Clear Sky Science · en
DNA Repair gene alterations and efficacy from gemcitabine and nab-paclitaxel with/without durvalumab and tremelimumab in metastatic pancreatic ductal adenocarcinoma
Why Some Pancreatic Cancers Respond Differently
Pancreatic cancer is one of the deadliest cancers, and even the best current drugs usually only extend life by months. Immune therapies, which have transformed treatment for several other cancers, have so far done little for most people with advanced pancreatic cancer. This study asks a hopeful question: is there a small group of pancreatic cancer patients whose tumors are wired in a special way that lets them benefit much more from adding immune therapy to standard chemotherapy?
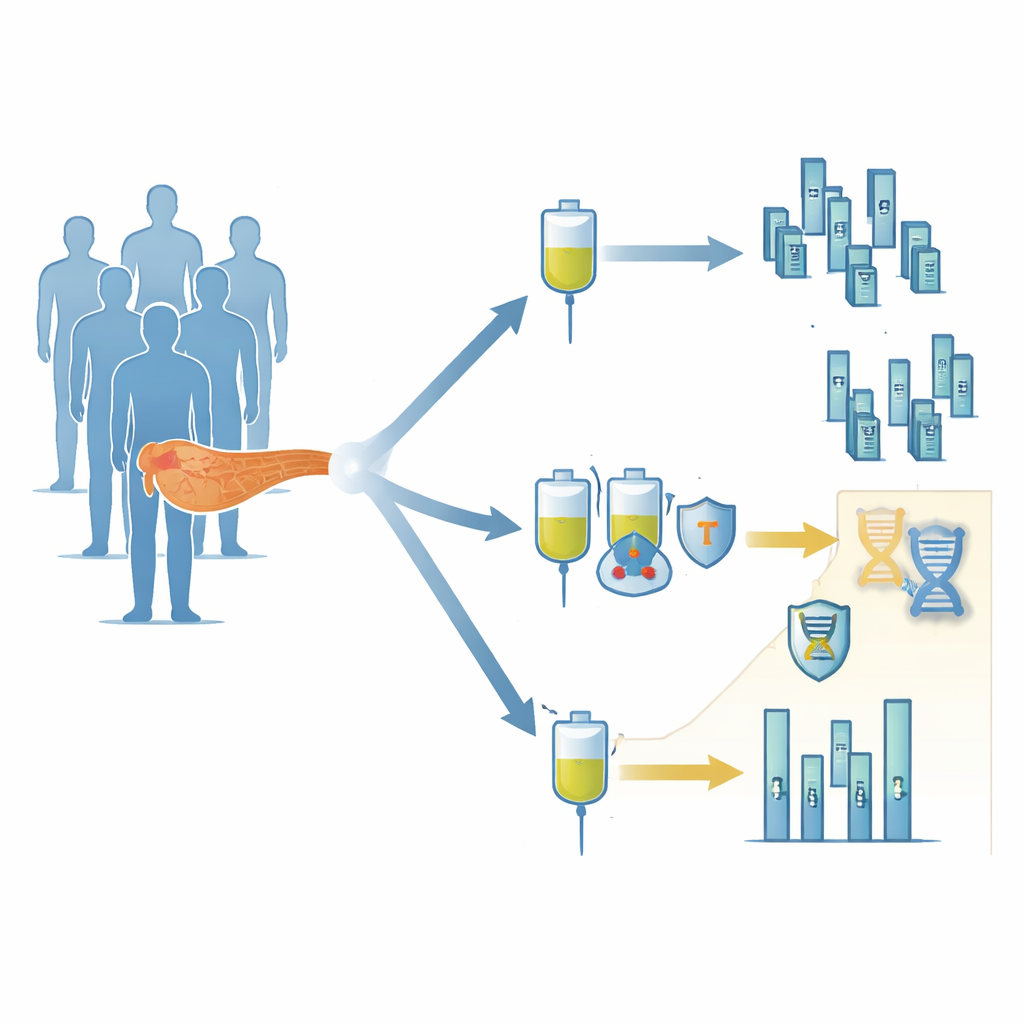
A Tough Cancer That Needs Better Options
Metastatic pancreatic ductal adenocarcinoma is usually diagnosed late, when it has already spread and is very hard to treat. Standard chemotherapy combinations have slowly improved survival, but only modestly. Immune checkpoint drugs, which remove brakes from the immune system, help in this disease only when the tumor has a rare defect called mismatch repair deficiency. Most patients do not have this defect, so doctors urgently need other reliable signs—biomarkers—that can flag who might truly benefit from immune-based treatments.
Testing Chemotherapy With and Without Immune Boosters
In the Canadian CCTG PA.7 clinical trial, 180 patients with newly diagnosed metastatic pancreatic cancer were randomly assigned to receive standard chemotherapy (gemcitabine and nab-paclitaxel) either alone or combined with two immune checkpoint drugs, durvalumab and tremelimumab. The researchers followed patients for more than six years to see who lived longer and whose disease stayed under control. Overall, the combination did not help the average patient: survival and time before the cancer grew were nearly the same in both treatment groups. At first glance, it looked as though adding immune therapy simply did not work in this setting.
Hidden Clues in Tumor DNA from a Blood Test
The team then searched for a deeper explanation using a blood-based test called circulating tumor DNA sequencing, which can detect genetic changes shed by tumors into the bloodstream. They focused on genes involved in fixing damage to DNA, including BRCA1, POLE, ATM and FANCA. Using a machine-learning approach, they found that when patients had mutations in at least two of these DNA repair genes, a striking pattern emerged. About 10 percent of patients fell into this group, and those who received the chemo–immunotherapy combination lived far longer than similar patients who received chemotherapy alone, with median survival extending from roughly 10 months to more than two years. Most of these patients also had noticeable tumor shrinkage on scans.
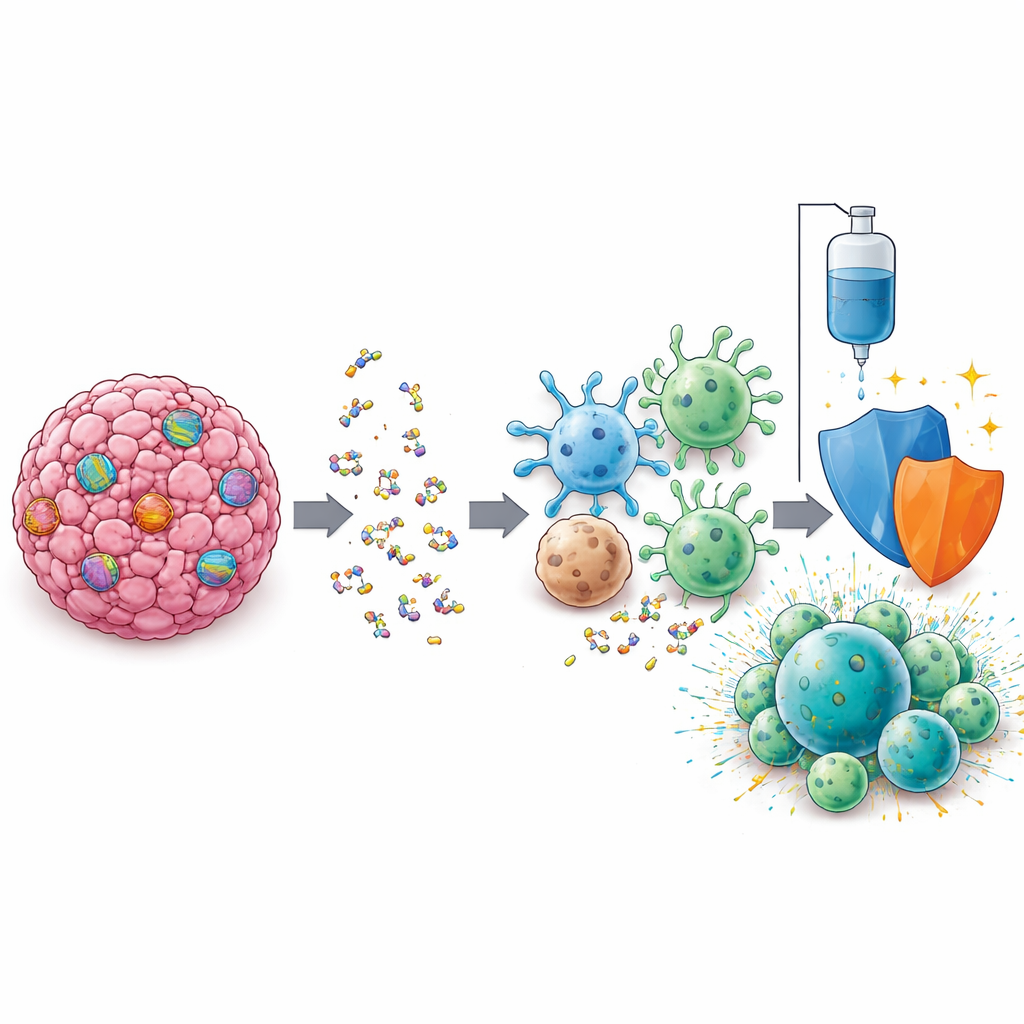
Checking the Signal in Stored Tumor Samples
To make sure the blood test was not misleading, the researchers also analyzed preserved tumor tissue from a subset of patients using whole-genome sequencing. In most cases, the same DNA repair gene mutations seen in the blood were confirmed in the tissue. The study also showed that these tumors did not start out with an unusually high overall mutation burden, suggesting that the specific pattern of DNA repair defects—rather than just a large number of mutations—may help sensitize tumors to the combination of chemotherapy and immune therapy. Still, the authors note that their analysis was exploratory and involved relatively small numbers, so the findings need to be tested in additional trials.
What This Could Mean for Patients
For the average person with advanced pancreatic cancer, adding dual immune therapy to standard chemotherapy did not improve survival. But this study highlights a small, actionable subgroup of patients whose tumors carry multiple faults in key DNA repair genes and who may gain years of extra life from the combination. Because these alterations can be detected through a blood test, they could, with further validation, help doctors quickly identify which patients are most likely to benefit from chemo–immunotherapy. In a disease where time is short and options are limited, such a biomarker-guided strategy could make a meaningful difference for a select group of patients.
Citation: Renouf, D.J., Topham, J.T., Loree, J.M. et al. DNA Repair gene alterations and efficacy from gemcitabine and nab-paclitaxel with/without durvalumab and tremelimumab in metastatic pancreatic ductal adenocarcinoma. Nat Commun 17, 3631 (2026). https://doi.org/10.1038/s41467-026-70120-z
Keywords: pancreatic cancer, immunotherapy, DNA repair genes, biomarkers, circulating tumor DNA