Clear Sky Science · en
DRP1 induces neuroinflammation via transcriptional regulation of NF-ĸB.
Why this brain inflammation story matters
Many brain disorders, including Alzheimer’s and Parkinson’s disease, are now known to be linked to chronic inflammation in the brain. This study uncovers a surprising new player in that process: a protein called DRP1, previously famous for shaping mitochondria, also turns out to help switch on inflammatory genes. Understanding this hidden role opens up new ways to think about calming harmful brain inflammation without shutting down essential immune defenses.
A hidden switch inside brain immune cells
Our brains contain resident immune cells called microglia and support cells called astrocytes. When these cells sense trouble, they release chemical signals that can protect or damage nearby neurons. The researchers used mice and cultured brain cells exposed to a bacterial component known as LPS, a common experimental trigger for inflammation. They focused on DRP1, a protein best known for cutting mitochondria into smaller pieces, and asked whether it also affects the way microglia and astrocytes respond to inflammatory challenges.
DRP1 makes inflammation genes louder
By partially reducing DRP1 levels in mice, the team found that the usual surge of inflammatory genes after LPS exposure was markedly blunted, especially for a gene called Lcn2, which encodes the protein lipocalin-2. Using precision techniques to collect single cells from mouse brains, they showed that microglia and astrocytes are the main sources of this lipocalin-2 burst, with dopamine-producing neurons contributing to a lesser degree. In mice overproducing the Parkinson’s-linked protein α-synuclein, lipocalin-2 was also elevated, and again this rise was normalized when DRP1 was reduced, suggesting a broader connection to neurodegenerative conditions.
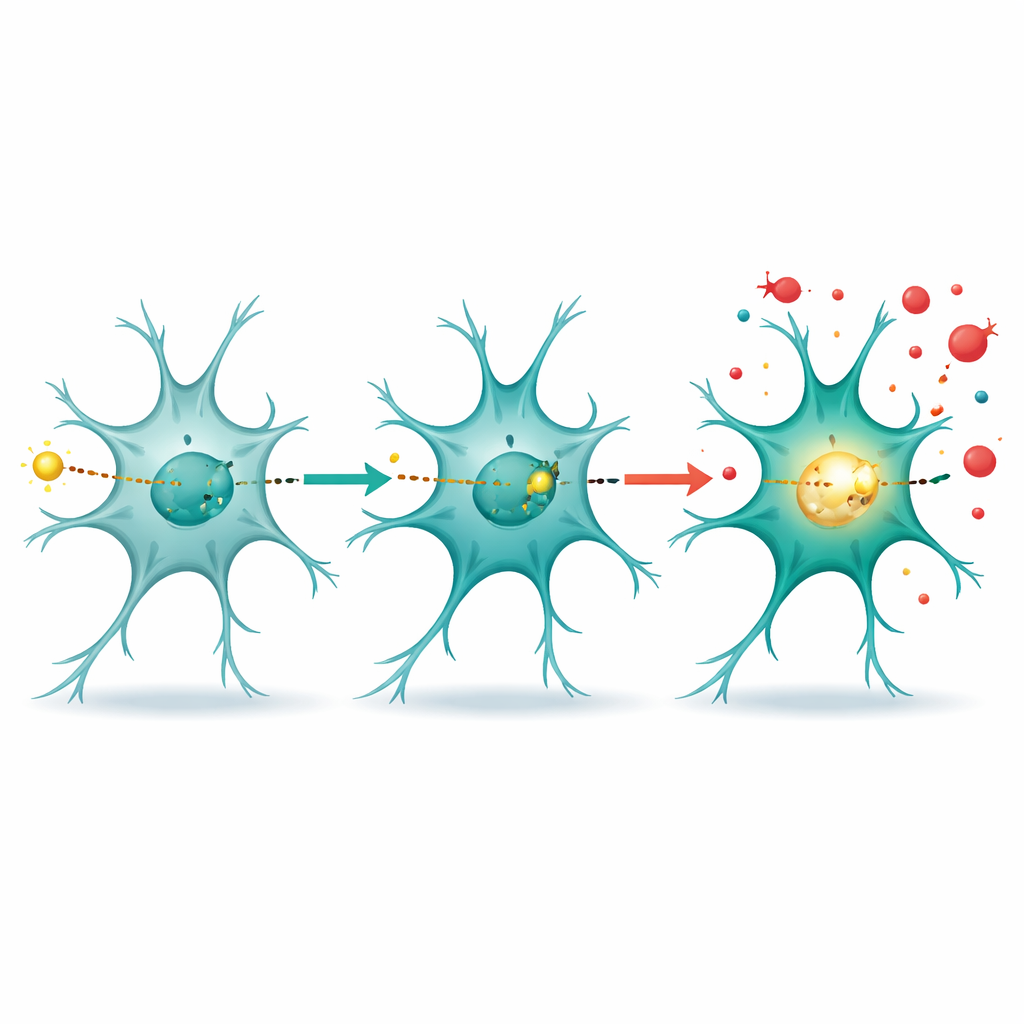
A surprising role in the cell nucleus
The most unexpected discovery came when the scientists looked at where DRP1 travels inside cells. After inflammatory stimulation, DRP1 moved from the cell fluid into the nucleus, the compartment that houses DNA. There, DRP1 bound to a specific stretch of DNA that controls the gene Rela, which produces the p65 subunit of NF-κB, a master switch for inflammation. Reporter assays showed that this DNA region became more active when DRP1 was present and less active when DRP1 was knocked down, indicating that DRP1 acts like a transcription factor, directly boosting NF-κB production and, in turn, many downstream inflammatory genes.
Lipocalin-2 as an amplifier and decoy
Because lipocalin-2 was the most highly induced NF-κB target in this study, the team examined its role more closely. When they removed or reduced the Lcn2 gene in microglia, LPS could no longer trigger the same strong wave of inflammatory molecules. Intriguingly, in cells completely lacking the functional lipocalin-2 gene, NF-κB became trapped at the inactive Lcn2 promoter and was less available to activate other inflammatory genes. In effect, the missing gene acted as a decoy, soaking up NF-κB and dampening the overall response. These results suggest that lipocalin-2 both amplifies inflammation when present and can, when deleted, divert NF-κB away from other harmful targets.
Inflammation without damaged mitochondria
Since DRP1 is famous for its role in mitochondrial fission, the team tested whether the protective effects of lowering DRP1 were simply due to healthier mitochondria. They carefully measured mitochondrial shape and energy production in microglia after LPS treatment and found no lasting damage under their conditions, even though inflammation was strong. Early, brief changes in mitochondrial shape resolved without functional problems. This indicates that in this model DRP1 drives brain inflammation largely through its nuclear gene-regulating role rather than through mitochondrial injury.
What this means for future brain therapies
Overall, the study reveals that DRP1 is more than a mitochondrial sculptor: it is also a nuclear switch that helps turn on NF-κB and a powerful inflammatory axis centered on lipocalin-2. By showing that lowering DRP1 or disrupting the lipocalin-2 arm of this pathway can soften inflammatory responses, the work points to new strategies for selectively cooling harmful brain inflammation that contributes to diseases like Parkinson’s and Alzheimer’s, while leaving beneficial immune functions more intact.
Citation: Lai, Y., Fan, R.Z., Brown, H.J. et al. DRP1 induces neuroinflammation via transcriptional regulation of NF-ĸB.. Nat Commun 17, 4032 (2026). https://doi.org/10.1038/s41467-026-70780-x
Keywords: neuroinflammation, microglia, NF-kappaB, lipocalin-2, Parkinsons disease