Clear Sky Science · en
TYK2 mediates neuroinflammation in Alzheimer’s disease brains with TDP-43 pathology
Why this study matters for brain health
Alzheimer’s disease is usually described in terms of plaques and tangles, but many patients also show a second, quieter kind of damage involving a protein called TDP-43 and persistent brain inflammation. This study digs into how a specific immune switch, TYK2, helps drive that inflammation and nerve cell loss. Because there are already drugs that target this switch in other diseases, the work opens a realistic path toward more personalized, inflammation-focused treatments for a subset of people with Alzheimer’s and related disorders.
Hidden RNA signals in vulnerable brain cells
The authors focused on a strange molecule called double-stranded RNA found in the fluid of nerve cells. Normally, our cells use single-stranded RNA as a working copy of DNA instructions. When double-stranded RNA appears in the wrong place, the cell treats it as a danger signal, similar to a virus. By examining brain tissue from people who had Alzheimer’s disease after death, the team found that this double-stranded RNA builds up specifically in neurons that also contain abnormal clumps of TDP-43 protein in the cell body, rather than in the nucleus where it belongs. This pairing did not overlap with the classic Alzheimer’s hallmarks—amyloid plaques and tau tangles—suggesting an additional damage pathway that affects only certain neurons.
Inflammation turned up across the thinking brain
To see how broad this danger response might be, the researchers analyzed large gene activity datasets from several brain regions involved in memory and thinking. They found that interferon-stimulated genes—genes that are switched on when cells sense viral-like danger—were consistently among the most strongly activated pathways in Alzheimer’s brains compared with healthy brains. Regions that deteriorate early in the disease, such as the parahippocampal gyrus and frontal areas, showed especially strong activation. This pattern supports the idea that neurons are responding to internal distress signals, including double-stranded RNA, and that chronic activation of this antiviral program may contribute to ongoing brain inflammation and degeneration.
Pinpointing who might benefit from existing drugs
Because not all people with Alzheimer’s show the same molecular patterns, the team built a refined machine-learning pipeline to predict which patients might respond to drugs that dampen interferon signaling. They used subtle RNA “stutter marks” called cryptic exons—abnormal extra sequence pieces that appear when TDP-43 fails to do its usual RNA editing job—as a readout of TDP-43 trouble in the brain. Patients whose brain samples contained multiple such cryptic exons were labeled as having TDP-43 pathology. In this subgroup, but not in others, the model predicted that two existing drugs—baricitinib and ruxolitinib, both Janus kinase inhibitors already approved for other conditions—were more likely to slow disease-related molecular changes. This suggests that future clinical trials should target these drugs specifically to patients whose molecular signatures indicate TDP-43-related inflammation.
A central switch: TYK2 and a more precise blocker
To understand the mechanism, the researchers recreated the harmful double-stranded RNA signal in several human neural cell models. They then used a genome-wide CRISPR knockout screen—systematically turning off genes across the genome—to see which losses kept cells alive despite the RNA stress. One kinase, TYK2, emerged as a top hit alongside interferon receptors and regulatory factors, firmly placing it at the center of the toxic response. When TYK2 levels were reduced genetically, cells were largely protected from double-stranded RNA–induced death. The team then tested deucravacitinib, a highly selective TYK2-blocking pill recently approved for psoriasis. Across multiple neuron-like cell types, this drug prevented the damaging response at much lower doses than broader JAK inhibitors, and it shut down downstream inflammatory signaling in protein-level analyses.
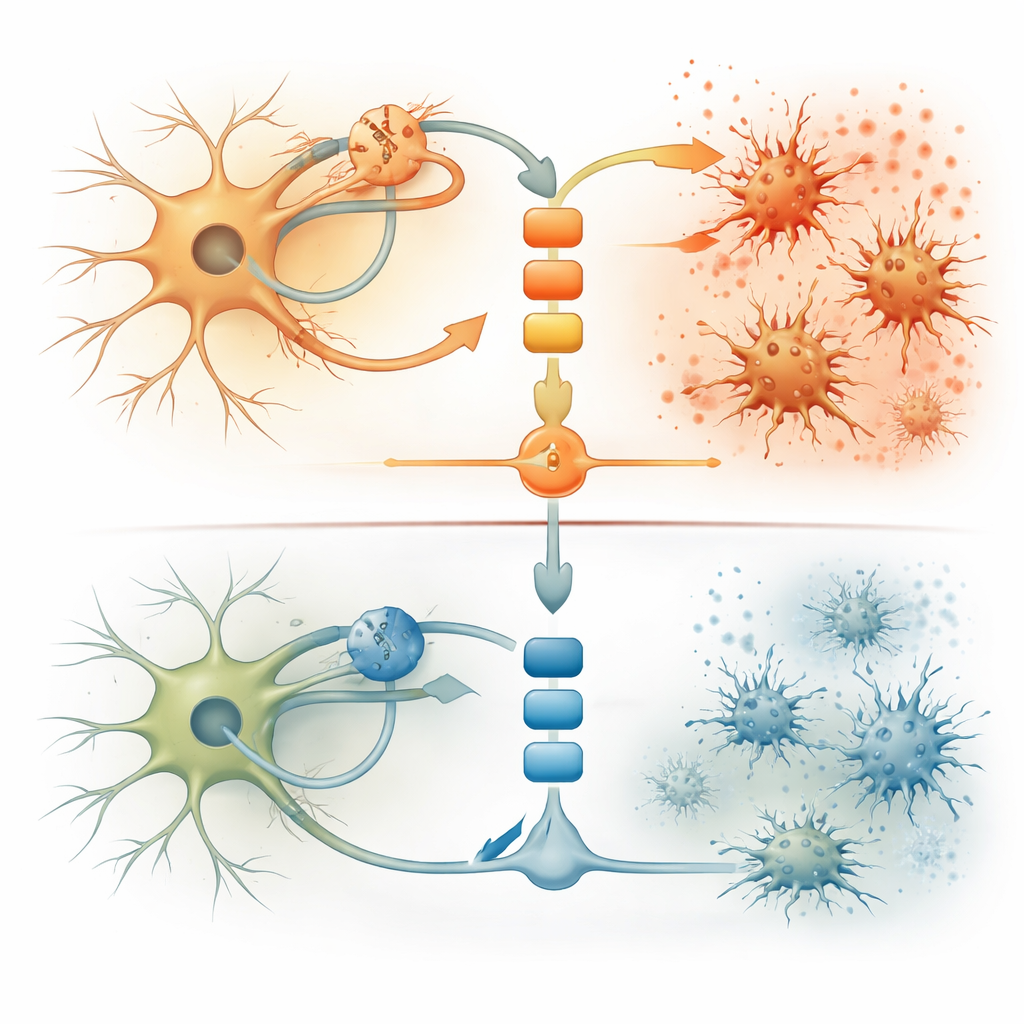
Finding a simple blood clue to a complex brain process
For any future therapy to be tailored to the right people, doctors will need a marker that reflects this particular kind of brain inflammation. The researchers turned to large genetic and blood protein datasets from tens of thousands of volunteers. People who naturally carry a partial loss-of-function variant in the TYK2 gene—essentially a built-in dial-down of this immune switch—had lower blood levels of a chemokine called CXCL10. In their cell models, double-stranded RNA strongly boosted CXCL10 release, while TYK2-targeting drugs brought levels back down. Even when TDP-43 was experimentally pushed out of the nucleus and into clumps, CXCL10 rose and was best normalized by the selective TYK2 inhibitor. This positions CXCL10 in blood or spinal fluid as a promising, testable biomarker for patients whose neurodegeneration is driven by this pathway.
What this means for future treatment
Overall, the study supports a picture in which certain Alzheimer’s brains—and those with related conditions like ALS—develop a slow buildup of double-stranded RNA tied to TDP-43 dysfunction. This, in turn, chronically triggers an antiviral alarm system inside neurons, with TYK2 as a key control knob, ultimately leading to inflammation and cell death. By combining human brain tissue, advanced cell models, CRISPR genetics, large-scale protein measurements, and population data, the authors make a strong case that TYK2 is a viable, disease-modifying target. Although current TYK2 drugs may not yet cross the blood–brain barrier efficiently, the work lays the groundwork for brain-penetrant TYK2 inhibitors and CXCL10-based tests that could usher in more precise, personalized therapies for a defined subgroup of people with Alzheimer’s and TDP-43 pathology.
Citation: König, L.E., Rodriguez, S., Hug, C. et al. TYK2 mediates neuroinflammation in Alzheimer’s disease brains with TDP-43 pathology. Nat Commun 17, 3967 (2026). https://doi.org/10.1038/s41467-026-70243-3
Keywords: Alzheimer’s disease, neuroinflammation, TDP-43, TYK2, double-stranded RNA