Clear Sky Science · en
Maintenance of intestinal CX3CR1+ macrophage homeostasis defines post-treatment control in SIV-infected macaques
Why the Gut Matters in HIV-Like Infections
The search for an HIV cure has long focused on hidden virus reservoirs in blood and lymph nodes. But the gut is one of the first and hardest-hit tissues in infection, and damage there can keep the immune system in a simmering state of inflammation for years. This study, using monkeys infected with an HIV-like virus (SIV), shows that a particular group of gut immune cells called macrophages can make the difference between viral resurgence after stopping treatment and long-lasting control of the virus without drugs.
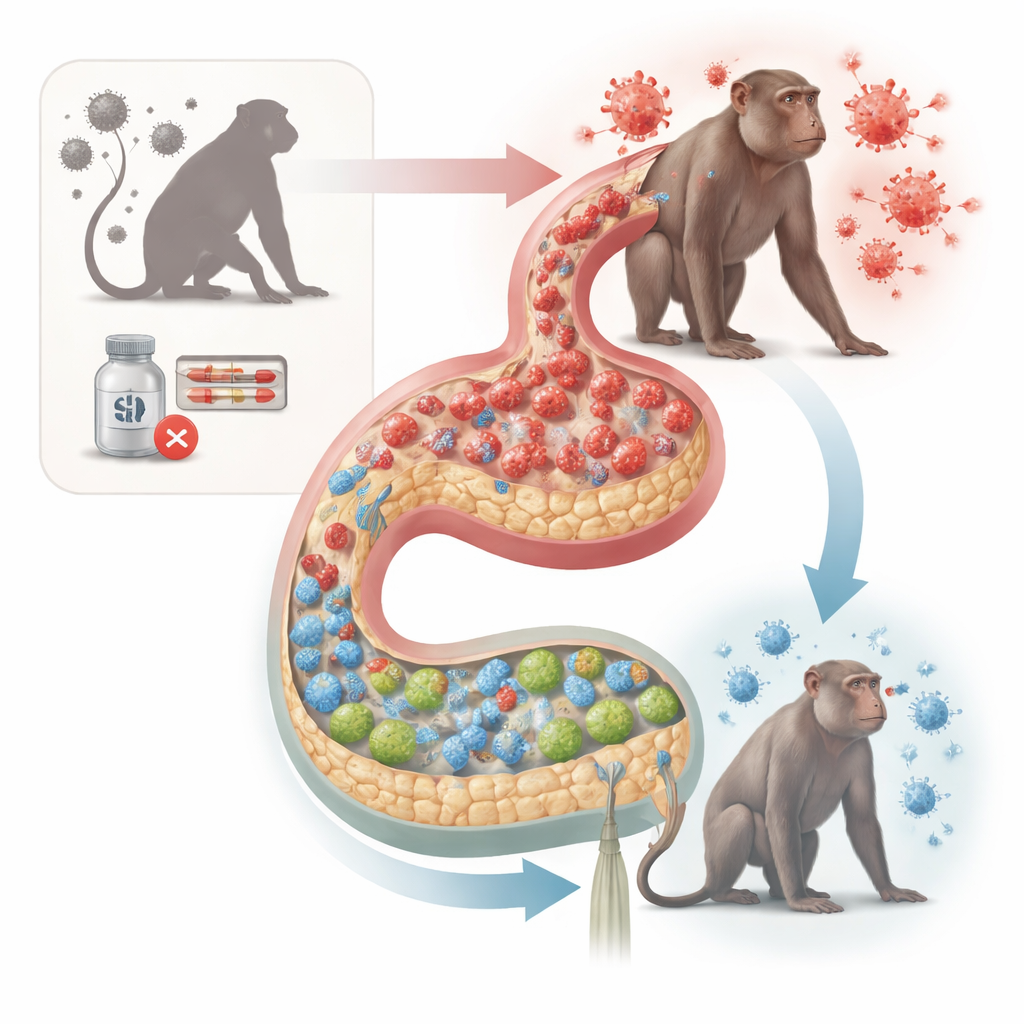
Different Paths After Stopping Treatment
The researchers followed 37 male cynomolgus macaques infected with SIV. Some animals never received antiretroviral therapy (ART), while others started ART either early or late after infection and then stopped it under close monitoring. After treatment interruption, a subset of monkeys kept the virus at very low levels—these were “post-treatment controllers.” Others, called non-controllers, experienced strong viral rebound. Although all treated animals had received similar amounts of virus exposure before ART, only those that became controllers fully regained blood CD4 T cells and had much smaller viral reservoirs in blood, lymph nodes, and gut tissue.
Guardian Cells in the Intestinal Wall
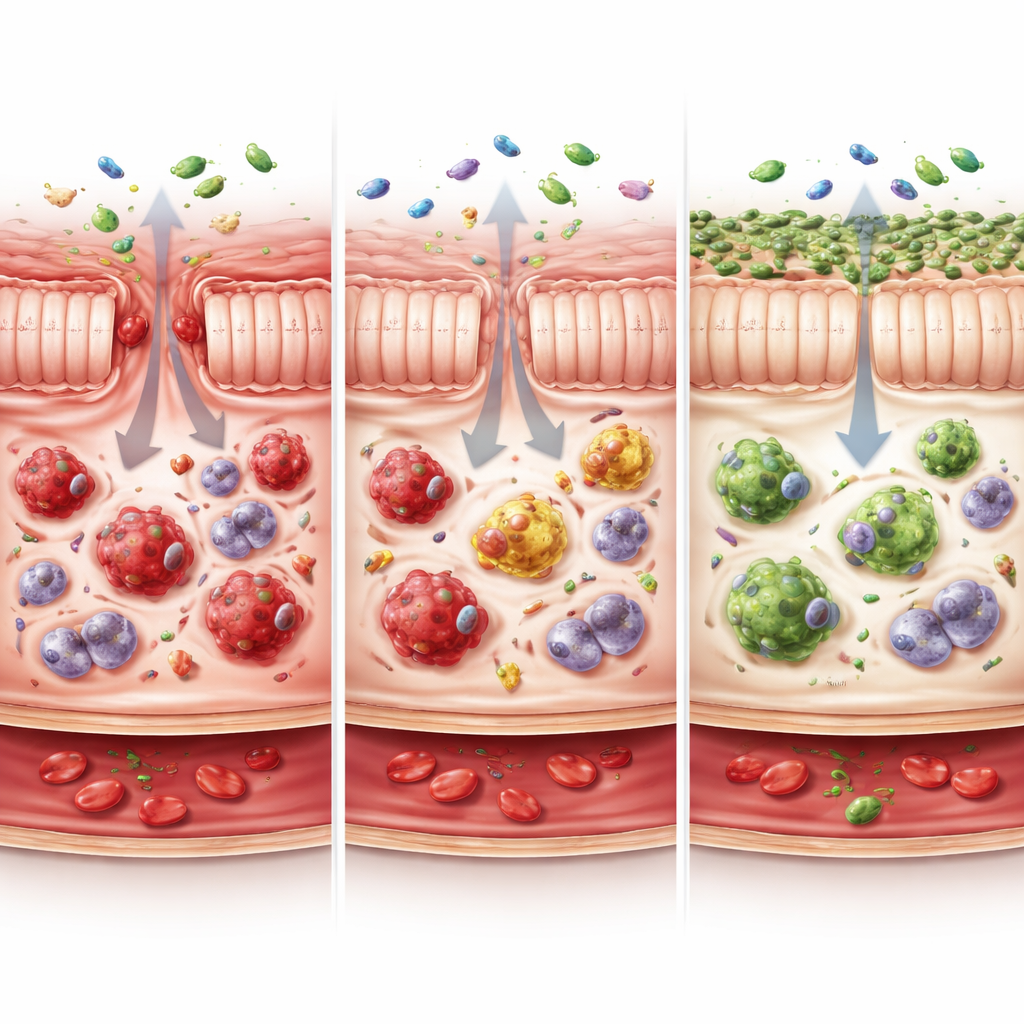
To understand why some animals controlled the virus, the team zoomed in on the intestinal lining, a major battleground in HIV and SIV. They focused on macrophages, immune cells that patrol tissues, clear microbes, and help repair damage. These cells come in different flavors. In healthy animals, most intestinal macrophages showed high levels of a surface molecule called CX3CR1, a signature of mature, calming cells that support tissue repair and immune balance. Chronic SIV infection, however, shifted this balance: the gut became dominated by CX3CR1-low macrophages associated with inflammation, while the protective CX3CR1-high cells dwindled. This skewed profile appeared early after infection and persisted in untreated animals.
Balanced Macrophages, Calmer Immunity
Post-treatment controllers broke this pattern. Even after stopping ART, their intestines largely maintained or restored the CX3CR1-high macrophage population and limited the build-up of CX3CR1-low cells, creating a profile much closer to uninfected monkeys. The more CX3CR1-high macrophages an animal had, the lower its viral load and the smaller its viral DNA reservoir. These “guardian” cells were closely linked to the presence of regulatory CD4 T cells—immune cells that dampen excessive responses—and to a healthier mix of T helper cells that protect the gut barrier. By contrast, CX3CR1-low macrophages were tied to heightened T cell activation, signs of exhaustion, and a shift toward more inflammatory T helper cells.
Inflammation That Spreads Beyond the Gut
The ripple effects of macrophage imbalance were seen well beyond the intestine. Animals with many CX3CR1-low macrophages had highly activated neutrophils—another type of white blood cell that can damage tissues when over-stimulated—and elevated levels of inflammatory molecules in the blood. Their draining lymph nodes also showed expanded pools of activated macrophages and depleted, overworked CD4 T cells. In controllers, by contrast, neutrophils were less activated, lymph node macrophages looked more quiescent, and blood levels of several inflammatory cytokines were much lower, all pointing to a body-wide state of reduced immune stress.
What This Means for Future HIV Cures
Altogether, the study suggests that keeping a stable population of CX3CR1-high macrophages in the gut is a hallmark of animals that can hold SIV in check after stopping ART. Rather than simply reflecting low virus levels, these cells appear to help preserve the gut barrier, support regulatory T cells, and prevent runaway inflammation that would otherwise feed viral persistence. For a lay reader, the key message is that a successful “functional cure” for HIV may depend not only on attacking the virus directly, but also on nurturing the right kinds of guardian cells in the intestine. These cells could serve as biomarkers to identify individuals with a good chance of post-treatment control and as targets for therapies aimed at restoring a calm, well-regulated immune environment.
Citation: Hua, S., Benmeziane, K., Desjardins, D. et al. Maintenance of intestinal CX3CR1+ macrophage homeostasis defines post-treatment control in SIV-infected macaques. Nat Commun 17, 3111 (2026). https://doi.org/10.1038/s41467-026-69848-5
Keywords: HIV cure, gut immunity, macrophages, SIV macaque model, immune inflammation