Clear Sky Science · en
Fluorescence-based mapping of condensate dielectric permittivity uncovers hydrophobicity-driven membrane interactions
Why Droplets Inside Cells Matter
Inside our cells, many crucial reactions happen not in hard-walled compartments, but in soft, liquid-like droplets called biomolecular condensates. These tiny droplets help organize the cell’s crowded interior and control where and when chemistry happens. Yet one basic question has remained murky: what makes some droplets cling to cellular membranes while others stay away? This study shows that a simple physical property—how easily a material responds to electric fields, closely tied to how watery or oily it is—can explain and even predict how these droplets interact with membranes.

Seeing Invisible Properties With Light
The authors developed a way to “see” the local electrical environment inside and around droplets using a special fluorescent dye called ACDAN. When this dye is excited by light, its color shifts depending on how freely nearby water molecules can move and reorient, which in turn reflects the local dielectric permittivity—a measure of how polar or hydrophobic the surroundings are. By recording a full spectrum of emitted colors at every pixel in a microscope image and analyzing those spectra mathematically, the team converts color information into a quantitative map of permittivity with pixel-level resolution.
Droplets From Oil-Like to Water-Like
Using this optical method, the researchers examined a variety of model condensates made from proteins, short peptides, and polymers. They discovered that droplets span a surprisingly wide range of dielectric permittivities, from values similar to oils to values approaching that of pure water. This means condensates can be far more hydrophobic or hydrophilic than previously appreciated. They also found that the type of phase separation chemistry (for example, charged polymers sticking together versus neutral molecules separating) is not enough to predict permittivity. Instead, factors like how much water is trapped inside the dense phase and how proteins are structured and packed make a major difference.
Tracking Crowding and Chemical Changes
The team then used permittivity maps to follow how droplets and their surroundings change as the overall mixture is altered. In classic water-based mixtures of two polymers, and in protein-rich condensates, increasing crowding or salt concentration changed the water content and thus the permittivity of both the dense droplets and the more dilute phase outside. Adding small amounts of ATP—a molecule best known as the cell’s energy currency—acted as a “hydrotrope,” subtly loosening interactions, increasing water content, and shifting the permittivity of the coexisting phases in different ways. These changes could be detected sensitively by the dye, even when conventional composition measurements would be difficult.
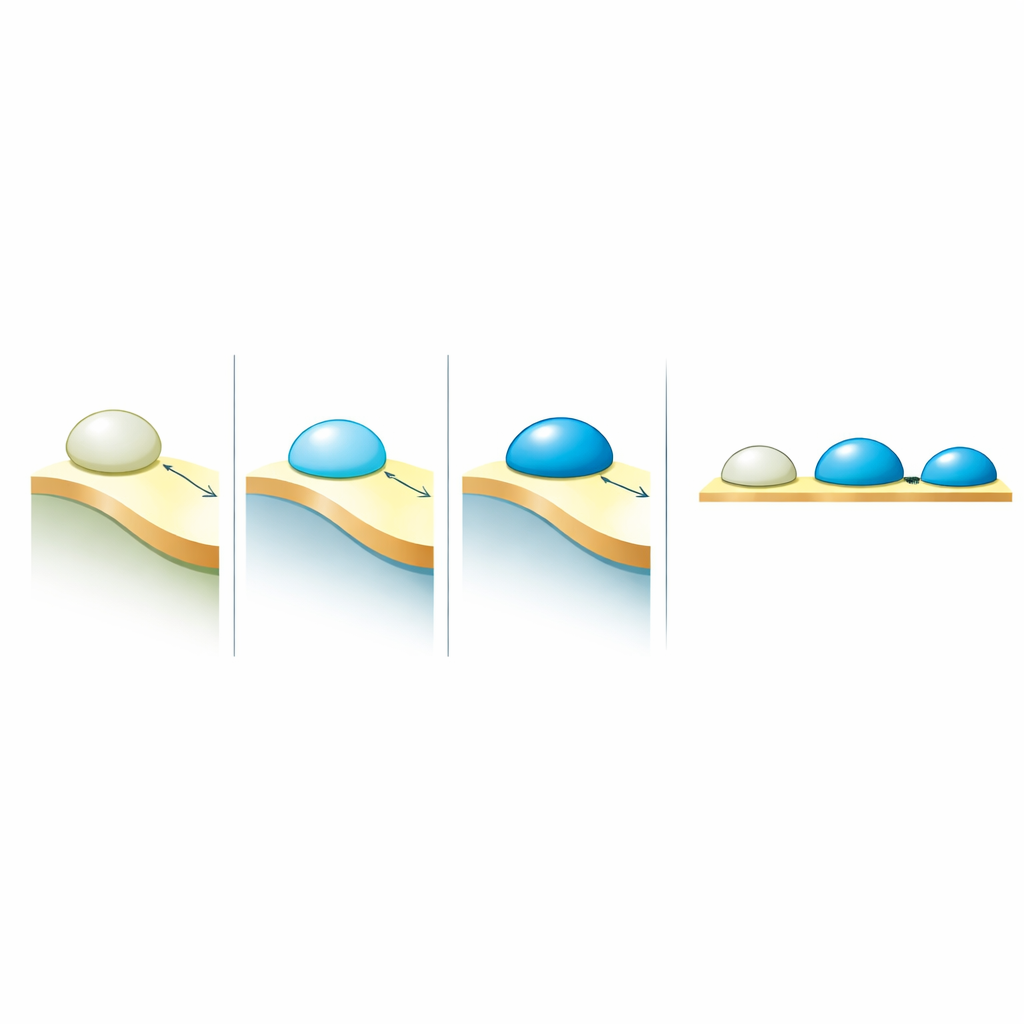
How Droplets Decide to Wet Membranes
The key biological question was how these electrical properties relate to the way droplets behave at membranes. When a condensate touches a lipid membrane, it can barely attach, partially spread, or strongly wet and reshape the membrane. This behavior is captured by a contact angle—a geometric measure of how much the droplet spreads. By comparing many systems from earlier experiments, the authors found a simple rule: the degree of wetting increases linearly with the permittivity contrast between the dense condensate and the surrounding dilute phase. In other words, it is not the absolute “polarity” of the droplet that matters most, but how different it is from what surrounds it. Larger contrasts produce stronger attraction to membranes, while smaller contrasts weaken that affinity.
Why This Matters for Cells and Disease
These findings reveal a unifying physical principle for how soft droplets inside cells interact with membranes: membrane affinity is set by the dielectric mismatch between coexisting phases, rather than by condensate properties alone. Because molecules like ATP can tune this contrast by altering local water structure, cells may use such small solutes to regulate where condensates form, which membranes they stick to, and how they help repair or reshape those membranes. The mapping method introduced here, which is compatible with live-cell imaging, offers a new window into the hidden electrical landscapes of condensates and may help explain how changes in crowding, hydration, and droplet aging contribute to health and disease.
Citation: Sabri, E., Mangiarotti, A. & Dimova, R. Fluorescence-based mapping of condensate dielectric permittivity uncovers hydrophobicity-driven membrane interactions. Nat Commun 17, 3155 (2026). https://doi.org/10.1038/s41467-026-71273-7
Keywords: biomolecular condensates, dielectric permittivity, membrane wetting, fluorescence imaging, cellular phase separation