Clear Sky Science · en
Dual phagocytosis-checkpoint blockade revitalizes immune surveillance in mouse models of glioblastoma
Turning the Body’s Clean-Up Crew Against Brain Cancer
Glioblastoma is one of the deadliest forms of brain cancer, in part because it is so skilled at hiding from the immune system. This study explores a new way to unmask these tumors by teaching the body’s own “clean-up crew” cells to recognize and devour cancer cells more effectively. The work suggests that blocking two specific molecular shields on tumor cells can awaken a powerful chain reaction that rallies multiple arms of the immune system against glioblastoma, at least in mice.
How Tumors Tell Immune Cells “Don’t Eat Me”
Our immune defenses include cells like macrophages, which patrol tissues, swallow damaged or abnormal cells, and show their fragments to T cells to spark a broader attack. Many cancers, however, protect themselves by displaying surface molecules that act as “don’t eat me” signals. One such molecule, CD47, has been a major experimental target, but drugs blocking CD47 alone have produced uneven benefits. The authors of this paper looked for other similar shields in glioblastoma and found that another molecule, CD24, is abundantly displayed on glioma cells, while its partner receptor, Siglec-10, sits on immune cells. This pattern suggests a direct line of communication from tumor to macrophage that tells the immune system to stand down.
A Two-Front Attack on Tumor Shields
Using mouse and human glioblastoma cell lines, the researchers tested antibodies that block either CD24 or CD47, or both together. They observed that blocking CD24 or CD47 individually modestly increased how often macrophages engulfed tumor cells that expressed CD24. When they blocked both signals at once, however, macrophages swallowed far more cancer cells, confirming that the two “don’t eat me” routes work in parallel. In glioma cells that lacked CD24, adding CD24 back restored this sensitivity, reinforcing that CD24 is a key switch controlling whether the dual treatment is effective.
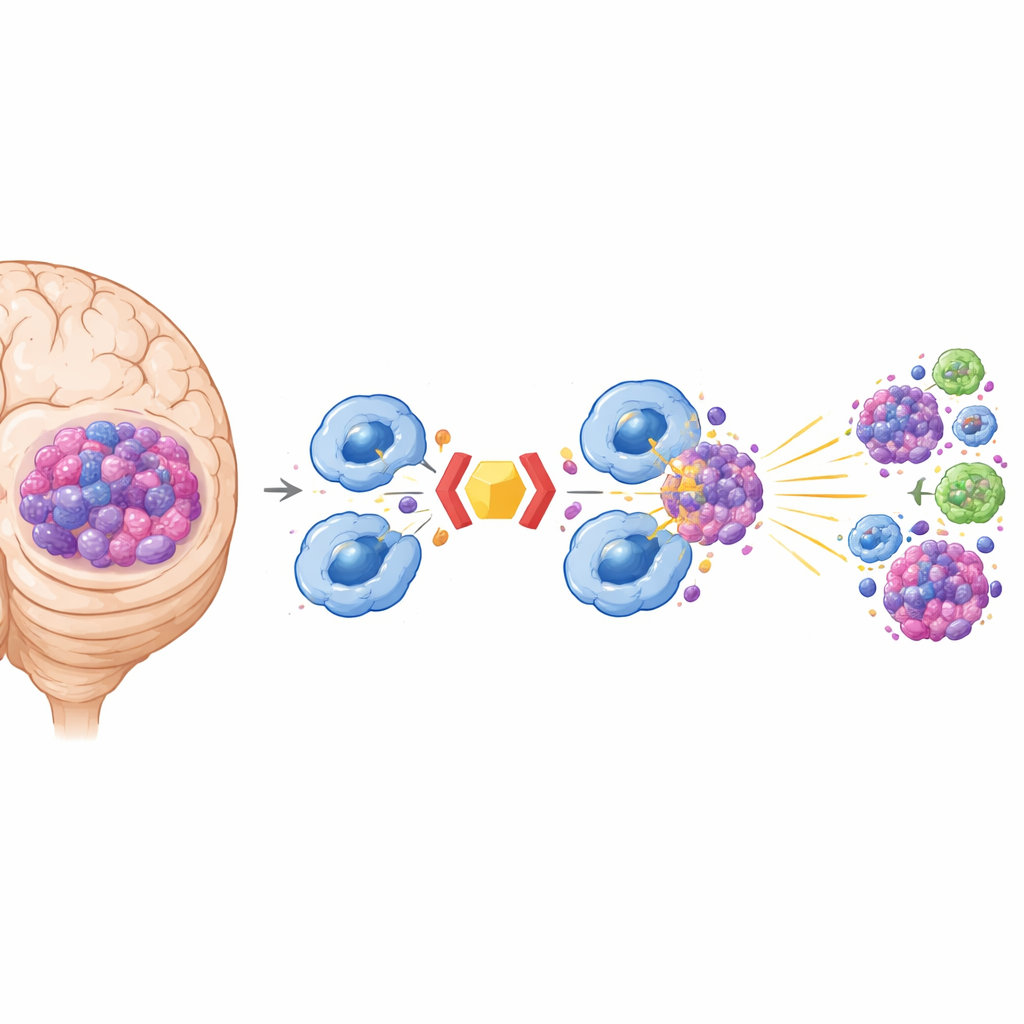
From Eating Tumors to Alerting the Whole Immune System
Macrophages do more than just digest tumors. After they consume cancer cells, they process tumor proteins and display them on their surface, where T cells can “see” them and become activated. In the mouse models, dual blockade of CD24 and CD47 led to stronger presentation of a test tumor antigen and vigorous proliferation of both helper (CD4+) and killer (CD8+) T cells. Inside macrophages, this enhanced eating triggered the cGAS–STING pathway, a molecular alarm system that senses DNA from engulfed tumor cells and drives the production of inflammatory molecules called type I interferons. These signals helped reshape the tumor environment from “cold” and immunosuppressive to “hot,” with more activated macrophages and increased infiltration of T cells into brain tumors.
Linking Innate Defenses to Modern Immunotherapy
In mouse glioblastoma models, especially those designed to mimic the treatment-resistant nature of human tumors, dual blockade clearly improved survival compared with blocking only one checkpoint. This benefit depended on intact cGAS–STING signaling in host macrophages and on the presence of CD8+ T cells, showing that both innate and adaptive immunity were needed. The authors also observed that as tumors became inflamed and T cells entered, they ramped up the PD1–PDL1 pathway, a known escape route that can blunt T cell activity. When the researchers combined dual phagocytosis checkpoint blockade with a PD1-blocking antibody—a drug class already used in the clinic—they saw further gains: more tumor-fighting T cells, fewer suppressive myeloid cells, smaller tumors, and longer survival. Mice that beat their tumors under this triple therapy even resisted tumor re-challenge, indicating a lasting immune memory.

What This Could Mean for Future Brain Cancer Treatment
For a non-specialist, the main message is that this work offers a blueprint for helping the immune system “see” and attack glioblastoma more effectively. By lifting two separate “don’t eat me” signals at once, macrophages can both destroy tumor cells directly and broadcast a strong alarm that wakes up T cells. Adding PD1 blockade then prevents the tumor from shutting those T cells back down. While the findings come from mice and will require careful translation using human-ready drugs, they point toward combination strategies that unite macrophage-targeted therapies with existing T cell checkpoint inhibitors. If similar effects are seen in people, such approaches could eventually give patients with glioblastoma a more durable and powerful immune defense against this aggressive cancer.
Citation: Ha, J., Wang, Y., Ma, Y. et al. Dual phagocytosis-checkpoint blockade revitalizes immune surveillance in mouse models of glioblastoma. Nat Commun 17, 3709 (2026). https://doi.org/10.1038/s41467-026-70221-9
Keywords: glioblastoma, macrophages, immune checkpoint blockade, cGAS-STING, cancer immunotherapy