Clear Sky Science · en
Genetic variations interact with polybrominated diphenyl ether exposure to alter lipid homeostasis
Why everyday chemicals and blood fats matter
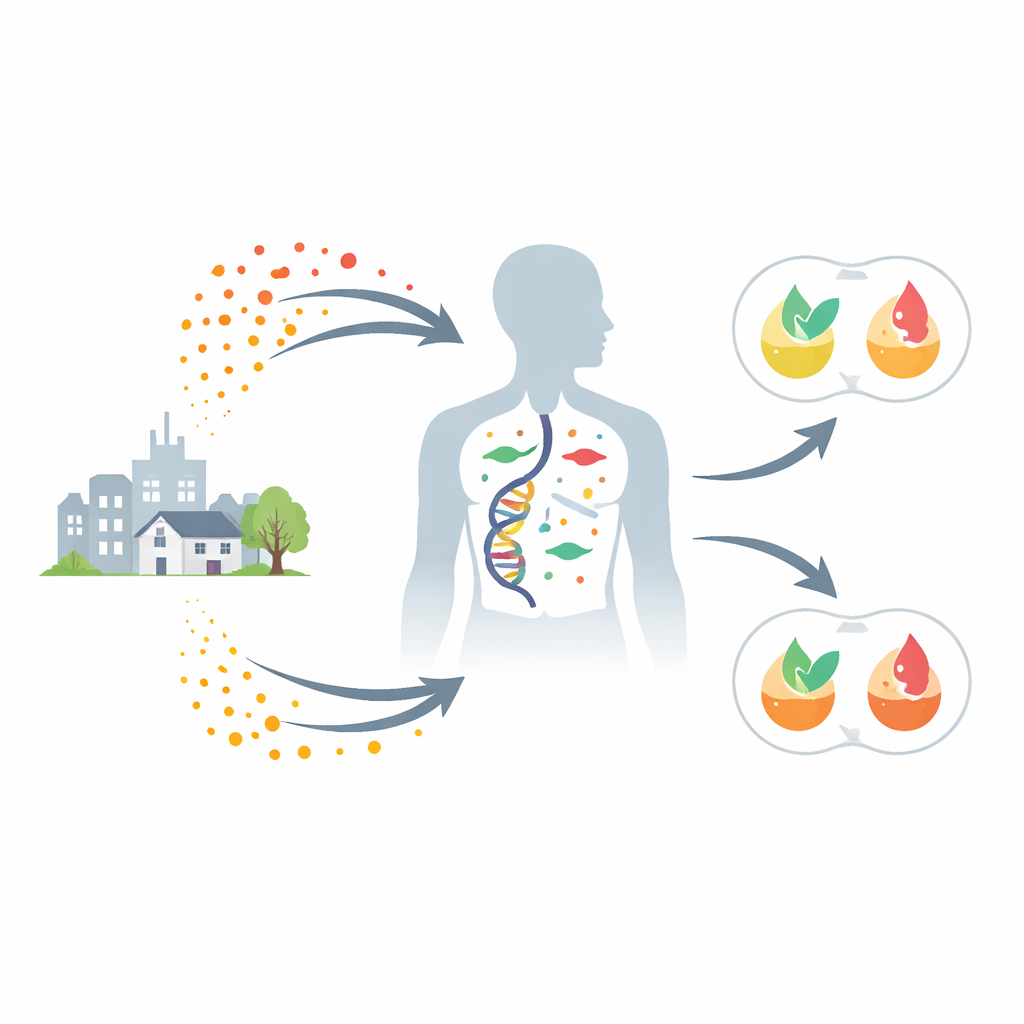
Many common products—from sofas to electronics—contain flame-retardant chemicals called polybrominated diphenyl ethers (PBDEs). These compounds can build up in our bodies and have been linked to unhealthy levels of blood fats such as cholesterol and triglycerides. Yet not everyone exposed to the same amount of PBDEs develops problems. This study asks a question that matters for public health and personal risk: do our genes decide who is most vulnerable to these invisible pollutants?
Unseen pollutants in a nationwide snapshot
Researchers drew on the China National Human Biomonitoring cohort, a large, nationally representative project tracking environmental exposures and health. From more than 21,000 participants, they focused on 871 adults with detailed measurements: PBDE levels in blood, genetic data, hundreds of small molecules (metabolites), and standard blood fats such as total cholesterol, “bad” LDL, “good” HDL, and triglycerides. People were split into high- and low-exposure groups based on their total PBDE levels. Those with higher PBDE exposure tended to have higher total cholesterol and triglycerides and lower HDL, even though age, weight, smoking, and drinking were similar between groups. This pattern suggested that PBDEs do affect lipid balance—but also hinted that something more than exposure alone was at work.
Genes that change how bodies react to PBDEs
To find that “something more,” the team scanned the entire genome for variants that changed the impact of PBDEs on blood fats. They uncovered 3,571 genetic markers across 622 regions of the genome whose effects depended on PBDE exposure. Most of these variants sit in non-coding DNA, where they influence how strongly nearby genes are switched on or off rather than altering the proteins themselves. Many lay in stretches of DNA that act as regulatory hubs, marked by open chromatin and special histone tags, and overlapped binding sites for key transcription factors that help control metabolism. When the researchers combined these PBDE-sensitive variants into polygenic risk scores, they found that people with higher scores showed much worse lipid profiles—but only if their PBDE exposure was high. Under low exposure, the same genetic risk barely mattered, highlighting a powerful gene–environment interaction.
Clues from small molecules in the blood
Next, the scientists asked how these gene–pollutant interactions actually disturb metabolism. Using metabolomics, they examined hundreds of small molecules circulating in blood and looked for those that tracked both with the genetic risk scores and with lipid levels. Several stood out, especially the amino acid glycine, along with citrate, bile acids, and glycerol-3-phosphate. Pathway analysis showed that networks involved in antioxidant defense, bile acid formation, and energy production were heavily affected. Mediation analysis indicated that glycine in particular acts as a crucial middle player: genetic variants that altered lipid levels under PBDE exposure often did so largely by shifting glycine levels. Because glycine is needed to make bile acids that help the body dispose of cholesterol, this pointed to a specific route by which pollutants and genes could team up to raise blood fats.
A single DNA letter that tips the balance
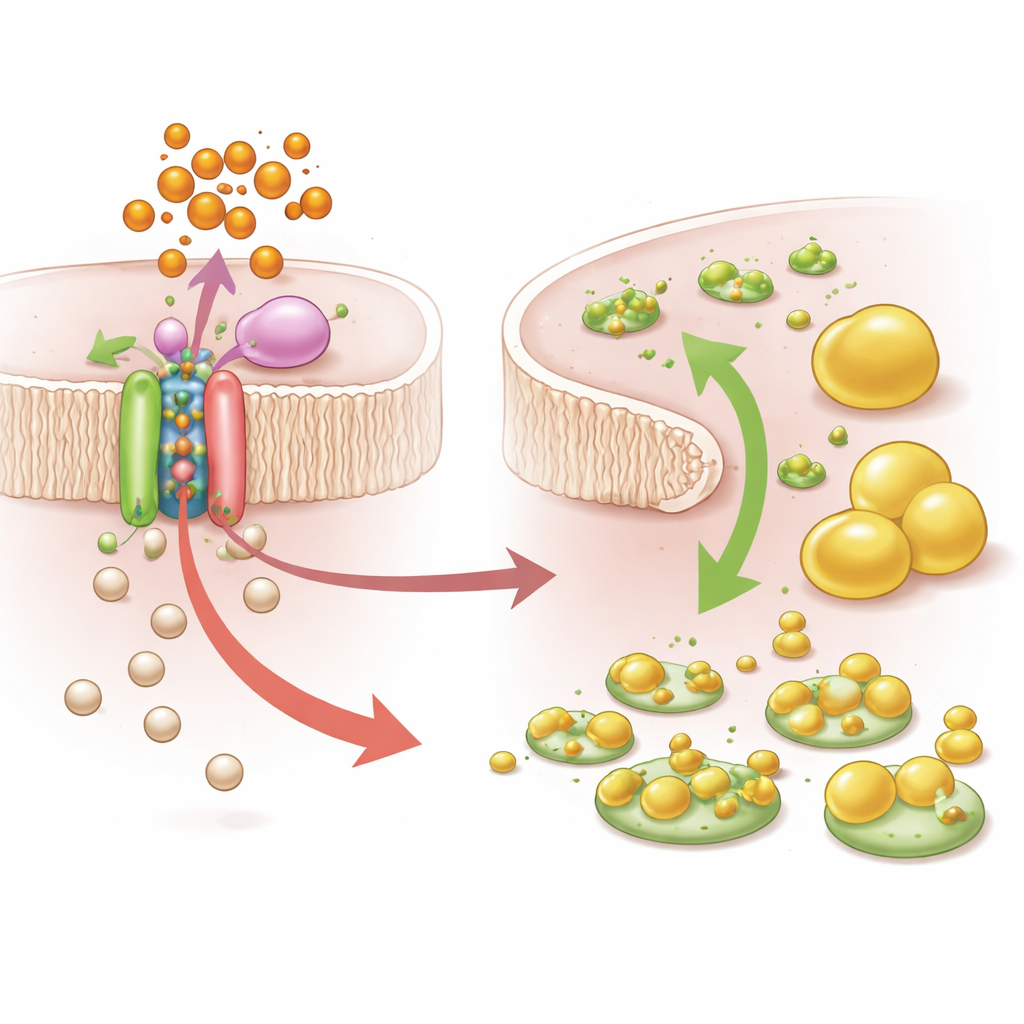
Digging deeper, the team homed in on one genetic variant, rs9869609, strongly linked to PBDE-related increases in total and LDL cholesterol. This variant sits near a gene called SLC6A20, which encodes a transporter that helps move glycine into cells. Using data from human tissues and engineered liver cells, the researchers showed that the “risk” version of rs9869609 boosts the binding of a transcriptional repressor protein, BHLHE40, which in turn dampens SLC6A20 activity. When cells carrying the risk variant were exposed to a common PBDE (BDE-47), SLC6A20 levels dropped further, glycine inside the cells fell, and cholesterol built up. Additional experiments confirmed that blocking SLC6A20 or reducing glycine limited the formation of glycine-linked bile acids and promoted cholesterol accumulation, matching the patterns seen in people with high exposure and high genetic risk.
What this means for health and prevention
Taken together, the study reveals a detailed pathway from everyday chemical exposure to disturbed blood fats, but only in genetically susceptible people. PBDEs appear to worsen cholesterol control by amplifying the effects of certain DNA variants that weaken glycine transport and bile acid production, reducing the body’s ability to clear cholesterol. For the general reader, the message is that neither genes nor environment act alone: subtle differences in our DNA can decide how harmful a given level of pollution will be. In the future, tools like the polygenic risk score developed here could help identify individuals who most need protection from specific pollutants, while the mechanistic insights around glycine and SLC6A20 may guide targeted therapies or nutritional strategies to buffer the metabolic impact of unavoidable exposures.
Citation: Hu, N., Li, B., Lu, Y. et al. Genetic variations interact with polybrominated diphenyl ether exposure to alter lipid homeostasis. Nat Commun 17, 3529 (2026). https://doi.org/10.1038/s41467-026-70222-8
Keywords: environmental pollutants, cholesterol, gene–environment interaction, metabolomics, PBDE flame retardants