Clear Sky Science · en
Heterogeneous endocrine cell composition defines human islet functional phenotypes
Why tiny clusters in the pancreas matter
Diabetes affects millions of people, yet scientists are still uncovering how the tiny cell clusters in the pancreas that control blood sugar differ from person to person. This study takes a deep look at those clusters, called islets, in hundreds of organ donors without diabetes to see how their internal makeup varies and how that variation might shape future diabetes risk and treatment.
Looking closely at human islets
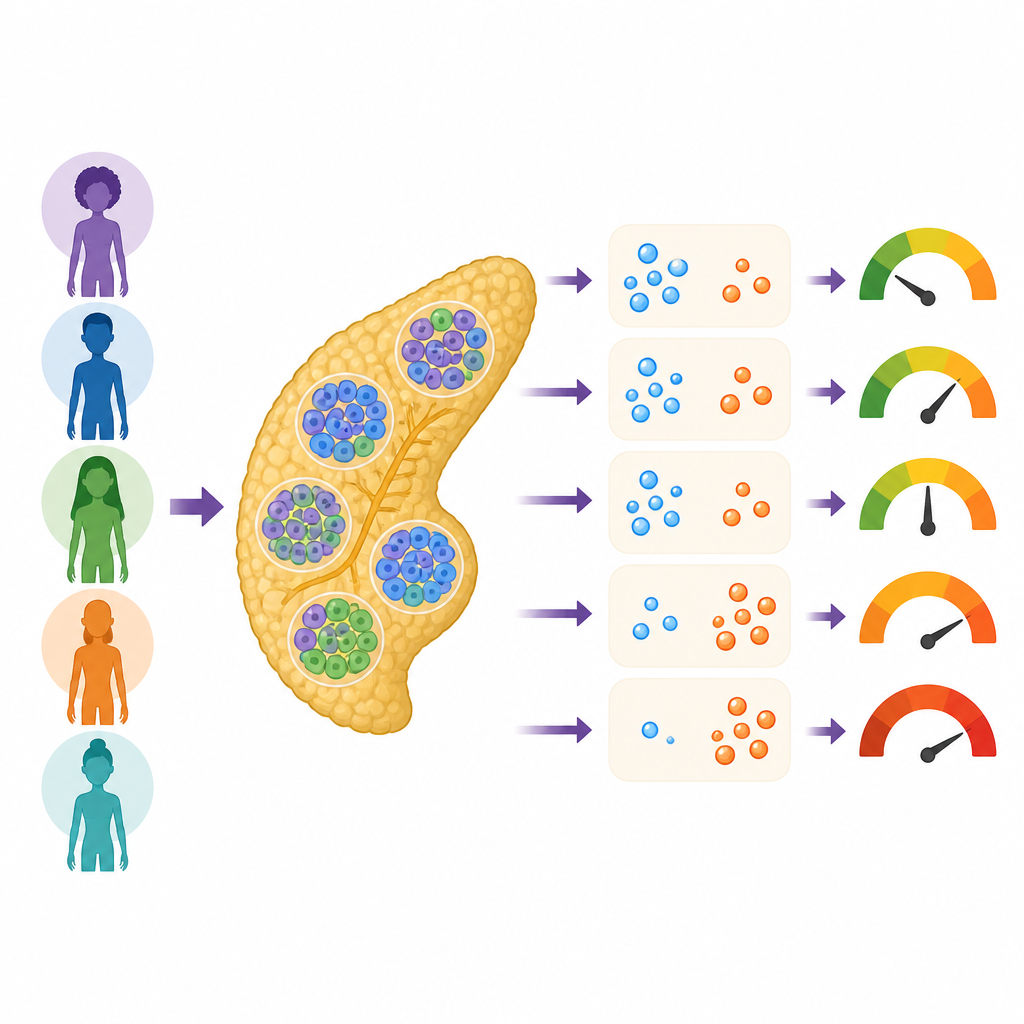
The researchers worked with the Integrated Islet Distribution Program, a national network that collects human pancreatic islets from organ donors and sends them to scientists. For 299 donors without diagnosed diabetes, the team combined three kinds of tests: how well the islets released hormones, what their cells looked like under the microscope, and what the donors’ DNA revealed about ancestry and genetic risk for diabetes. These islets came from men and women of different ages, body sizes, and reported racial and ethnic backgrounds, creating a rich snapshot of human diversity.
Big differences in hormone release
Each islet contains several types of endocrine cells, including beta cells that release insulin to lower blood sugar, alpha cells that release glucagon to raise it, and delta cells that release somatostatin to put the brakes on their neighbors. When the team exposed the islets to changing sugar levels and other chemical signals, they saw striking donor-to-donor differences in how much insulin and glucagon was released. Some of this variation related to familiar traits such as body mass index and long-term blood sugar levels, but these factors explained only part of the picture.
The surprise role of the rare delta cells
Under the microscope, the scientists measured what fraction of each islet was made up of beta, alpha, and delta cells. On average, about 58 percent of the endocrine cells were beta cells, 34 percent were alpha, and just 8 percent were delta, though the mix varied widely among donors. As expected, more beta cells generally meant stronger insulin release, and more alpha cells meant more glucagon. The surprise was how much influence the small delta cell population had. Islets with a higher percentage of delta cells tended to release less insulin and showed weaker responses across several tests, even after taking donor age, sex, body size, and processing conditions into account.
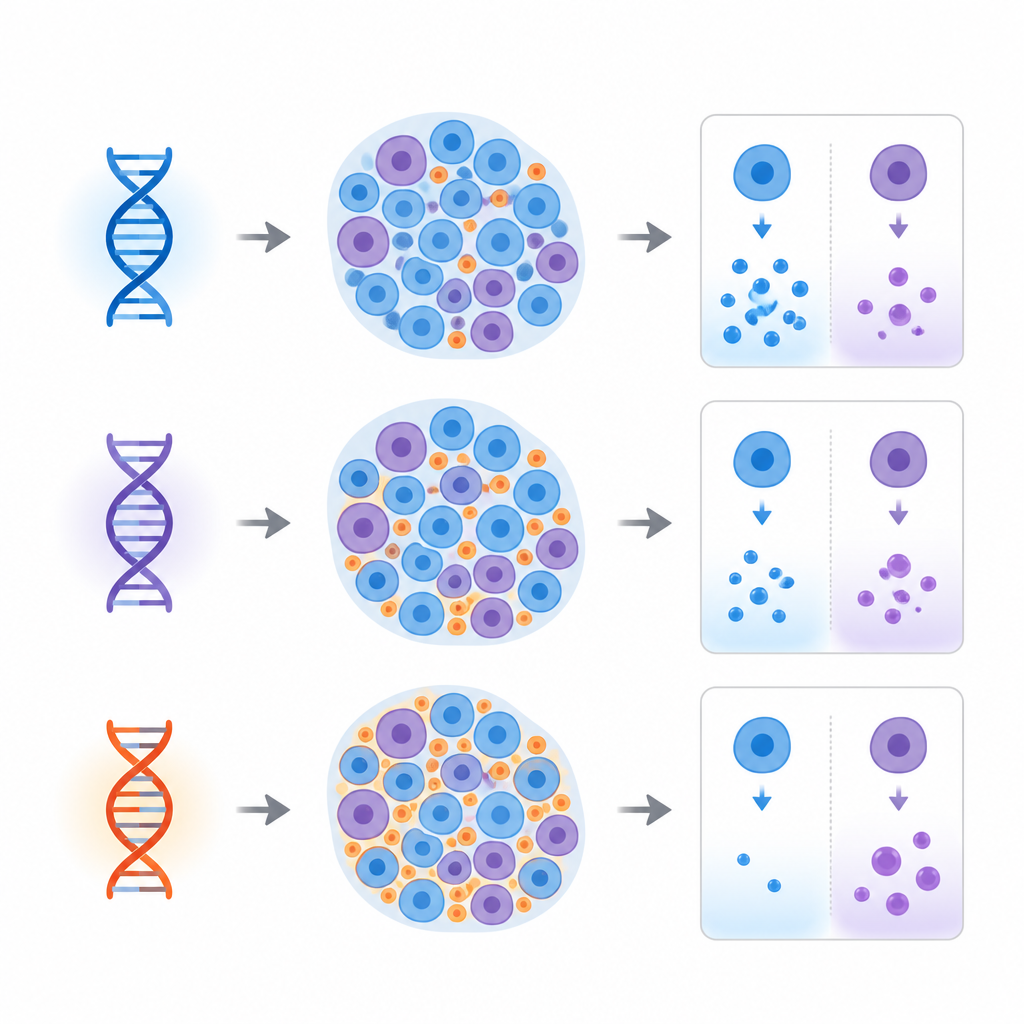
Linking cell mix to ancestry and genetic risk
The team then asked whether these cell mixtures and hormone patterns lined up with ancestry and inherited diabetes risk. Using DNA data, they predicted each donor’s genetic ancestry and calculated risk scores based on many known genetic variants for type 1 and type 2 diabetes. They found that islet composition was tied to both reported race or ethnicity and genetically predicted ancestry. For example, donors with East Asian ancestry tended to have relatively more beta cells and fewer alpha cells. Most notably, people with a higher genetic risk score for type 2 diabetes tended to have a larger share of delta cells in their islets. Additional analysis of single-cell gene activity from another dataset showed that many genes linked to type 2 diabetes are especially active in delta cells, strengthening the link between this rare cell type and future diabetes risk.
What this means for diabetes care
For a layperson, the key idea is that not all pancreatic islets are built the same, and those differences are connected to our genes, background, and how our bodies handle blood sugar. Even a small shift in the balance between beta, alpha, and delta cells can change how much insulin is available and how tightly blood sugar is controlled. This work suggests that paying attention to islet cell composition, especially the often overlooked delta cells, could improve how scientists interpret lab studies, design beta cell replacement therapies, and understand why diabetes looks different from one person to another.
Citation: Evans-Molina, C., Pettway, Y.D., Saunders, D.C. et al. Heterogeneous endocrine cell composition defines human islet functional phenotypes. Nat Commun 17, 4223 (2026). https://doi.org/10.1038/s41467-026-70689-5
Keywords: human islet cells, insulin and glucagon, delta cells, type 2 diabetes risk, genetic ancestry