Clear Sky Science · en
Photoinduced radical-mediated atomic dispersion of noble metal nanoparticles
Turning Precious Metals into Super-Efficient Catalysts
Noble metals like palladium and platinum sit at the heart of many modern technologies, from cleaning car exhaust to making medicines and plastics. But these metals are rare and expensive, and much of the metal in conventional catalysts is locked away inside tiny particles where it cannot work. This study describes a gentle light-driven method that can break those particles down into individual atoms, dramatically boosting how efficiently each precious metal atom is used and offering a greener way to upgrade and even recycle industrial catalysts.
Why Breaking Metal Particles Matters
Traditional catalysts often use nanoparticles of noble metals dispersed on an oxide support. Only atoms on the surface of each particle actively participate in reactions; atoms buried inside are essentially wasted. Scientists have long pursued “single-atom catalysts,” where every metal atom is exposed and anchored on a support, maximizing performance per atom. Existing ways to create such materials typically demand high temperatures, special gas atmospheres, or complicated chemical treatments. These approaches are energy-intensive, costly, and sometimes hard to scale. A simple, low-temperature route that could convert existing nanoparticle catalysts into single-atom versions would therefore be highly attractive for industry.
Using Light as a Gentle Transformer
The authors show that ultraviolet light can drive this transformation under everyday conditions. They disperse palladium nanoparticles supported on titanium dioxide (TiO2) in a very dilute hydrochloric acid solution in acetonitrile, then shine UV light at room temperature. Before illumination, electron microscopy clearly reveals clustered palladium particles on the TiO2 surface. After one hour of light exposure, the particles visually disappear, yet chemical analysis shows that palladium is still present and is now spread uniformly across the surface. Advanced imaging that can “see” single atoms confirms that the metal has been converted into isolated palladium atoms anchored to the oxide. Spectroscopic measurements tracking how carbon monoxide binds to the surface also shift from signatures of particles to those characteristic of single atoms, supporting this structural change. 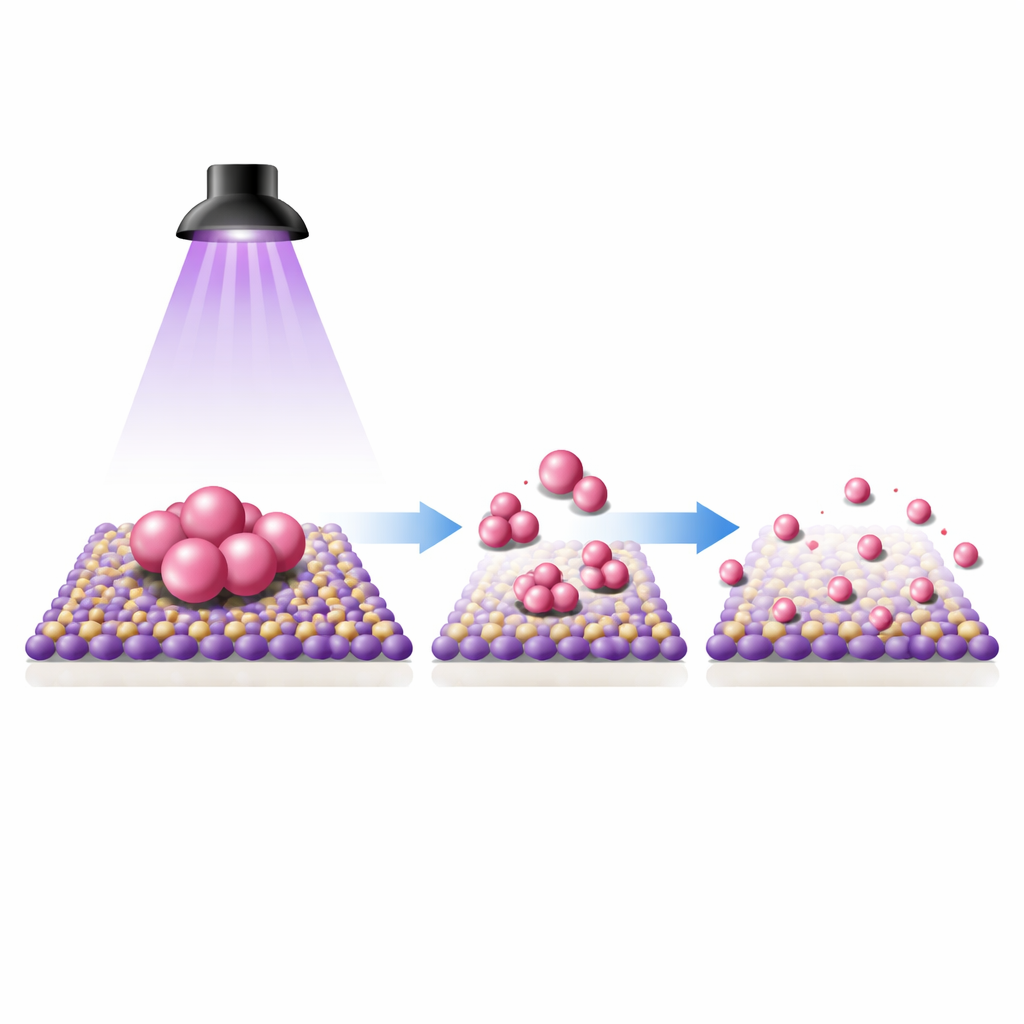
How Radicals Do the Heavy Lifting
To understand how this light-driven reshaping occurs, the researchers probe the chemical species formed during illumination and run computer simulations based on quantum mechanics. When UV light hits TiO2, it creates energetic electrons and “holes” that migrate to the surface and react with the surrounding solution. Oxygen captures electrons to form superoxide radicals, while acetonitrile and chloride ions react with holes to generate organic radicals and chlorine radicals. Experiments that selectively quench these short-lived species show that both chlorine and superoxide radicals are essential for breaking the bonds holding palladium atoms together in nanoparticles. Simulations reveal that chlorine radicals first attach to the metal, drawing away electron density and weakening palladium–palladium bonds. Superoxide then attacks, and chloride from the solution coordinates the freed atoms to form a mobile intermediate complex. This complex is electrostatically attracted to the positively charged TiO2 surface, where it sheds chloride and, with help from organic radicals and nearby nitrogen and oxygen sites, locks into place as a single palladium atom bound in a stable local environment.
From Lab Mechanism to Versatile Tool
Once the mechanism was clarified, the team tested how broadly the strategy applies. They showed that similar light treatments can convert not only palladium but also platinum and rhodium nanoparticles into single atoms on TiO2, and that the process also works on another oxide support, tungsten oxide (WO3). Larger palladium particles prepared by a conventional chemical reduction route could likewise be broken down under longer UV exposure. Most importantly for practical use, commercial palladium-on-carbon catalysts and even industrial waste catalysts that had lost activity were successfully rejuvenated: after treatment alongside TiO2 under UV light, palladium became atomically dispersed and the catalysts’ performance in a standard hydrogenation reaction jumped by nearly 18-fold for commercial material and 26-fold for waste material. These upgraded catalysts also remained highly active over repeated cycles, and the authors demonstrate that the process can be run in a continuous-flow setup and even driven by sunlight. 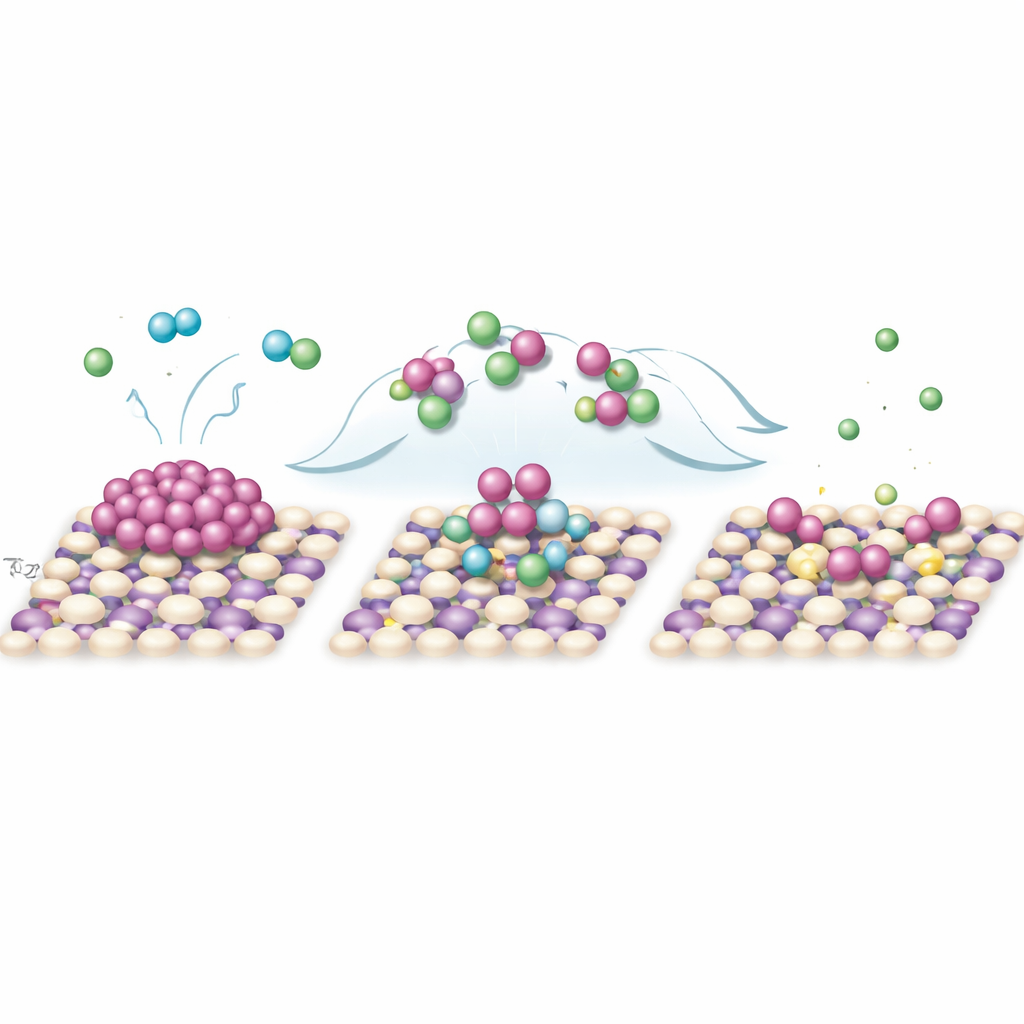
A Simple Path to Greener Catalysis
For non-specialists, the key message is that shining light on a carefully chosen mixture can reorganize precious metals from clumps into individually anchored atoms, without the need for high heat or harsh conditions. This light-powered “unclumping” greatly increases how effectively each expensive metal atom is used and can breathe new life into spent industrial catalysts. Because the method works for several important metals, different supports, and real-world materials, it offers a promising, environmentally friendly route to more efficient and sustainable catalytic processes in the chemical and energy industries.
Citation: Chen, X., Zhao, Q., Zhang, J. et al. Photoinduced radical-mediated atomic dispersion of noble metal nanoparticles. Nat Commun 17, 3934 (2026). https://doi.org/10.1038/s41467-026-70742-3
Keywords: single-atom catalysts, photocatalysis, noble metal nanoparticles, green chemistry, catalyst regeneration