Clear Sky Science · en
General and selective ruthenium-catalyzed hydrogenation of primary amides to primary amines under mild conditions
Turning Tough Bonds into Useful Building Blocks
Chemists rely on amines—small, nitrogen‑containing molecules—to make everything from medicines and agrochemicals to dyes and plastics. A huge and underused reservoir of potential amines is locked inside amides, the sturdy chemical links that also hold proteins together. This study introduces a new way to gently "unlock" a wide variety of primary amides and convert them into valuable primary amines using hydrogen gas, offering a cleaner, more versatile route to many everyday and high‑tech products. 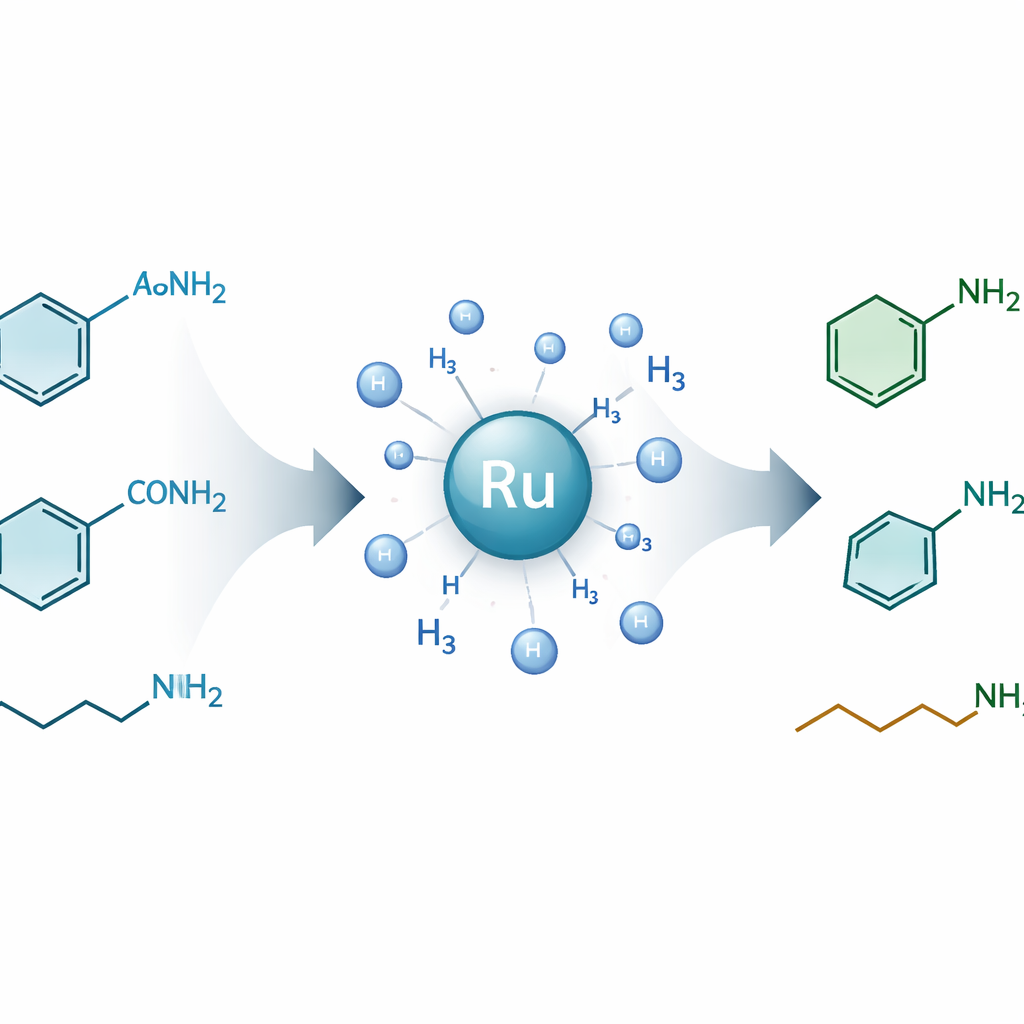
Why Breaking Amides Is So Hard
Amides are among the most resilient bonds in organic chemistry, which is why nature uses them to build proteins and why industry uses them in durable materials. That same strength, however, makes them notoriously difficult to modify. Converting amides into amines sounds simple—just remove oxygen and add hydrogen—but in practice the reaction can wander down many side routes, chopping the molecule apart or stitching fragments together in unwanted ways. Existing methods typically work only for a narrow set of amides, demand very high temperatures and pressures, or give mixtures of products that are hard to separate.
A New Helper for Hydrogen
The authors tackled this long‑standing problem by designing a specific molecular catalyst based on the metal ruthenium paired with a custom three‑armed phosphine ligand carrying methoxy groups (Triphos(p‑anisole)). This catalyst, formed in situ from inexpensive components, activates hydrogen gas and steers it toward the right bonds inside the amide. Crucially, the reaction is carried out under comparatively mild conditions for such a difficult task—about 115 °C and a hydrogen pressure of 10 bar, assisted by a modest amount of ammonia and a strongly hydrogen‑bonding solvent. Together, these ingredients let the catalyst break the tough carbon–oxygen connection in the amide while preserving the fragile carbon–nitrogen link that defines the desired amine.
From Simple Molecules to Medicines and Materials
Using this catalyst system, the team systematically showed that a broad range of primary amides can be transformed into their corresponding primary amines in good to excellent yields. Aromatic amides, which contain ring‑shaped fragments common in pharmaceuticals, were converted to benzylic amines, including versions decorated with halogens, ethers, and other useful groups. Heteroaromatic amides—those containing nitrogen or oxygen within the ring—likewise gave heterocyclic amines important in drug discovery and coordination chemistry. The method also works for straight‑chain and cyclic aliphatic amides, enabling the preparation of biogenic amines such as dopamine, tyramine, phenethylamine, and histamine, which play key roles as neurotransmitters and signaling molecules in the body. 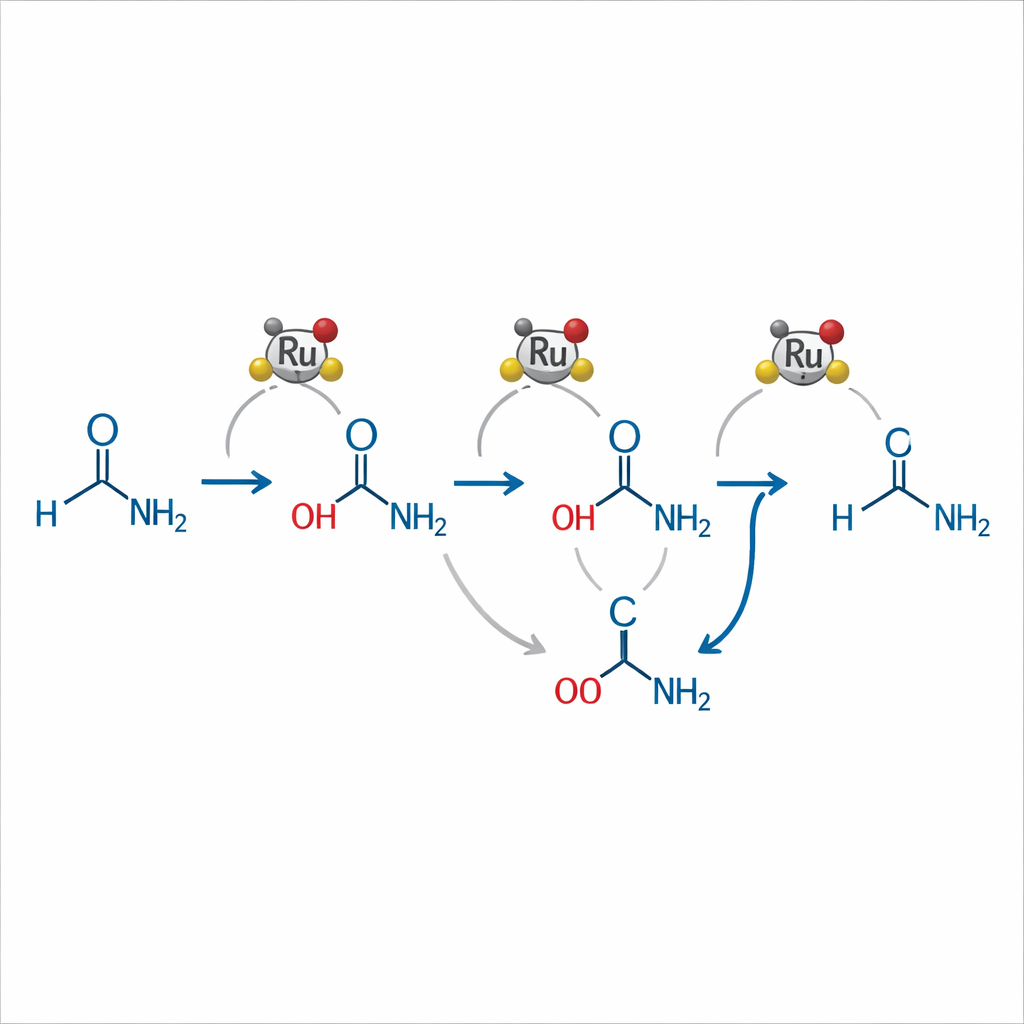
New Access to Everyday Functional Molecules
Beyond small bioactive molecules, the catalyst can turn fatty amides—derived from natural oils and fats—into long‑chain fatty amines. These compounds are workhorses of modern life, used as surfactants, fabric softeners, corrosion inhibitors, and additives in asphalt and other materials. The new route offers a complementary alternative to the current industrial practice of starting from nitriles, potentially allowing more flexible use of renewable feedstocks such as vegetable oils. The researchers further showed that their catalyst behaves differently with secondary and tertiary amides, favoring primary amides and underscoring its selectivity.
How the Catalyst Guides the Reaction
To understand how their system works, the authors combined careful product studies with high‑pressure nuclear magnetic resonance experiments. They propose that the ruthenium complex first forms a dihydride species that adds hydrogen across the amide, yielding an unstable "hemiaminal" intermediate. From there, two main paths are possible: dehydration to an imine that is then reduced to the amine, or cleavage that briefly forms an alcohol, which is converted back into an imine and finally into the amine. Ammonia plays a subtle but central role—shifting reaction equilibria away from unwanted coupling products, promoting the conversion of alcohols into imines, and stabilizing a resting state of the catalyst that can be reactivated by hydrogen.
A Step Toward Greener Chemical Manufacturing
In practical terms, this work provides a general, selective tool for turning robust amides into versatile primary amines using hydrogen gas, a clean reducing agent that produces water as the only by‑product. By working under significantly milder conditions than previous methods and tolerating many different molecular structures, the ruthenium–Triphos(p‑anisole) catalyst opens new possibilities for sustainable synthesis of pharmaceuticals, specialty chemicals, polymers, and fatty amine‑based materials from abundant amide precursors.
Citation: Kuloor, C., Goyal, V., Ma, Z. et al. General and selective ruthenium-catalyzed hydrogenation of primary amides to primary amines under mild conditions. Nat Commun 17, 3525 (2026). https://doi.org/10.1038/s41467-026-69794-2
Keywords: amide hydrogenation, ruthenium catalysis, primary amines, green chemistry, fatty amines