Clear Sky Science · en
Remodeling synaptic connections via engineered neuron-astrocyte interactions
Why changing brain connections matters
Our brains are wired with trillions of tiny junctions called synapses, where nerve cells pass signals to one another. These connections are not fixed like a circuit board; they are constantly being strengthened, weakened, added, or removed. Subtle imbalances in this wiring are thought to underlie many brain disorders, from autism to Alzheimer’s disease. Yet scientists have had few precise tools to selectively reshape synaptic connections without simply turning activity up or down. This study introduces a synthetic biological method to gently “edit” synapses through the brain’s own support cells, revealing how circuits can become leaner yet more flexible—and how this might one day be harnessed for therapy. 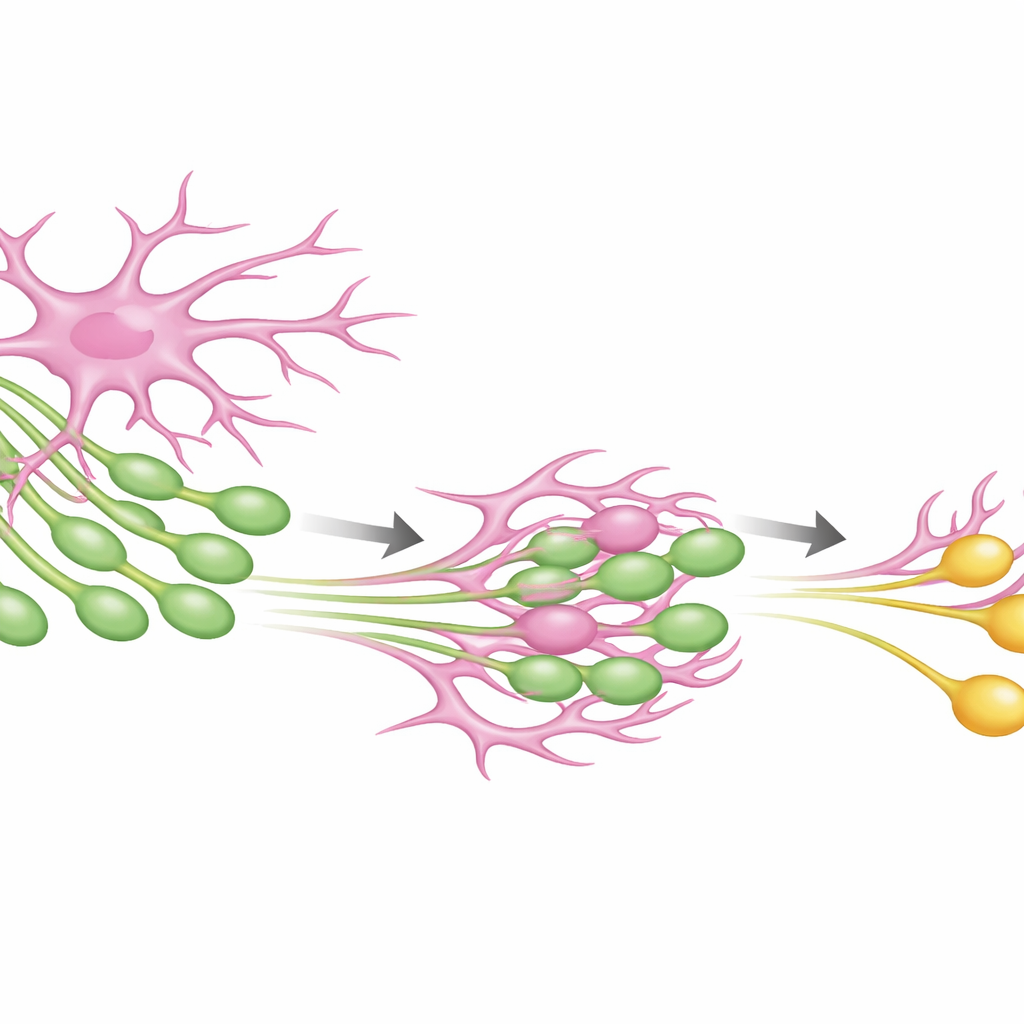
A new way to make brain cells touch
The authors built an artificial “Velcro” system that makes chosen cells stick together very tightly. They used a well-known glowing protein, GFP, as the “handle” on one cell type, and a tiny antibody-like partner (a nanobody) that grabs GFP as the “hook” on the other cell type. Both parts were anchored on cell surfaces so that when a GFP-bearing cell encountered a nanobody-bearing neighbor, their membranes locked together. In lab dishes, this strong contact triggered a process resembling trogocytosis—a kind of cellular nibbling—where the receptor-bearing cell pinched off and internalized small patches of the partner’s membrane and nearby contents. The stronger the molecular grip between the two engineered proteins, the more efficiently this nibbling occurred, and the process proved versatile across many cell types without killing the cells.
Enlisting brain support cells to nibble neurons
The team then turned to astrocytes, star-shaped support cells that naturally wrap around synapses and help remove unwanted connections. They expressed the GFP “ligand” in neurons and the nanobody “receptor” in astrocytes. In cultured neurons, astrocytes repeatedly nibbled tiny bits of neuronal membrane wherever their processes touched axons, dendrites, or cell bodies. Importantly, synaptic proteins located near the GFP tag were dragged along into astrocytes, showing that the engineered interaction could selectively pull synaptic material away from neurons. This confirmed that the synthetic system—dubbed SynTrogo, for Synthetic Trogocytosis—can redirect how closely astrocytes engage with neuronal connections and what cellular cargo they take up.
Thinning out connections in a memory circuit
To see what SynTrogo does in a living brain, the researchers targeted a classic memory pathway in the mouse hippocampus, where CA3 neurons connect to CA1 neurons. They put the GFP handle on CA3 axons and the nanobody receptor on CA1 astrocytes. Under these conditions, fluorescent signal from CA3 axons accumulated within astrocytic territories, indicating active nibbling. Microscopy at nanometer resolution showed that astrocyte membranes formed unusually tight, interlocking interfaces with presynaptic boutons, sometimes partially wrapping pieces of the axon that contained vesicles and other organelles. Across this pathway, the density of excitatory synapses dropped by roughly a quarter, especially where engineered astrocytes were present, while inhibitory connections modestly increased—changing the balance of inputs onto CA1 neurons without destroying axons outright.
Stronger survivors and more adaptable circuits
Surprisingly, the synapses that remained after this pruning were not weaker. Electrical recordings showed fewer excitatory events overall, matching the reduced number of synapses, but the remaining presynaptic terminals released neurotransmitter more readily and had a larger readily releasable pool of vesicles. Three-dimensional reconstructions revealed that surviving boutons were bigger, packed with more synaptic vesicles and mitochondria, and faced enlarged postsynaptic spines with expanded contact zones. Many of these spines acquired a specialized internal structure, the spine apparatus, associated with calcium handling and receptor trafficking. Functionally, this remodeled network displayed stronger long-term potentiation, a hallmark of synaptic plasticity, and mice performed better in contextual fear memory tests, retaining memories longer while still being able to extinguish them through training. 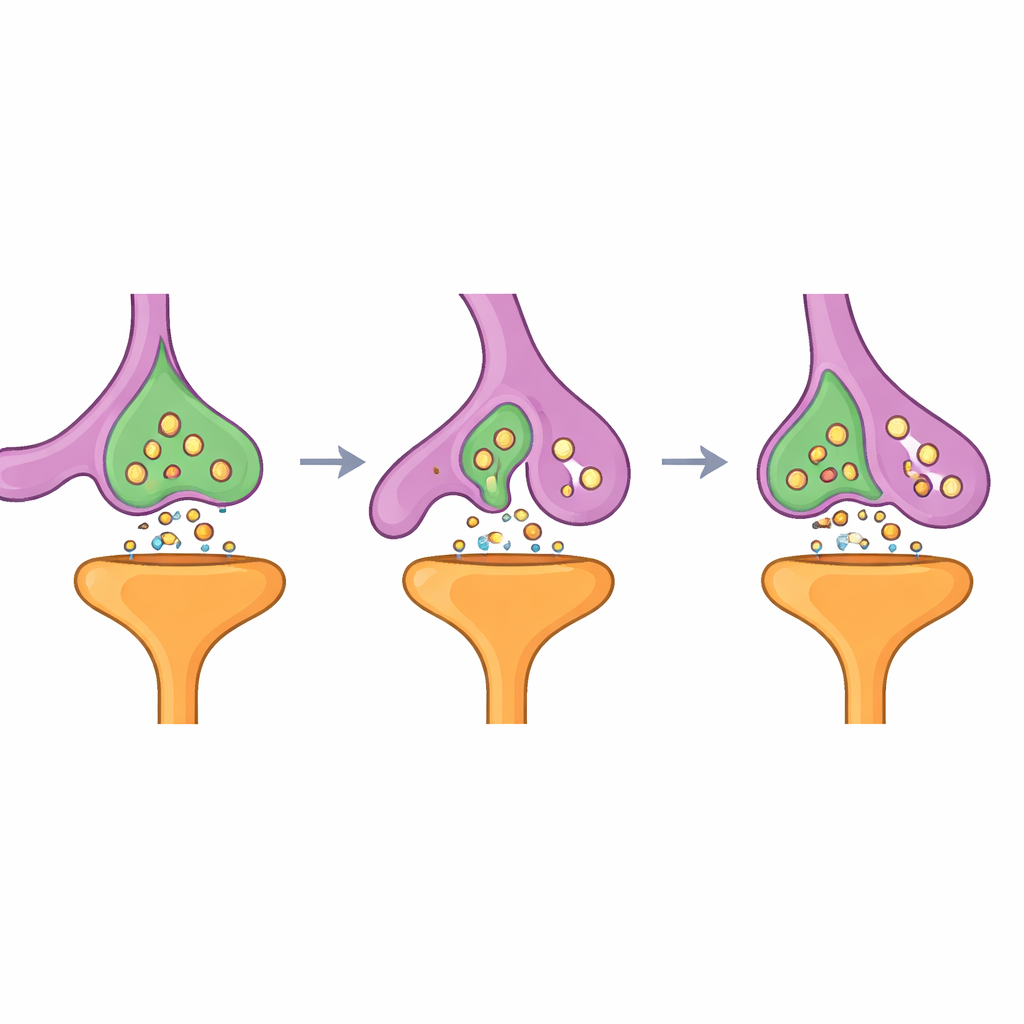
What this could mean for brain health
To a non-specialist, the key message is that carefully reducing the number of synapses in a circuit does not necessarily blunt brain function; it can actually sharpen it. SynTrogo shows that by guiding astrocytes to selectively nibble on certain neuronal contacts, it is possible to thin out connections while prompting surviving synapses to grow stronger and more adaptable. This synthetic approach separates structural rewiring from simple changes in electrical activity, offering a powerful new way to study how circuit architecture shapes learning, memory, and resilience. In the long run, strategies inspired by SynTrogo might help rebalance overly dense or disorganized connections in conditions such as autism, schizophrenia, or early Alzheimer’s disease, turning controlled synapse loss into an opportunity for healthier circuit remodeling.
Citation: Kim, S.H., Won, W., Kim, G.H. et al. Remodeling synaptic connections via engineered neuron-astrocyte interactions. Nat Commun 17, 3490 (2026). https://doi.org/10.1038/s41467-026-71440-w
Keywords: synaptic pruning, astrocytes, neural plasticity, synthetic biology, hippocampus