Clear Sky Science · en
Modulating coordinate site occupancy in high-entropy spinel electrocatalysts
Why this new material matters for clean energy
Splitting water into oxygen and hydrogen with electricity is a cornerstone of many clean energy technologies. But this process, especially the step that produces oxygen, wastes a lot of energy because current catalysts are not efficient enough. This study explores a new way to build better catalysts from "high-entropy" oxides—materials that mix many different metals into one crystal—and shows how carefully placing specific atoms inside that crystal can make water splitting faster, more stable, and less energy-hungry.
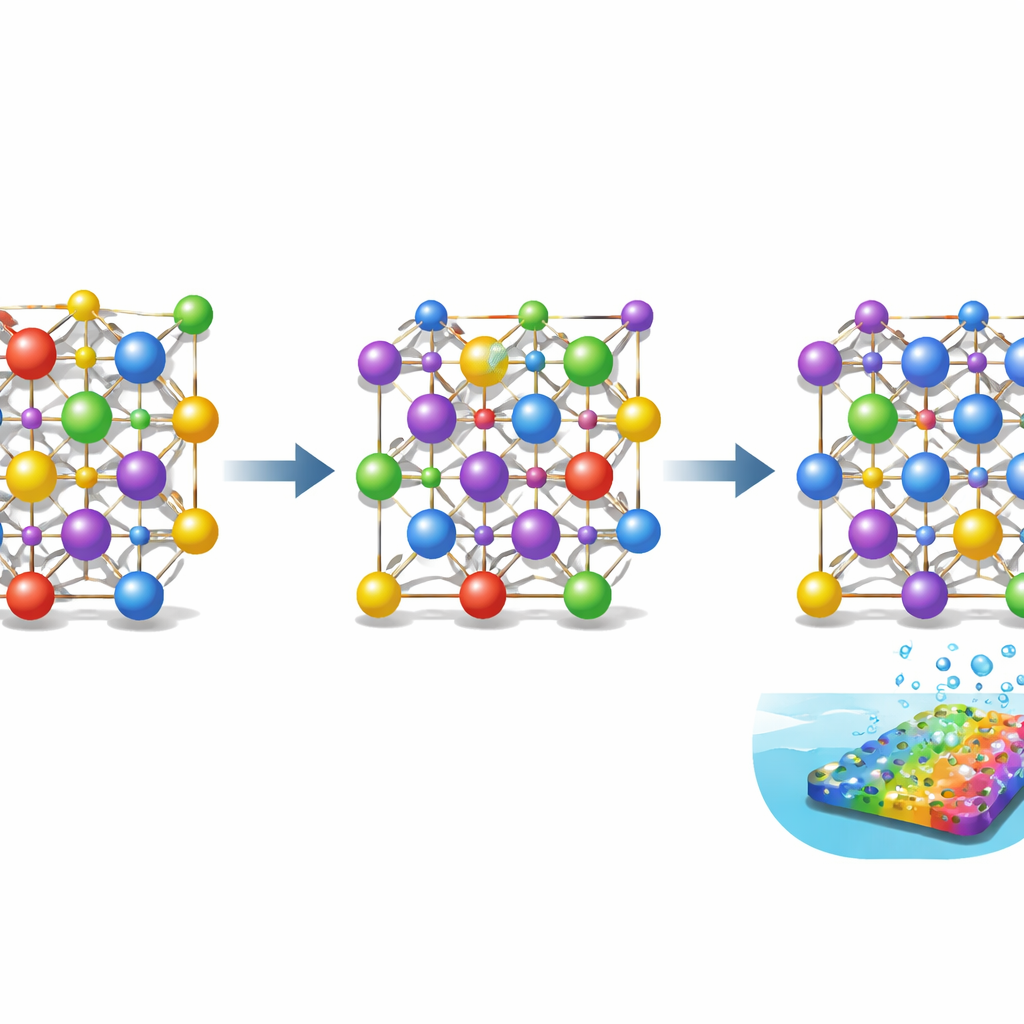
Building order inside highly mixed materials
High-entropy materials are like alloys on overdrive: instead of one or two metals, they can contain five, six, or more, all mixed together at nearly equal amounts. In this work, the researchers focus on a spinel oxide, a crystal structure with two kinds of pocket-like sites for metal atoms—tetrahedral and octahedral. Traditionally, scientists assumed that when several metals are mixed to form a high-entropy spinel, the different atoms simply spread out evenly. Recent evidence, however, shows that even if the overall mix is even, the local arrangement can be quite uneven, with certain metals preferring one type of pocket over the other. That hidden pattern matters, because it strongly affects how electrons move through the material and how well it drives chemical reactions.
A targeted extra ingredient that reshapes the lattice
To gain control over these atomic positions, the team started from a five-metal spinel made of cobalt, iron, chromium, manganese, and nickel. They then added a sixth metal—zinc, gallium, magnesium, or aluminum—chosen because each has a known preference for either the tetrahedral or octahedral pockets. By introducing, for example, zinc, which strongly favors tetrahedral sites, they could gently push cobalt atoms out of those sites and into octahedral ones instead. Advanced electron microscopy and X-ray measurements at synchrotron facilities confirmed that the overall crystal structure stayed intact, but the detailed distribution of atoms between the two types of pockets shifted in systematic ways. Zinc in particular increased the number of cobalt atoms sitting in octahedral positions while keeping the material structurally stable.
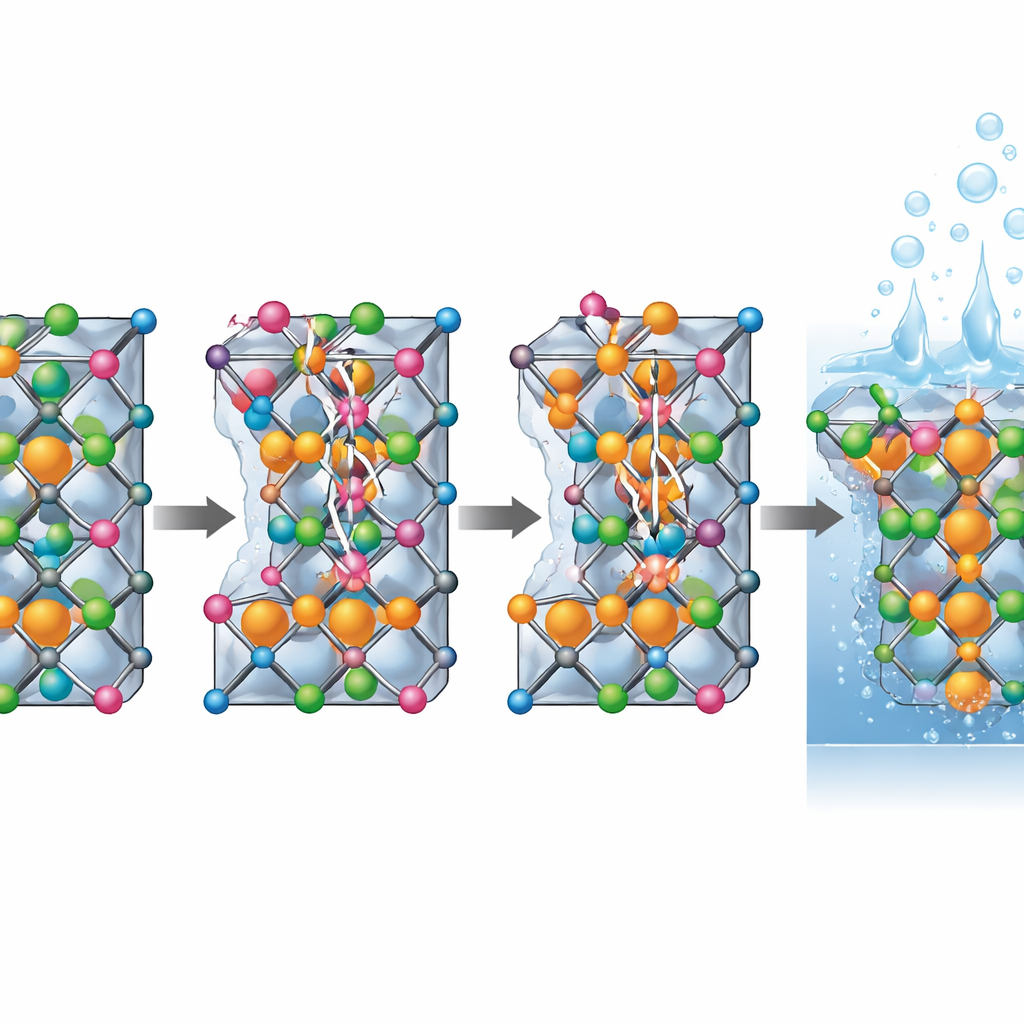
Following electrons and reactions at the atomic scale
The researchers combined experiments with quantum mechanical simulations to understand why this rearrangement matters. Calculations showed that zinc truly prefers the tetrahedral pockets and most stably replaces cobalt there, nudging cobalt into octahedral positions. Further modeling of the oxygen evolution reaction—the step where water is turned into oxygen gas—revealed that cobalt in specific octahedral states provides the most active sites for the reaction. These sites bind reaction intermediates just strongly enough to speed the process without trapping them. Electronic structure calculations also showed that when more cobalt sits in these octahedral sites, especially in a particular charge state, the material becomes more conductive, allowing electrons to flow more easily during operation.
Putting the new catalysts to the test
To see if this atomic rearrangement translates into real-world performance, the team tested thin films of the different high-entropy spinels in alkaline solution. The zinc-containing version stood out: it required less extra voltage to drive oxygen production at both modest and high current levels and showed the smallest kinetic penalty, as reflected in its low Tafel slope. It also conducted electricity better than the undoped material and maintained its performance for many hours of continuous operation, unlike the reference catalyst, which slowly degraded. Additional "operando" X-ray measurements—taken while the catalyst was actually working—showed that cobalt and, to a lesser extent, nickel atoms change their oxidation states as the voltage is increased, forming active oxyhydroxide-like species. Some of these changes are partially retained after cycling, hinting at a self-activation process supported by the carefully tuned local environment.
What this means for better water-splitting devices
This study shows that in complex multi-metal oxides, performance is not just about which elements are present, but exactly where they sit in the crystal. By using a sixth, site-selective metal like zinc to steer cobalt into the most favorable positions, the researchers created a more efficient, more conductive, and more durable oxygen-evolving catalyst. For non-specialists, the key takeaway is that subtle atomic "seat assignments" inside a crowded lattice can have an outsized impact on how well a material converts electricity into chemical fuel. The work points to coordination site occupancy—a measure of which atoms sit in which pockets—as a powerful design knob for next-generation electrodes in water-splitting and other clean energy technologies.
Citation: Baek, J., Hamkins, K.S., Li, Y. et al. Modulating coordinate site occupancy in high-entropy spinel electrocatalysts. Nat Commun 17, 3540 (2026). https://doi.org/10.1038/s41467-026-70982-3
Keywords: water splitting, oxygen evolution reaction, high-entropy oxides, spinel electrocatalyst, atomic site engineering