Clear Sky Science · en
Ligand engineering tailors hydrophobic microenvironments for efficient electrocatalytic oxidation of fatty alcohol
Turning Grease into Green Chemistry
Everyday products from soaps and shampoos to lubricants rely on fatty alcohols—oily molecules that are notoriously hard to process cleanly in water. Traditional methods to turn these alcohols into more useful fatty acids often need harsh chemicals and precious metals. This study shows how clever molecular design can sidestep those drawbacks, using electricity and a smartly tailored catalyst to transform greasy molecules in water more efficiently and sustainably.
Why Oily Molecules Are So Hard to Tame
Fatty alcohols do not like water, and most industrial electrochemical systems run in water-based solutions. That mismatch means the oily reactants tend to hover far from the solid catalyst surface, slowing the reaction and giving oxygen formation a head start. Standard nickel hydroxide catalysts work well for small, water-loving alcohols like methanol but struggle with long-chain fatty alcohols. The result is sluggish conversion, wasted electrical energy, and a lot of unwanted oxygen gas instead of the desired fatty acids.
Borrowing a Trick from Nature’s Enzymes
Nature has already solved a similar problem. Enzymes such as chymotrypsin use clusters of aromatic amino acids to build “hydrophobic pockets” that attract oily molecules and hold them at just the right spot for reaction. Inspired by this idea, the researchers built nickel-based metal–organic frameworks (Ni‑MOFs), where nickel atoms are linked by organic molecules bearing one, two, or three benzene rings. By stepping up the number of rings, they could tune how water-repelling and structurally robust the catalyst’s internal environment would be, aiming to create enzyme-like pockets on an inorganic scaffold.
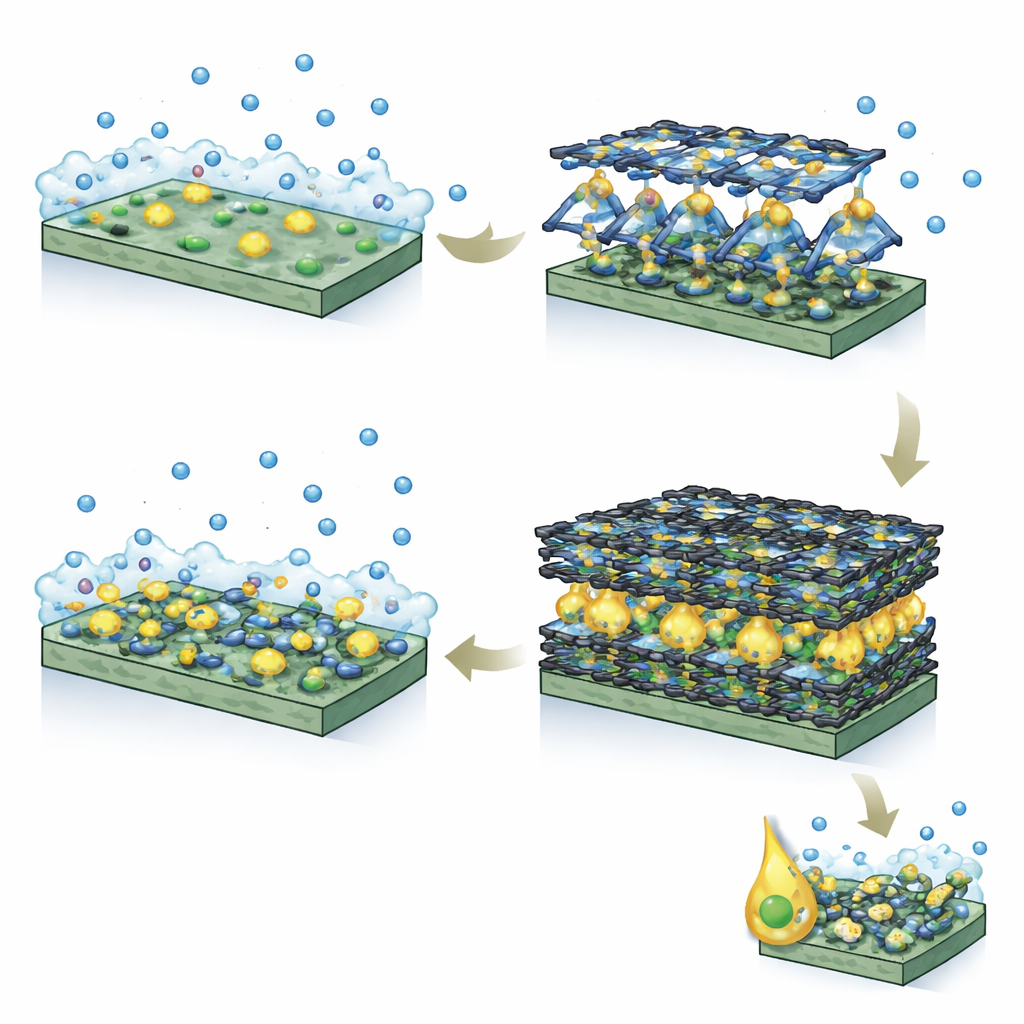
Designing a Better Microscopic Home for Fatty Alcohols
The team synthesized three layered Ni‑MOFs and compared them with conventional nickel hydroxide. They found that the version with the longest ligand, built from a terphenyl (three-ring) unit called Ni‑TPDC, created the most hydrophobic surroundings. Experiments measuring how octanol droplets spread and how much oily dye the materials absorbed showed that Ni‑TPDC attracted roughly twice as much fatty alcohol as nickel hydroxide. Computer simulations backed this up, revealing that octanol molecules clustered strongly against the Ni‑TPDC surface due to enhanced van der Waals attractions in its aromatic-lined channels.
Keeping the Catalyst’s Framework Intact
A common weakness of MOF-based electrodes is that they fall apart under the harsh conditions needed for oxidation reactions, losing their carefully designed internal architecture. Here, detailed X-ray and vibrational spectroscopy revealed a crucial difference among the three Ni‑MOFs. In the materials with shorter ligands, the organic linkers were stripped away during operation, and the structure collapsed into an amorphous nickel hydroxide-like phase. In contrast, Ni‑TPDC retained its layered crystalline framework even after many activation cycles and hours of electrolysis. The extra benzene ring enabled strong stacking between neighboring ligands, acting like a set of interlocking tiles that hold the lattice together while only the outer surface partially converts into the active nickel oxyhydroxide layer.
Faster Reactions, Fewer Side Products
When the researchers used octanol as a test fatty alcohol, Ni‑TPDC dramatically outperformed standard nickel hydroxide. At relevant voltages, it delivered about three times the production rate of octanoic acid while maintaining a Faradaic efficiency above 80%, compared with roughly 30% for nickel hydroxide, where oxygen formation dominates. Careful analysis showed that Ni‑TPDC did not simply have more active nickel sites or a much larger surface area; instead, its hydrophobic microenvironment fed fatty alcohols to the reaction zone more effectively, easing mass transport bottlenecks. The optimized catalyst completed full conversion of octanol to octanoic acid in just 3.5 hours with essentially 100% selectivity, reaching production rates competitive with state-of-the-art thermal oxidation methods that typically rely on precious metals.
From Lab Concept to Practical Energy Systems
To test real-world potential, the authors integrated Ni‑TPDC into a flow cell where fatty alcohol oxidation at the anode is paired with hydrogen generation at the cathode. Because oxidizing octanol is easier than splitting water to oxygen, the overall cell voltage dropped by nearly 0.2 volts at practical current densities, reducing the energy cost of hydrogen production while simultaneously making valuable fatty acid. The system ran stably for 48 hours, with high yields of both hydrogen and octanoic acid. For non-specialists, the takeaway is that by designing a catalyst’s tiny surroundings to be more “oil-friendly” yet structurally tough, it is possible to convert greasy molecules cleanly in water using renewable electricity, opening a path toward greener manufacturing of everyday chemicals and more efficient hydrogen-powered energy schemes.
Citation: Du, R., Chen, Z., Zhang, B. et al. Ligand engineering tailors hydrophobic microenvironments for efficient electrocatalytic oxidation of fatty alcohol. Nat Commun 17, 3628 (2026). https://doi.org/10.1038/s41467-026-70501-4
Keywords: organic electrocatalysis, fatty alcohol oxidation, hydrophobic catalysts, metal-organic frameworks, green hydrogen