the crowded resin than highly reactive ones, and that exposing certain positions on the acceptor sugar during the reaction can relieve crowding and improve yields.
Building a Library of Complex Milk Sugars
Armed with these conditions, the researchers assembled nine different sialylated human milk oligosaccharides of varying size and branching, including molecules decorated not only with sialic acid but also with another important sugar, fucose. A highlight is the successful automated synthesis of DSLNF II, a heavily branched, fucosylated and disialylated seven‑sugar chain that has resisted enzyme‑based methods and previously required an intricate, hand‑crafted total synthesis. Along the way, the team uncovered how subtle changes far from the reaction site—such as swapping one type of temporary protective group for another on a distant sugar—can dramatically speed up or slow down sialic acid attachment.
From Protected Chains to Ready‑to‑Use Tools
Because automated assembly relies on protective groups to control which positions react, the final products initially carry many temporary chemical masks. Removing all of these without damaging fragile sialic acid and fucose linkages is challenging. The authors devised a gentle, stepwise deprotection sequence beginning with a zinc–copper reduction, followed by controlled acetylation, mild base treatment, and finally hydrogenation. This protocol consistently yielded clean, fully unmasked glycans, each bearing a short spacer at one end so they can be easily attached to slides, particles, or proteins for biological testing.
Opening the Door to Systematic Sugar Studies
By solving the bottleneck of reliably adding sialic acid on a solid support, this work turns automated glycan assembly into a much more powerful platform for exploring the biology of human milk sugars and other sialylated glycans. Researchers can now access panels of closely related structures in days, vary branching patterns or the presence of fucose in a controlled way, and directly test how these differences affect immunity, infection, or disease. In practical terms, the study brings us closer to routine, on‑demand production of complex milk sugars as tools for research, potential therapeutics, or components of next‑generation infant nutrition.
Citation: Kuo, YT., Le Mai Hoang, K. & Seeberger, P.H. Synthesis of sialylated human milk oligosaccharides by automated glycan assembly. Nat Commun 17, 4214 (2026). https://doi.org/10.1038/s41467-026-73028-w
Keywords: human milk oligosaccharides, sialic acid, automated glycan assembly, carbohydrate synthesis, infant nutrition