Clear Sky Science · en
Electrochemical tyrosine-click bioconjugation enables multiplexed cytokine sensing and immunoprofiling in native serum
Why tiny signals in blood matter
Doctors and scientists often want to read the body’s immune signals from just a few drops of blood, so they can track infections, cancer, or the effects of pollution in real time. These signals are carried by small proteins called cytokines, but measuring several of them at once in untreated blood is hard. The sensors used today often lose activity, give unstable readings, or take hours to prepare. This study introduces a quick surface chemistry trick that helps build better electrochemical sensors, making it easier to profile immune responses directly in complex samples like native serum.
A new way to anchor proteins on electrodes
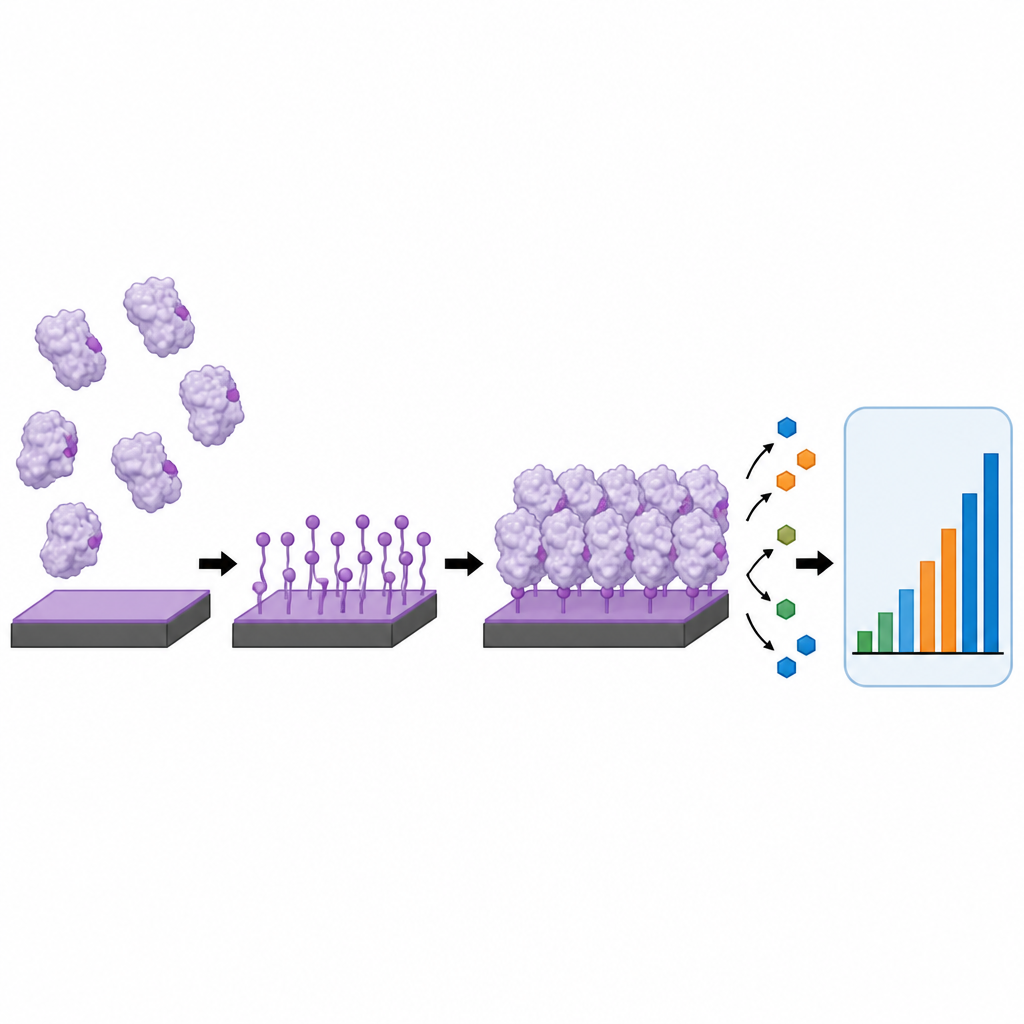
At the heart of many electrochemical biosensors is a flat electrode coated with a carefully arranged layer of proteins, such as antibodies or enzymes. How these proteins are attached greatly affects sensor performance. Common methods, like simple sticking or slow chemical coupling, often leave proteins in random orientations, form clumps, or wash off over time. The authors developed an approach they call interfacial electrochemical tyrosine click, or i-eY-Click, which uses a mild electrical signal to activate a thin film on a carbon surface. This activated film then reacts selectively with tyrosine building blocks on the outside of proteins, forming a thin, dense, and stable protein layer in under three minutes without needing extra reagents or genetic modification.
Fast, gentle chemistry that keeps proteins working
The team first confirmed that this tyrosine-focused reaction really was fast, selective, and robust. They showed that the special film on the electrode could be switched into its reactive form at a gentle voltage that does not harm proteins. Compared with a standard amide coupling method, i-eY-Click attached molecular probes about twenty times faster and reached higher coverage in just a few minutes than the older chemistry did in an hour. Microscopy and surface measurements revealed that the new method produced smooth, uniform layers that stayed in place even after harsh washing, while proteins that were only physically adsorbed formed uneven patches and were easily removed. Importantly, tests with several enzymes showed that their activity and substrate sensitivity were largely preserved after this electrical “click” attachment.
Turning better chemistry into better immune sensors
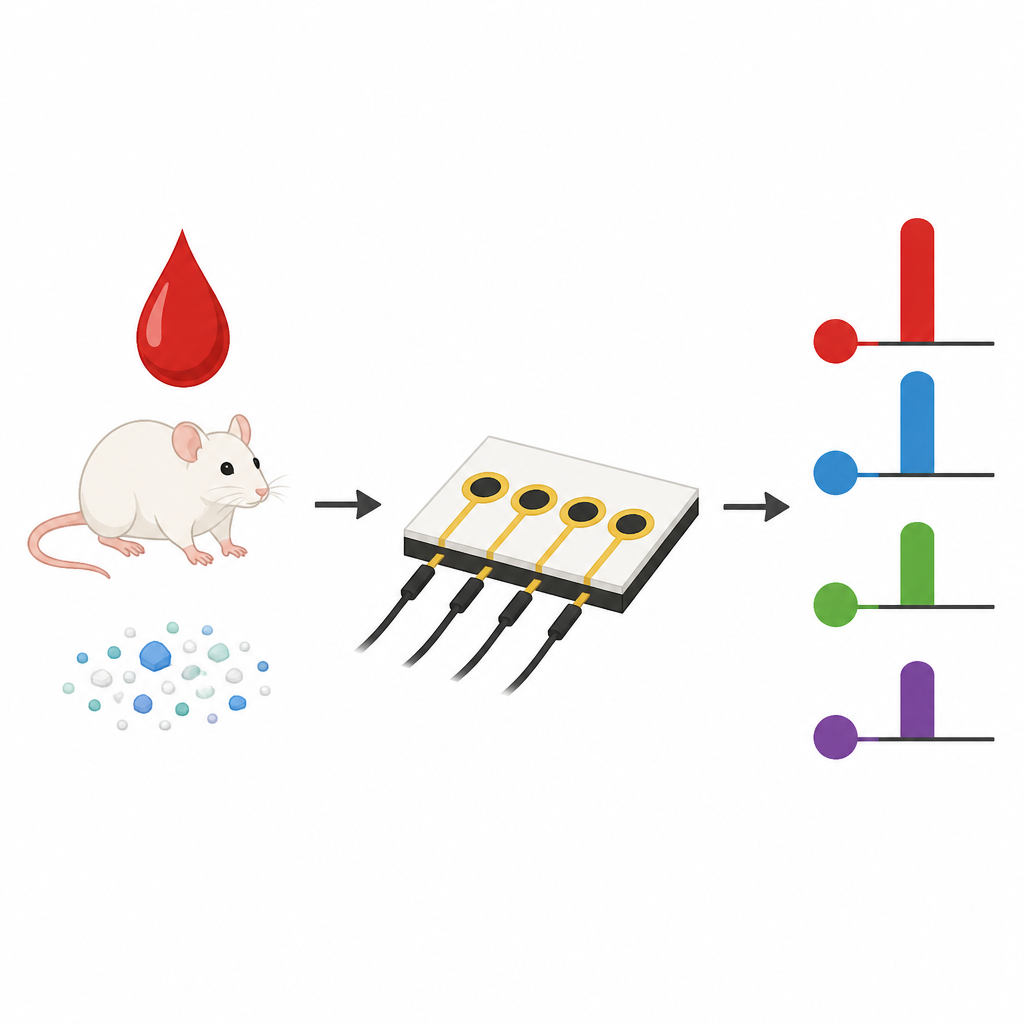
Building on this surface chemistry, the researchers fabricated small chips carrying arrays of carbon microelectrodes. Each spot on the array was coated by i-eY-Click with a different antibody that recognizes a specific inflammatory cytokine, including IL-6, IL-1β, TNF-α, and IFN-γ. When a small blood sample is added, the target cytokines bind to their matching antibodies, and a standard enzyme signal step converts this binding into an electrical current. Compared with sensors made using the traditional coupling chemistry, the new chips needed only minutes to prepare, showed stronger signals, and detected cytokines at lower concentrations. In undiluted mouse serum, they delivered higher sensitivity, lower detection limits, and much better reproducibility from spot to spot and from batch to batch, crucial features for practical diagnostic use.
Watching the body respond to nanoplastics
To demonstrate real-world value, the authors used their platform to monitor how the immune system of mice responded over time to different nanoplastic particles, including polylactic acid and charged polystyrene beads. After a single injection, they collected tiny serum samples at several time points and measured the four cytokines in parallel. The chips revealed distinct time courses of cytokine changes that depended on particle surface charge and degradability. Some particles triggered brief, moderate spikes in inflammatory markers, while positively charged or degradable particles were associated with stronger or more prolonged responses. These patterns were consistent with conventional laboratory tests and with tissue imaging that showed differing levels of brain damage and inflammation.
What this means for future health and environmental monitoring
This work shows that carefully designed surface chemistry can unlock more reliable and sensitive electrochemical biosensors. By using a quick, tyrosine-targeted electrical click reaction, the authors create durable, well-organized protein layers that support multiplexed cytokine measurements in tiny volumes of native serum. While the nanoplastic study is exploratory, it illustrates how such chips could be used to track immune responses to materials, disease, or therapies over time. In the long run, this approach could help power compact, affordable devices that read immune signals at the bedside or in the field, providing clearer snapshots of how the body reacts to its environment.
Citation: Song, K., Liu, Y., Ma, Q. et al. Electrochemical tyrosine-click bioconjugation enables multiplexed cytokine sensing and immunoprofiling in native serum. Nat Commun 17, 4251 (2026). https://doi.org/10.1038/s41467-026-70815-3
Keywords: electrochemical biosensor, cytokine sensing, protein immobilization, nanoplastic exposure, immunoprofiling