Clear Sky Science · en
Extrusion fountains are hallmarks of chromosome organization emerging upon zygotic genome activation
How early life switches on its DNA
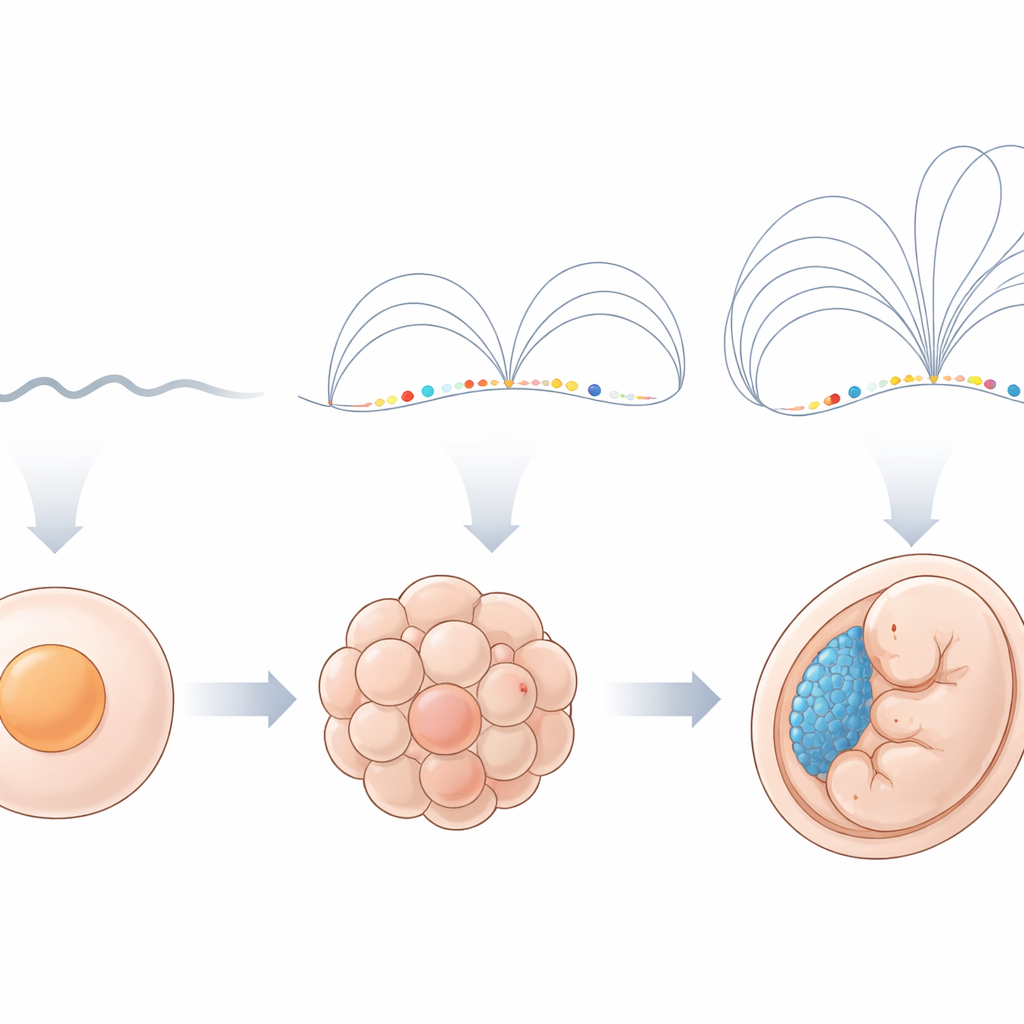
Every animal embryo faces the same challenge: at first, it runs on molecules from the mother, but very soon it must wake up its own DNA and start controlling its growth. This study asks what happens to the physical folding of chromosomes at that pivotal moment, and uncovers a surprising new structural pattern, nicknamed “fountains,” that seems to mark where key control switches in the genome turn on.
A quiet genome suddenly comes to life
In zebrafish and many other animals, the embryo’s own genes switch on in a burst called zygotic genome activation. Before this, chromosomes look relatively plain when viewed with Hi-C, a technique that measures which pieces of DNA are close together in 3D space. After activation, scientists usually see familiar patterns such as domains and loops that reflect how DNA is packed. The authors collected Hi-C data from sperm and multiple early stages of zebrafish embryos to watch this transition in detail. They confirmed that, before activation, chromosomes show few recognizable features, but soon afterward large-scale compartments and more local structures begin to appear.
Fountain patterns mark early control regions
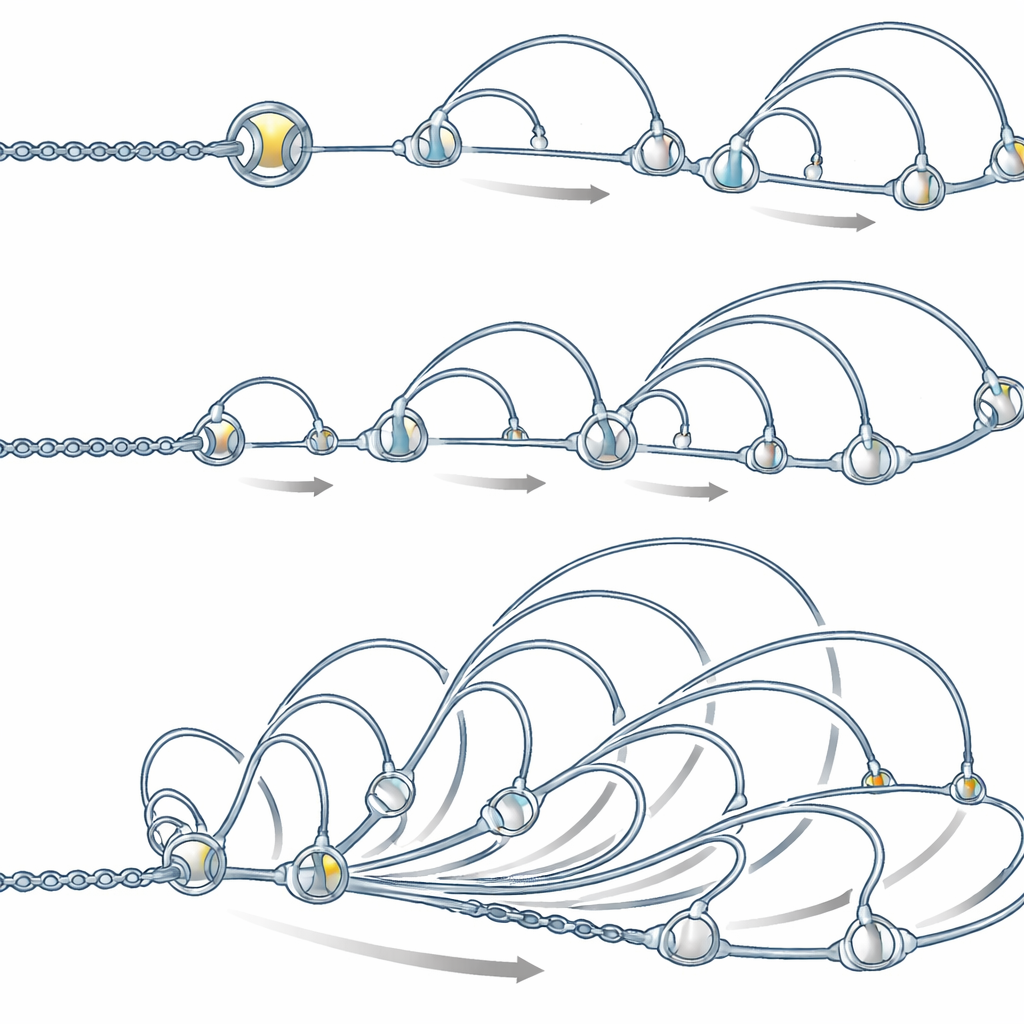
Looking closely at the first local features that appear, the team discovered distinct contact patterns they call fountains. In a Hi-C map, a fountain looks like a narrow base at a single DNA site that broadens into a fan of enriched contacts as one moves away along the chromosome. These shapes are different from the box-like domains and stripe-like features seen later. Using an automated detection tool, the authors found over a thousand such fountains shortly after genome activation in zebrafish, and similar patterns in early embryos of frog and medaka fish. Intriguingly, fountains tend to form in DNA regions that are open, active early in development, and decorated with chemical marks typical of enhancers—regulatory switches that help nearby genes turn on—rather than at promoters, where genes actually start being read.
Key starter proteins prepare spots where fountains form
To test whether fountains really depend on these early switches, the researchers focused on “pioneer” transcription factors—special proteins that can open tightly packed DNA. In zebrafish, three such factors (Pou5f3, Sox19b, and Nanog) are known to set up early enhancers. When embryos lacked all three, the characteristic fountain patterns largely vanished at the regions where chromatin accessibility and enhancer marks were lost. Individual mutants showed that when a pioneer factor failed to open DNA at a site, fountains at that site weakened or disappeared. At the same time, some fountains remained unchanged or even grew stronger, and these tended to coincide with enhancers that become active later in development or in specific tissues, hinting that fountains can also appear at “waiting” control elements before they fully turn on.
Loop-extruding rings shape the fountains
The authors then asked what might physically create these patterns. A leading candidate is cohesin, a ring-shaped protein complex known to grab DNA and reel it into loops in a process called loop extrusion. Measurements showed that cohesin accumulates at fountain bases, and that enhancer regions with more cohesin show stronger fountain patterns. Computer simulations of a flexible DNA chain, in which cohesin loads more frequently at certain sites and then extrudes loops outward, reproduced the observed fountain shapes, provided that loading at enhancers is several times higher than in the rest of the genome and that the two sides of each loop sometimes move out of sync, for example by colliding with other protein complexes.
Fountains appear across species and cell cycles
To see whether fountains are a general phenomenon, the researchers reanalyzed data from mouse embryonic stem cells and a mouse blood cell line. When they focused on enhancer regions in these cells, Hi-C–like maps again showed fountain-like fans of contacts, and these were strongly reduced when cohesin was experimentally depleted. During cell division, when cohesin temporarily leaves chromosomes, fountains disappeared; as cells entered the next growth phase and cohesin reloaded, fountains gradually re-emerged and later evolved into the more familiar domains and stripes. Similar enhancer-linked fountains have also been reported in worms, plants, fungi, and immune cells, often disappearing when cohesin is removed.
What this means for early development
Altogether, the findings suggest that, as an embryo first turns on its own genes or as a cell rebuilds its nucleus after division, chromosome folding begins at enhancer regions where cohesin is loaded more easily. These sites give rise to fountains—early, enhancer-centered folding elements that later mature into the complex 3D structures seen in fully developed cells. For a lay reader, the key message is that the same DNA switches that decide which genes turn on in early life also help sculpt the very shape of the genome, using cohesin rings as tiny machines that loop and organize chromosomes from the moment they awaken.
Citation: Galitsyna, A., Ulianov, S.V., Bazarevich, M. et al. Extrusion fountains are hallmarks of chromosome organization emerging upon zygotic genome activation. Nat Commun 17, 2787 (2026). https://doi.org/10.1038/s41467-026-69105-9
Keywords: zygotic genome activation, chromosome folding, cohesin loop extrusion, enhancers, embryo development