Clear Sky Science · en
Motor cortex somatostatin interneurons adaptively shape the structure of action sequences
Why the Timing of Our Movements Matters
Everyday actions like typing, playing piano, or pouring coffee feel effortless, but they depend on the brain’s ability to link many tiny movements into smooth, efficient sequences. This study asks a deceptively simple question: how does the brain’s motor cortex reorganize these action sequences when we practice, speed up, or change the rules? By watching a special group of inhibitory nerve cells in mice as they learn different lever-press tasks, the researchers reveal that these neurons help fine-tune the timing and structure of complex actions on the fly.
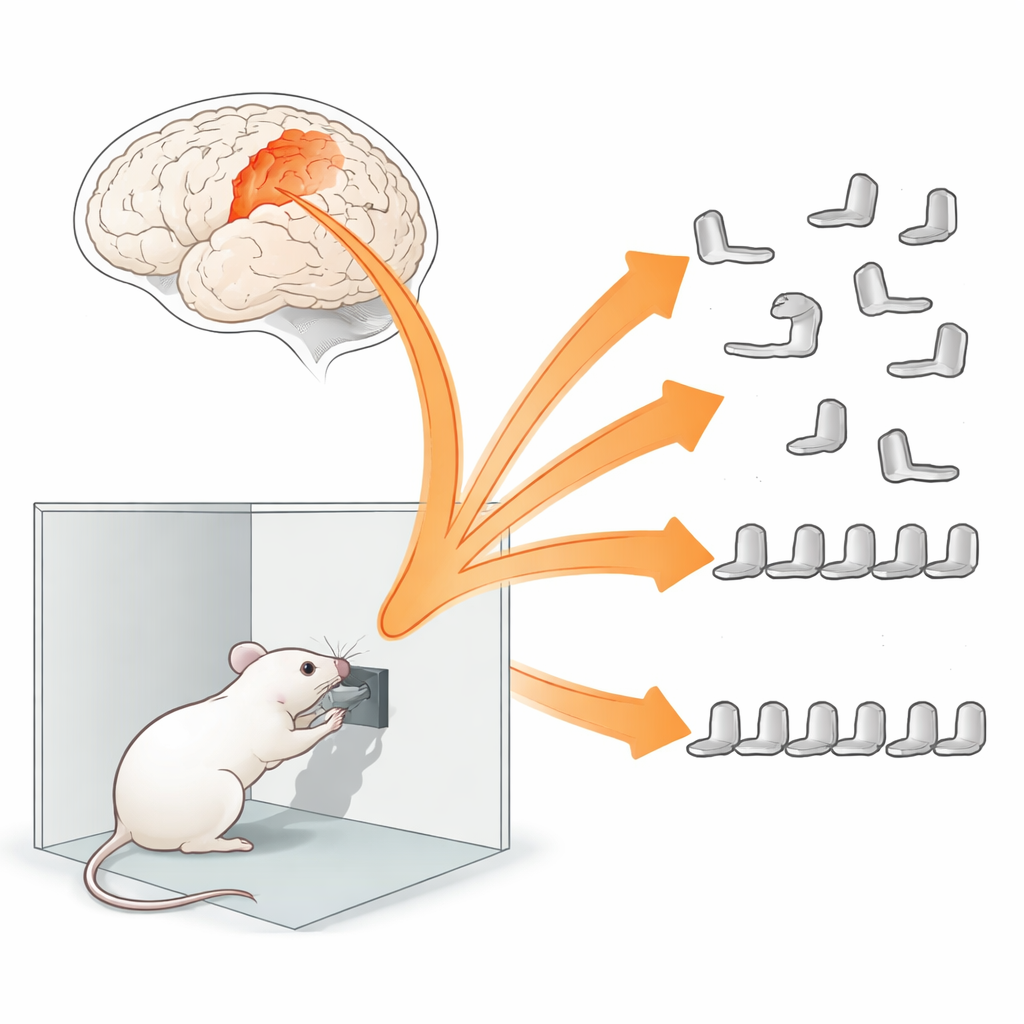
From Single Moves to Chained Actions
To explore how movement sequences are built, the team trained freely moving mice to press a lever for food. At first, the task was simple: one press per reward. Later, the animals had to press four times for each reward, and eventually they had to squeeze those four presses into a narrow time window, forming a fast, tightly timed sequence. While the mice learned, the scientists used tiny microscopes to record calcium signals—a proxy for electrical activity—from specific neurons in the primary motor cortex, the brain area that sends commands to the muscles. They focused on somatostatin interneurons, cells that dampen the activity of nearby excitatory neurons and are thought to regulate learning-related plasticity.
Specialized Cells for Shaping Sequences
During early training on the simple, single-press task, somatostatin interneurons in the deeper layers of motor cortex fired in a highly synchronized, action-locked way: their activity rose reliably around the time of each lever press. By contrast, nearby pyramidal neurons—the main output cells—were activated in a more staggered, sequential pattern. As the single-press behavior became well learned and routine over weeks of practice, the somatostatin cells’ responses shrank and became less correlated, even though their overall ability to generate calcium signals remained intact. This suggests that when a task becomes automatic and unchanging, these interneurons largely disengage from moment-to-moment control of the action.
When the Rules Change, the Network Adapts
The picture changed dramatically when the task demands increased. When mice were required to produce rapid four-press sequences within strict time limits, they reorganized their behavior: presses became faster, more tightly clustered, and more “efficient,” with fewer wasted presses that did not earn rewards. At the same time, somatostatin interneuron activity did not fade; instead, it was redistributed in time and strengthened. The researchers identified two distinct subpatterns in these cells. One group showed a brief, immediate burst around sequence initiation, while another group fired later, with the timing of their peaks tracking how long each particular sequence lasted. More efficient, well-structured sequences were accompanied by larger and more sustained somatostatin signals, and different sequence “classes” with distinct kinematics could be distinguished purely from these neurons’ activity profiles.
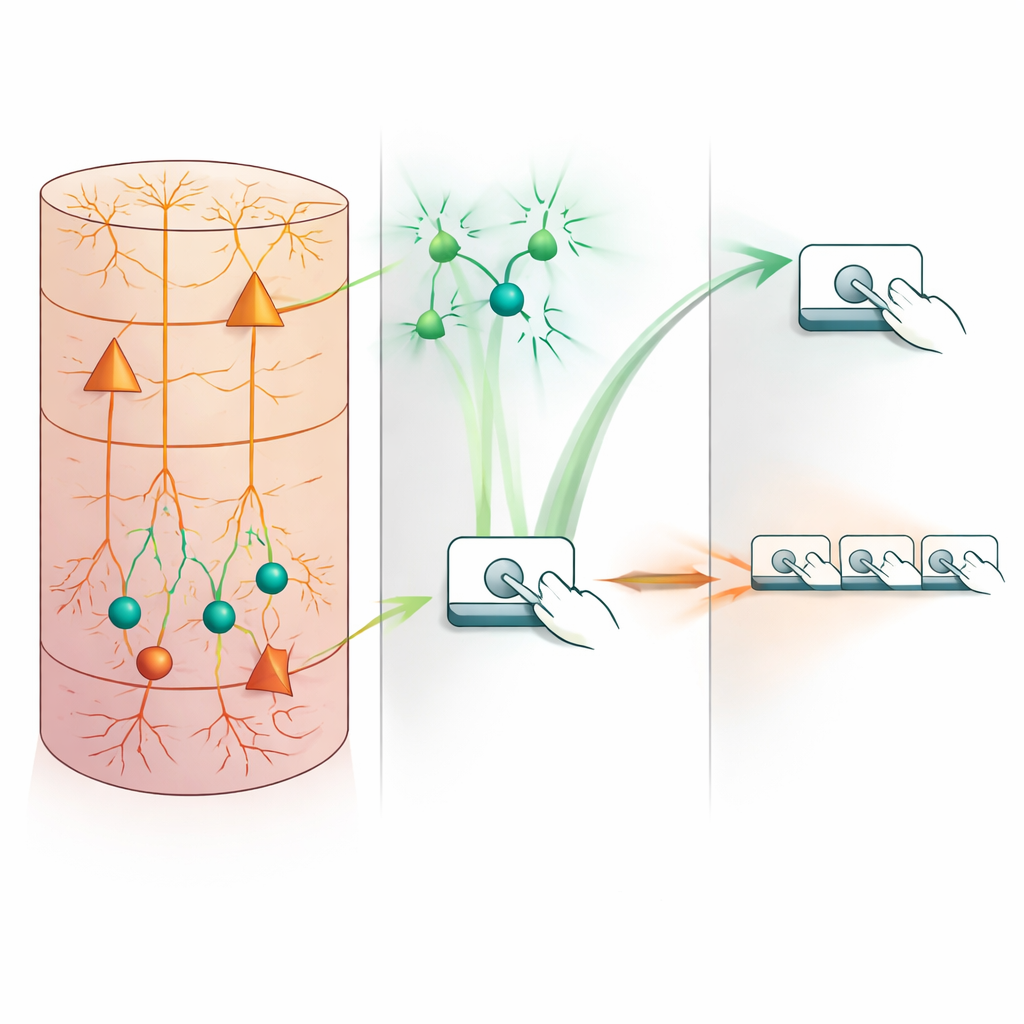
Switching Off the Brakes Scrambles the Rhythm
Correlation alone does not prove cause, so the authors next asked what happens when somatostatin interneurons are deliberately silenced. Using chemogenetic tools and closed-loop optogenetics, they selectively damped these cells in motor cortex while mice performed the fast, time-constrained sequences. In both cases, turning down somatostatin activity caused the animals to press more often within each sequence but in a less organized way: presses were stretched out in time, efficient high-speed sequences became rarer, and “incomplete” sequences that failed to meet the timing requirement became more common. Notably, overall movement or motivation was not simply reduced; in fact, lever-press rates could increase, but the extra presses did not earn more rewards. This points to a specific disruption of temporal organization rather than a loss of drive or strength.
What This Means for How We Move
Taken together, the findings suggest that somatostatin interneurons in deep layers of motor cortex do more than passively modulate activity: they help sculpt the timing and structure of complex action sequences, especially when tasks require speed, precision, or flexible reorganization. When a movement is simple and well practiced, their detailed control can relax. But when the brain needs to compress actions into tight packages or adapt sequences to new rules, these cells come back online to fine-tune when sequences start, how long they last, and how efficiently they achieve the goal. Understanding this circuit-level “timing control” may eventually inform new approaches to treating movement disorders, where actions become slow, fragmented, or poorly sequenced, by targeting not just the strength of motor commands but also the interneuron networks that keep our movements tightly and efficiently organized.
Citation: Lee, J.O., Bariselli, S., Sitzia, G. et al. Motor cortex somatostatin interneurons adaptively shape the structure of action sequences. Nat Commun 17, 4116 (2026). https://doi.org/10.1038/s41467-026-70353-y
Keywords: motor cortex, action sequences, inhibitory interneurons, movement learning, neural timing