Clear Sky Science · en
A single-cell and spatial atlas of early human olfactory development
How Our Sense of Smell Begins
The sense of smell shapes how we experience food, danger, and even other people, yet we know surprisingly little about how this system first forms in the human fetus. This study uses cutting-edge genetic mapping tools to create a detailed atlas of the early human nose, focusing on the tissue that will later detect odors. By tracking thousands of individual cells and where they sit in the developing nasal cavity, the researchers show how the smell-sensing lining of the nose is built and when its nerve cells first switch on their odor receptors.
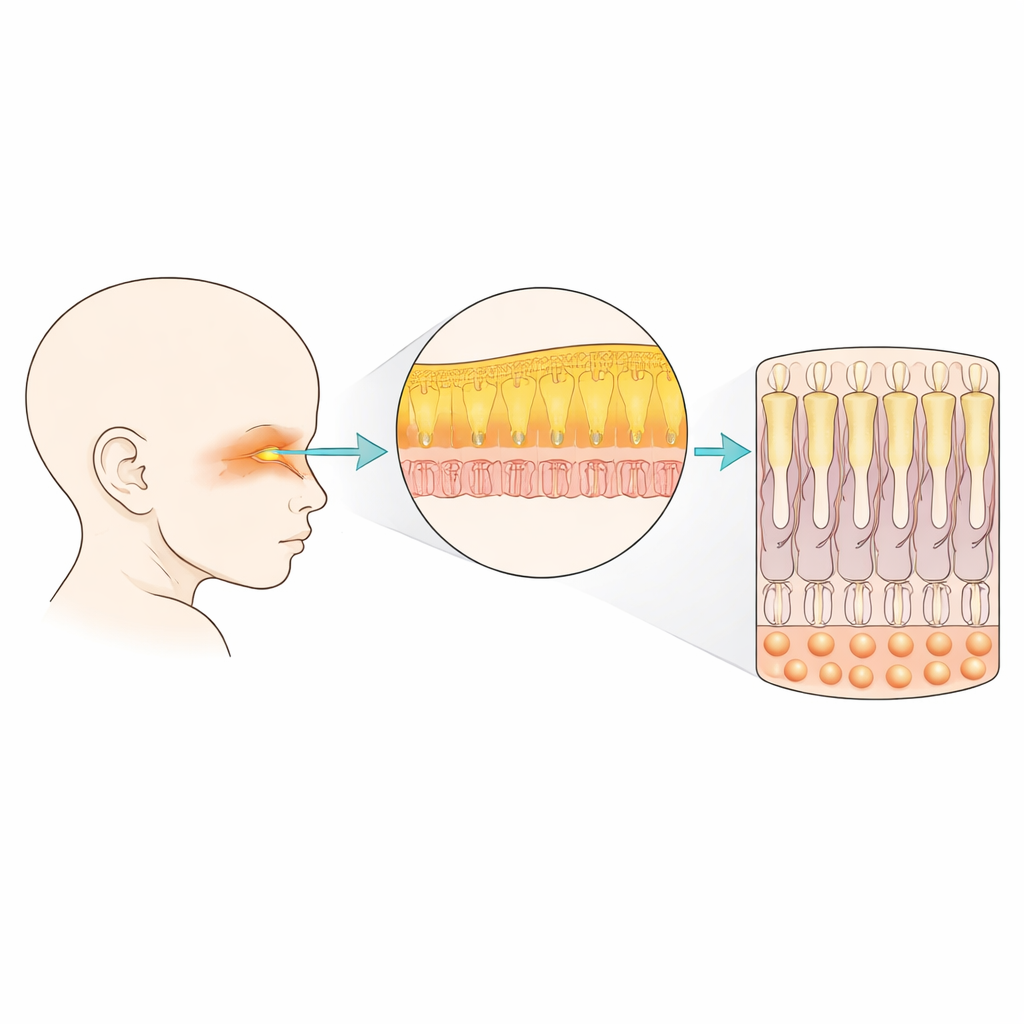
Building Blocks of the Early Nose
The human nose is more than a simple tube for air. Even in the first trimester, it contains cartilage, bone, blood vessels, immune cells, and several kinds of nerve and support cells. The team studied nasal tissue from human fetuses between 7 and 12 weeks after conception and analyzed nearly 42,000 individual cell nuclei. From these data, they could sort the cells into 32 distinct groups, including nerve cells, their precursors, structural cells, and the lining cells of both the smell-sensing and breathing parts of the nose. This revealed how early the nasal region already resembles a complex miniature organ rather than a simple sheet of cells.
Shaping the Smell-Sensing Lining
Within this complex tissue, the researchers focused on the olfactory epithelium—the specialized strip of tissue that houses smell-sensing nerve cells. They identified the main players: basal stem-like cells at the bottom, intermediate precursors, immature smell neurons, and supporting and microvillar cells nearer the surface. Over time, they saw a clear shift: early on, dividing stem and precursor cells are common; later, these give way to growing numbers of immature neurons and support cells. By comparing patterns of gene activity, they inferred how basal cells give rise to several branches of the tissue, feeding both the nerve lineage and non-neuronal support lineages, suggesting that the nose’s lifelong ability to renew its smell cells is wired in from very early development.
Mapping Cells in Their Native Neighborhood
Knowing what cell types exist is only half the story; where they sit in the tissue matters just as much. To place each cell type back into its physical context, the team used a technique that records the positions of hundreds of different RNA molecules in thin sections of fetal heads. This spatial map confirmed that the olfactory epithelium and the neighboring respiratory lining form distinct but adjacent territories. It also showed that the smell-sensing region is not uniform: its front and back segments and its upper and lower parts have different thicknesses and cell mixtures. Key developmental signals form patterned zones in the surrounding tissue, hinting that local chemical cues help carve out where smell cells form and how the epithelium expands along the nasal cavity.
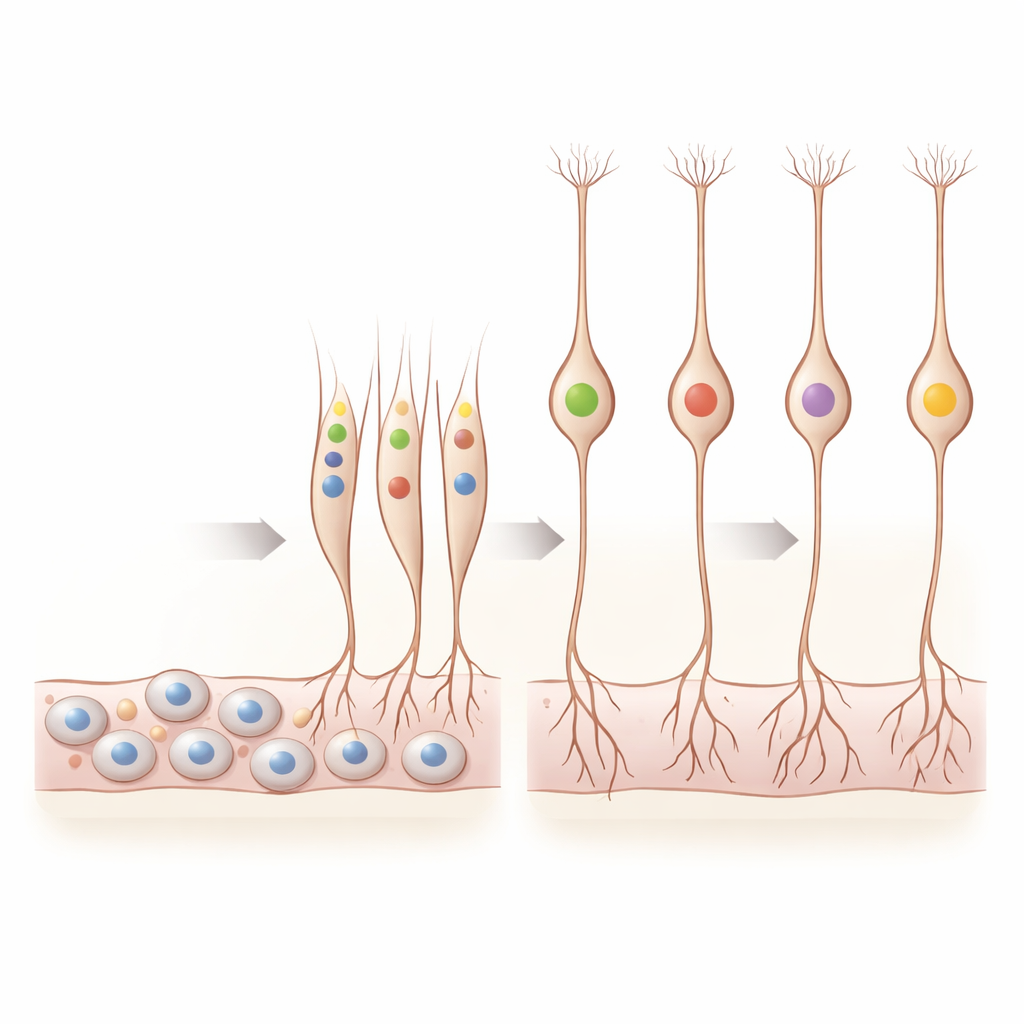
When Neurons Decide What to Smell
A central mystery in smell biology is how each sensory neuron ends up using just one odor receptor from the hundreds encoded in our DNA. By examining receptor genes in individual fetal neurons, the researchers found that this “one neuron–one receptor” rule is already taking shape in the first trimester. They detected 169 different odor receptor genes switched on, mostly in immature smell neurons. Many precursor cells had no receptor activity, but once neurons began to mature, a growing fraction expressed a single dominant receptor, with only a small minority briefly showing two or more. Over the weeks studied, cells with strong dominance of one receptor became more common, and receptor signals were tightly confined to the olfactory epithelium in space.
What This Means for Health and Disease
Together, these findings show that the basic architecture and core rules of the human smell system are put in place remarkably early before birth. The nasal lining already contains most of the major cell types found in adults, stem cells are organized into renewal pathways, and developing neurons are committing to single odor receptors within specific regions of the tissue. This atlas offers a reference map for researchers studying birth defects of the nose, inherited loss of smell, and conditions where smell is an early warning sign of brain disease. By revealing when and where things are supposed to happen, it provides a guide for understanding—and eventually correcting—what goes wrong when the sense of smell fails to develop properly.
Citation: Mbouamboua, Y., Lebrigand, K., Nampoothiri, S. et al. A single-cell and spatial atlas of early human olfactory development. Nat Commun 17, 3537 (2026). https://doi.org/10.1038/s41467-026-71595-6
Keywords: olfactory development, single-cell atlas, spatial transcriptomics, olfactory receptors, fetal nasal epithelium