Clear Sky Science · en
Ancestral neuronal receptors are bacterial accessory toxins
From Bacterial Weapons to Brain Wiring
Our brains rely on exquisitely precise connections between nerve cells, guided in part by unusual surface proteins called Teneurins. This study reveals that these key neuronal receptors did not start out as gentle guides at all. Instead, they began life in bacteria as components of toxic weapons used in microbial battles. By uncovering how these ancient toxins were repurposed into communication tools, the work offers a striking example of evolution turning instruments of war into the building blocks of complex multicellular life.
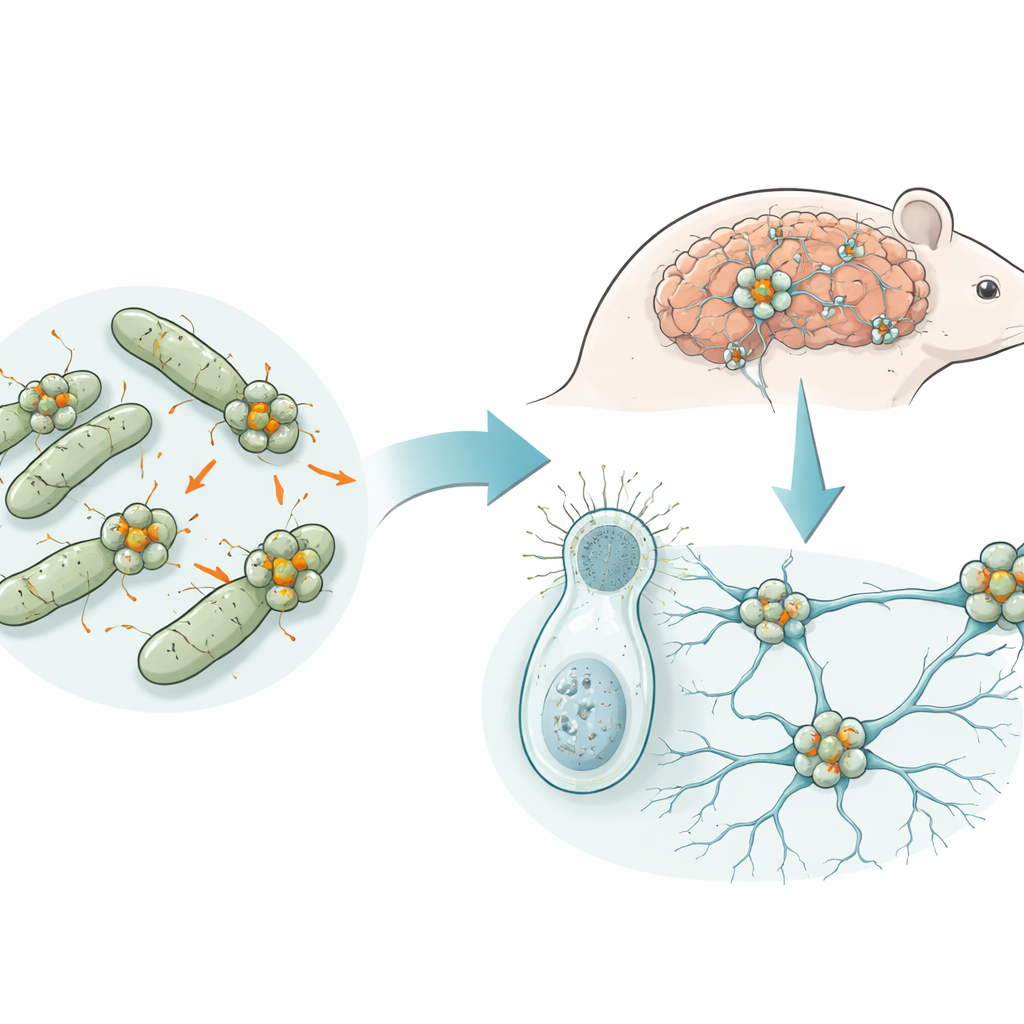
Ancient Proteins with a Surprising Past
Teneurins are large, intricate proteins that sit in the membranes of animal cells and help neurons find and connect to their correct partners. They are found across animals with nervous systems and even in some of their single-celled relatives, but bacteria do not have nerves at all. Earlier work hinted that Teneurins might have come from bacteria through horizontal gene transfer, where genes jump between species rather than passing down the family line. The authors set out to trace this ancestry and to understand what role the bacterial versions, called Teneurin-like proteins (TLPs), play in microbial life.
A Hidden Shell and a Toxic Core
Scanning hundreds of thousands of bacterial genomes, the team found that only a small fraction of species carry TLP genes, yet these species are scattered across many branches of the bacterial tree. TLPs share a conserved central architecture: a large protein "shell" built from repeated structural elements that spiral into a closed chamber. Using high-resolution cryogenic electron microscopy on a TLP from the bacterium Bacillus inaquosorum, the researchers showed that this shell closely resembles the central scaffold of animal Teneurins. However, instead of extending outward, the bacterial TLP’s tail folds back inside the shell to form a compact core that remains fully enclosed.
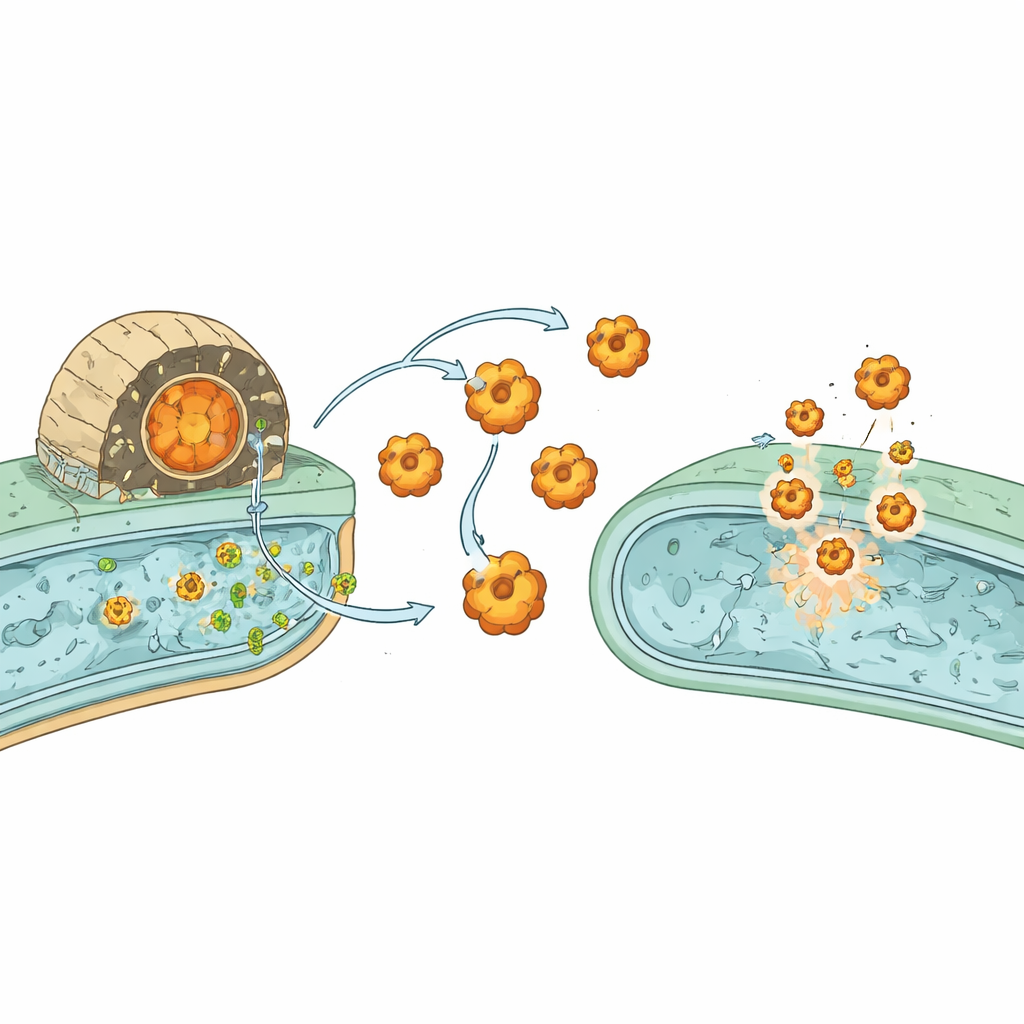
Bacterial Warfare and Built-In Protection
The team asked what this buried core does. Computer-based structure predictions across many TLPs revealed that these C-terminal cores resemble a wide array of enzymes known to damage cells, including nucleases, proteases, hydrolases, and ADP-ribosyl transferases—many of them classic toxin types. Experiments in Escherichia coli showed that expressing these cores could halt growth, puncture membranes, or drain key cellular molecules, confirming they act as toxins. Importantly, nearly every TLP gene sits next to a small partner gene that encodes a matching "immunity" protein. When co-produced, these immunity proteins bind the toxic cores and neutralize them, protecting the host bacterium while still allowing it to deploy toxins against competitors.
From Cell Attack to Cell Communication
Beyond specific case studies, the authors found that TLPs are especially enriched in bacterial families known for complex social behaviors, such as predation and cooperative swarming. This pattern, together with the modular toxin-plus-immunity layout, identifies TLPs as a distinct class of polymorphic toxins: customizable weapons that different bacteria tune to their ecological needs. The structural backbone of TLPs—the shell that packages their toxic cores—is essentially the same "superfold" that forms the heart of animal Teneurins. In animals, however, the toxic role has been lost. Instead, the Teneurin scaffold is linked to a membrane anchor and used as a stable surface receptor that mediates adhesion and signaling between neighboring cells, particularly neurons.
How a Weapon Helped Build the Nervous System
To a non-specialist, the central message is that a protein system once used by bacteria to poison their rivals was captured by early animal relatives and remodeled into a communication hub for nervous tissue. In bacteria, TLPs act as delivery platforms that package and protect small, highly variable toxins, paired with matching antidotes. When a similar structural scaffold moved into early eukaryotes—likely through gene transfer from bacteria being eaten as prey—it was repurposed: rather than firing toxins, it became a way for cells to recognize and adhere to one another. Over evolutionary time, this transformation helped support the emergence of organized tissues and, eventually, the sophisticated neuronal circuits that underlie animal behavior and cognition.
Citation: Raoelijaona, F., Szczepaniak, J., Schahl, A. et al. Ancestral neuronal receptors are bacterial accessory toxins. Nat Commun 17, 2753 (2026). https://doi.org/10.1038/s41467-026-69246-x
Keywords: horizontal gene transfer, bacterial toxins, teneurin receptors, nervous system evolution, cell-cell communication