Clear Sky Science · en
Sustainable continuous seawater electrolysis using atomic interface catalyst via liquid-medium strategy
Turning Seawater into Clean Fuel
As the world searches for cleaner fuels, hydrogen produced from water using renewable electricity is especially attractive. But large-scale hydrogen plants typically rely on scarce freshwater, and attempts to use seawater directly run into serious technical problems such as clogging, corrosion, and wasteful side reactions. This study presents a practical way to turn ordinary seawater into a steady stream of hydrogen without first removing the salt, using a cleverly designed filter system and a highly efficient catalyst built from carefully arranged metal atoms.
Why Seawater Is So Hard to Use
At first glance, seawater should be an ideal raw material for hydrogen: the oceans hold almost all of Earth’s liquid water. Yet seawater electrolysis—splitting water into hydrogen and oxygen with electricity—behaves very differently from electrolysis of pure water. At the negative electrode, the local buildup of hydroxide ions causes dissolved calcium and magnesium to form stubborn mineral crusts that foul the surface. At the positive electrode, chloride ions can be oxidized to chlorine-containing chemicals that corrode parts and pose safety hazards. Together, these effects make existing seawater devices inefficient, short‑lived, and difficult to scale.
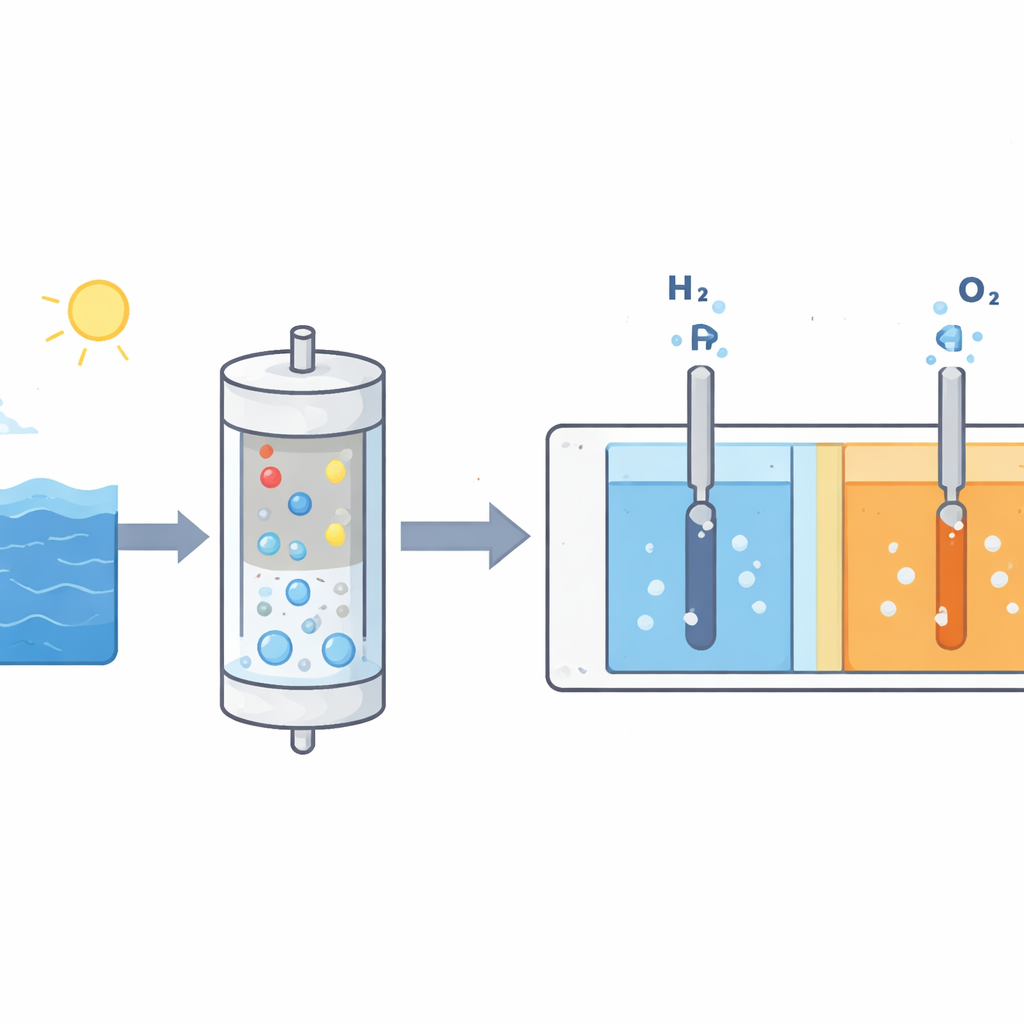
A Two-Stage System That Lets Only Water Through
The researchers built a system that keeps the troublesome salts at arm’s length. Seawater is pumped around the outside of a “balloon filter” module, while a concentrated alkaline electrolyte circulates inside. Only water molecules are allowed to migrate through the thin membrane of the balloon, driven by the ongoing electrolysis in the downstream cell. The salts and other ions in seawater remain outside. The incoming water then enters an anion‑exchange membrane electrolyzer, where it is split into hydrogen and oxygen in a tightly controlled environment. Because the rate at which water crosses the balloon is automatically tied to the rate of gas production, the electrolyte concentration stays nearly constant without complex control systems.
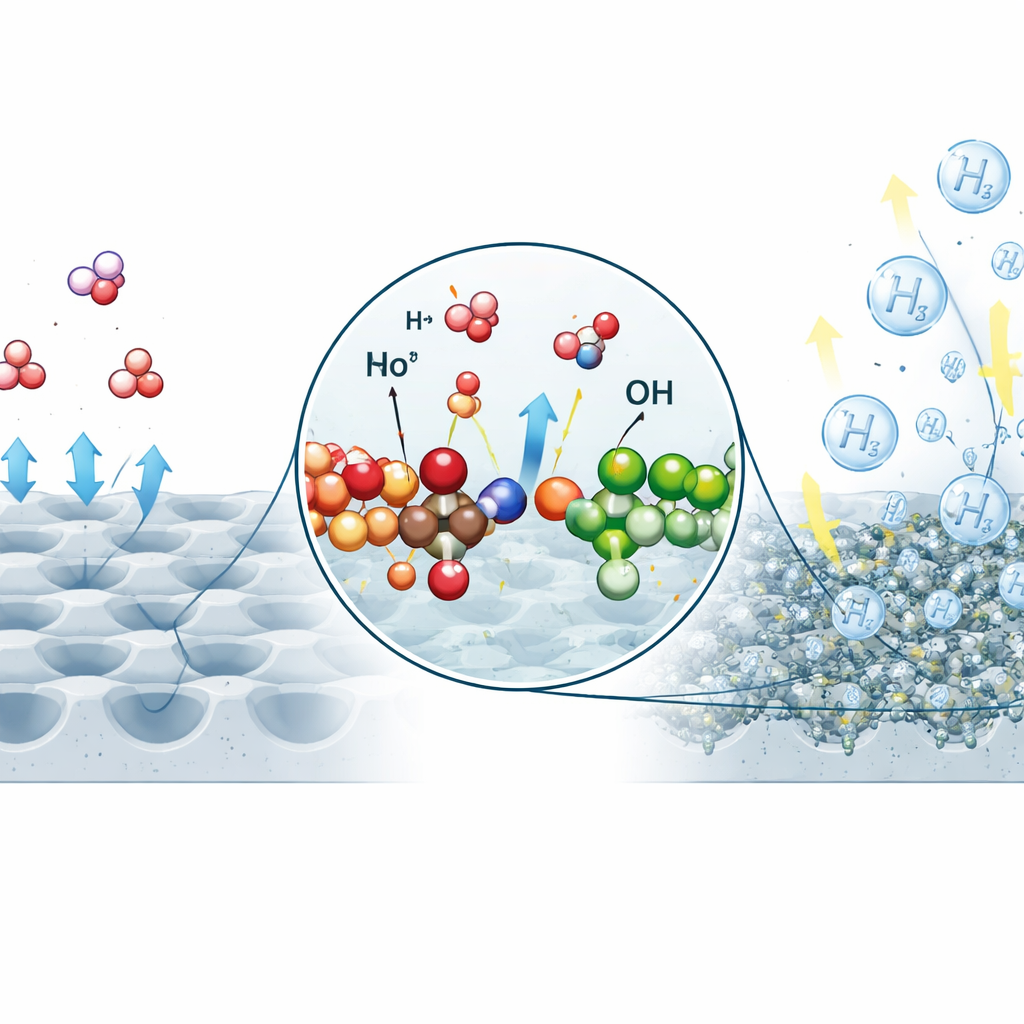
A Catalyst Built Atom by Atom
To make this filtered water split efficiently, the team designed a new catalyst made from nickel and molybdenum oxides arranged at the scale of individual atoms. Using a liquid-based preparation method, they grew a forest of tiny bowl‑shaped wells on a nickel support and then formed countless bridges where nickel atoms connect to molybdenum through oxygen atoms. These Mo–O–Ni bridges give each reaction site a split personality: one side helps break apart water molecules, while the other side helps assemble and release hydrogen gas. Microscopy and advanced X‑ray measurements confirmed the intended structure and showed that the nickel atoms near the bridges adopt an electronic state ideal for speeding up the reaction.
Fast Reactions, Stable Operation
In lab tests, the new catalyst needed only a very small extra voltage to start producing hydrogen and maintained rapid reaction rates even at industrial‑scale currents. Its nano‑rough, highly wettable surface allows gas bubbles to detach quickly, preventing them from blocking fresh water from reaching the active spots. Operando experiments—measurements made while the catalyst is actually working—revealed that the structure remains stable under operating conditions, and that the Mo‑rich regions help weaken water’s internal bonds while the Ni‑rich regions release hydrogen more easily. Computer simulations supported this picture by showing that the atomic bridges lower the energy barriers for both water splitting and hydrogen release compared with simpler materials.
A Long-Lived Seawater-to-Hydrogen System
When combined, the balloon filter and the Mo–O–Ni catalyst formed a seawater electrolysis system that ran continuously at high current for thousands of hours. Tests with real seawater from the Bohai Sea showed that almost no salt ions leaked into the electrolyte, no chlorine‑containing by‑products were detected, and the voltage required to keep the device running increased only slightly over time. In plain terms, the study demonstrates a realistic path to turning abundant seawater into clean hydrogen fuel without first desalinating it, by smartly separating where the salts are and precisely engineering how atoms on a catalyst surface work together.
Citation: Shi, Z., Shi, W., Zhang, C. et al. Sustainable continuous seawater electrolysis using atomic interface catalyst via liquid-medium strategy. Nat Commun 17, 3940 (2026). https://doi.org/10.1038/s41467-026-70234-4
Keywords: seawater electrolysis, green hydrogen, electrocatalyst, nickel molybdenum interface, anion exchange membrane