Clear Sky Science · en
Interfacial-mediated fast formation of toxic derivatives of phthalate esters
Hidden Dangers in Everyday Droplets
Many of the plastics that make modern life convenient quietly shed chemicals called phthalate esters into our air and water. These substances are already known to harm the lungs, liver, and developing children, and they have long been considered stubborn pollutants that break down only very slowly. This study reveals a surprising twist: at the surfaces of tiny airborne water droplets—like those in clouds, sea spray, or home humidifiers—phthalates can transform in minutes into new chemicals that are often even more toxic than the originals.
Why Tiny Droplets Matter
We tend to imagine pollution as something diluted in big volumes of air or water, but a huge amount of chemistry actually happens where air and water meet. The world is filled with microscopic droplets: in fog and clouds, in sea spray over the oceans, and in the mist from ultrasonic humidifiers indoors. The combined surface area of these droplets is enormous—cloud droplets alone offer tens of times more surface than the entire planet’s land and oceans. Yet most pollution models still assume that chemicals behave as if they were in uniform air or water, overlooking what happens at these thin interfacial skins.
A Fast-Acting Surface Factory
The researchers built a “contactless” reactor that mimics the working principle of a household ultrasonic humidifier. They sprayed water containing several common phthalates into a sealed chamber, generating microdroplets tens of micrometers across. By sampling the liquid over time and analyzing it with high‑resolution chromatography and mass spectrometry, they tracked how quickly one representative phthalate, diisobutyl phthalate (DiBP), disappeared and what it turned into. On droplet surfaces, about 97% of DiBP broke down within 12 minutes, with a half‑life of only 2.4 minutes—an astonishing speedup of 4 to 11 orders of magnitude compared with typical breakdown in bulk water or air, where these chemicals can persist for years.
How Water Surfaces Supercharge Reactions
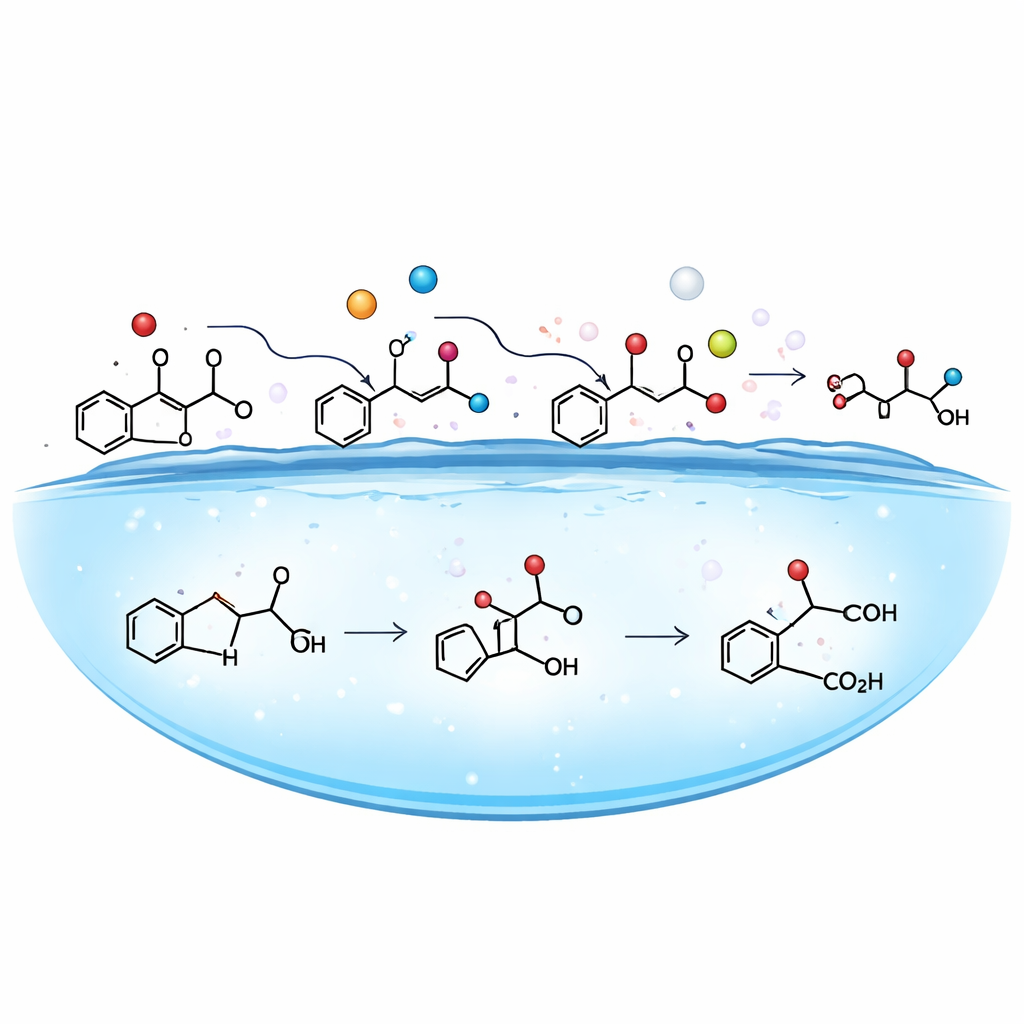
The key to this rapid transformation lies in how phthalates and water arrange themselves at the droplet surface. Computer simulations showed that DiBP prefers to sit at the air–water boundary, where its water‑loving and water‑fearing parts can both be partially satisfied. At the same time, the droplet surface spontaneously generates extremely reactive hydroxyl radicals—tiny oxidizing agents that form without added light, heat, or external chemicals. Experiments that selectively “quenched” different reactive species, together with electron spin measurements, confirmed that these hydroxyl radicals dominate the reaction. They attack the exposed parts of the phthalate molecules, snapping bonds and adding oxygen in a stepwise fashion. Detailed quantum mechanical calculations revealed that these surface‑enabled steps require much less energy than the same reactions in bulk water, explaining the enormous acceleration.
From Pollutant to Something Worse
By combining experimental measurements with automated structure searches, the team identified a suite of transformation products. The parent phthalate first loses pieces of its side chains and gains hydroxyl groups, and then is converted mainly into “carboxylated” products, including monoisobutyl phthalate and phthalic acid. Using advanced toxicology prediction tools, the authors compared the health hazards of these products with the original DiBP. The pattern was troubling: for several human health endpoints, the carboxylated products were much more harmful, showing up to 37.5‑fold higher predicted liver toxicity, 4.5‑ to 15‑fold greater potential to damage the respiratory system, and substantially stronger eye‑corroding power. Even the hydroxylated intermediates showed increased skin‑sensitizing potential, despite somewhat lower acute toxicity to aquatic organisms.
Implications for Homes, Clouds, and Policy
Because microdroplets have short lifetimes—from seconds in indoor mists to hours in fog—the full breakdown of phthalates to harmless end‑products is unlikely before the droplets evaporate. Instead, people and ecosystems are more likely to encounter the intermediate products, which this work shows can be more dangerous than the parent chemicals. Indoors, where people spend about 90% of their time, ultrasonic humidifiers act much like the experimental reactor, accelerating phthalate transformation right where people breathe. This helps explain why users may have lower levels of the original phthalates but could still face heightened risk from their more toxic derivatives. The study argues that environmental models and regulations must move beyond treating phthalates—and by extension many other ester‑containing chemicals—only in terms of their slow bulk‑phase decay. Instead, they must account for fast, surface‑driven chemistry in ubiquitous microdroplets and explicitly consider the toxicity of the products created along the way.
Citation: Li, X., Jiang, Q., Xia, D. et al. Interfacial-mediated fast formation of toxic derivatives of phthalate esters. Nat Commun 17, 2823 (2026). https://doi.org/10.1038/s41467-026-69495-w
Keywords: phthalate esters, microdroplets, air–water interface, transformation products, environmental health