Clear Sky Science · en
Acetylation-triggered degradation of MSX1 impairs palatal development
Why This Matters for Babies and Families
Cleft palate is among the most common birth defects, affecting how newborns breathe, feed, and later speak. Doctors know that both genes and environmental exposures can raise the risk, but exactly how these influences damage the developing palate has been a mystery. This study uncovers a hidden molecular “switch” inside facial cells that decides whether they live long enough for the palate to close properly, offering fresh ideas for prevention and early treatment.
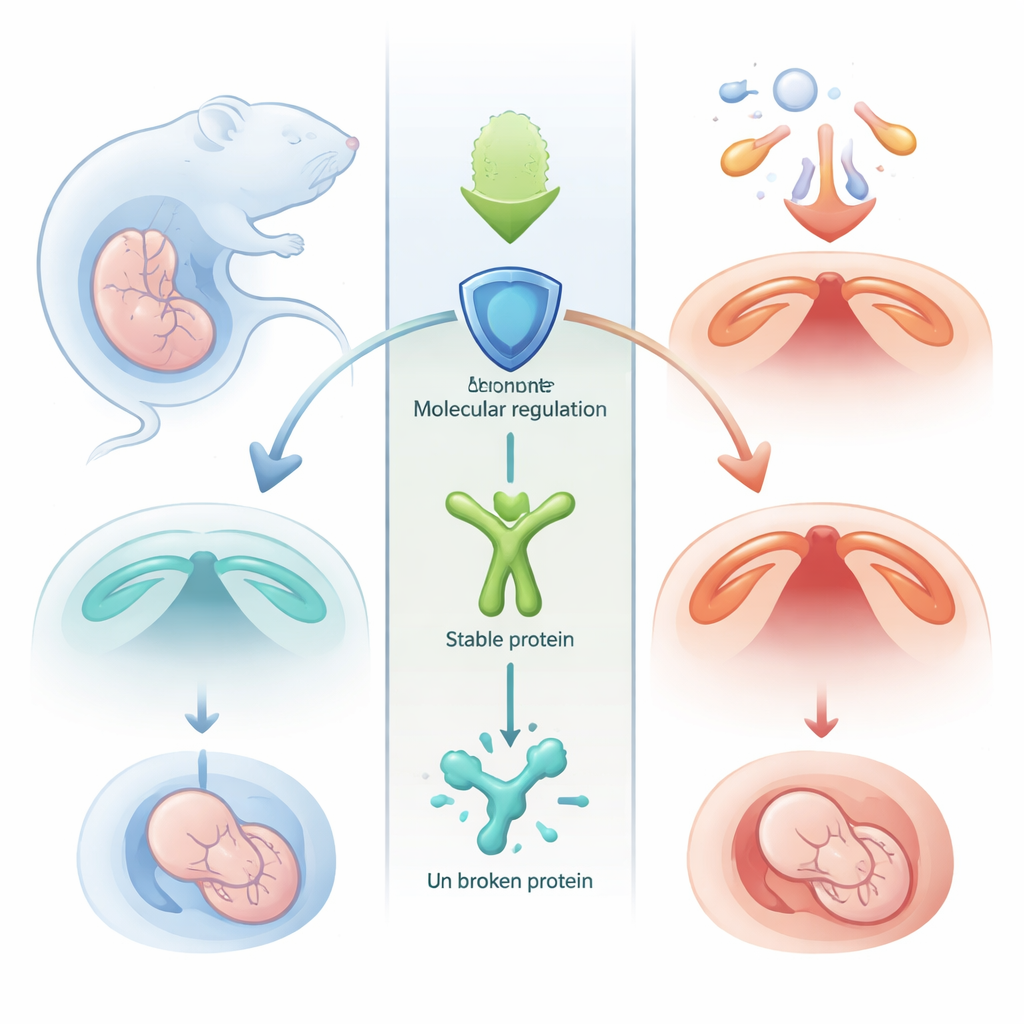
A Delicate Dance in the Developing Mouth
During early pregnancy, two shelves of tissue in the embryo’s mouth must grow toward each other and fuse to form the roof of the mouth. This process demands a careful balance between cell growth and cell death. If too many cells in the palate die at the wrong time, the shelves fail to meet, leaving an open gap known as a cleft palate. The authors focused on a protein called MSX1, long known as a key coordinator of facial development, and asked how its levels are controlled during this critical window.
When a Helpful Protein Is Marked for Destruction
Proteins inside cells are constantly being tagged, remodeled and sometimes destroyed after they are made. The team discovered that MSX1 carries a specific chemical tag—an acetyl group—at one site that acts like a disposal mark. When this tag is added, MSX1 becomes unstable and is quickly broken down by the cell’s protein-recycling machinery. In mouse palate cells, too much of this tagging dramatically lowers MSX1 protein levels even though the underlying MSX1 gene remains unchanged, revealing that the problem lies not in the DNA code but in how the protein is handled afterward.
The Guardian Enzyme That Keeps Cells Alive
Another protein, an enzyme called SIRT1, normally strips the acetyl tag off MSX1 and helps keep it stable. In healthy embryos, SIRT1 and MSX1 sit together in the cell nucleus, and SIRT1 activity protects MSX1 from premature destruction. Using a model of cleft palate triggered by a high dose of all-trans retinoic acid—a derivative of vitamin A that can act as a drug or pollutant—the researchers found that SIRT1 levels drop sharply. With this guardian weakened, MSX1 becomes over-acetylated, more heavily tagged for breakdown, and rapidly degraded. The result is a wave of excessive cell death in the palatal tissue and failure of the shelves to fuse.
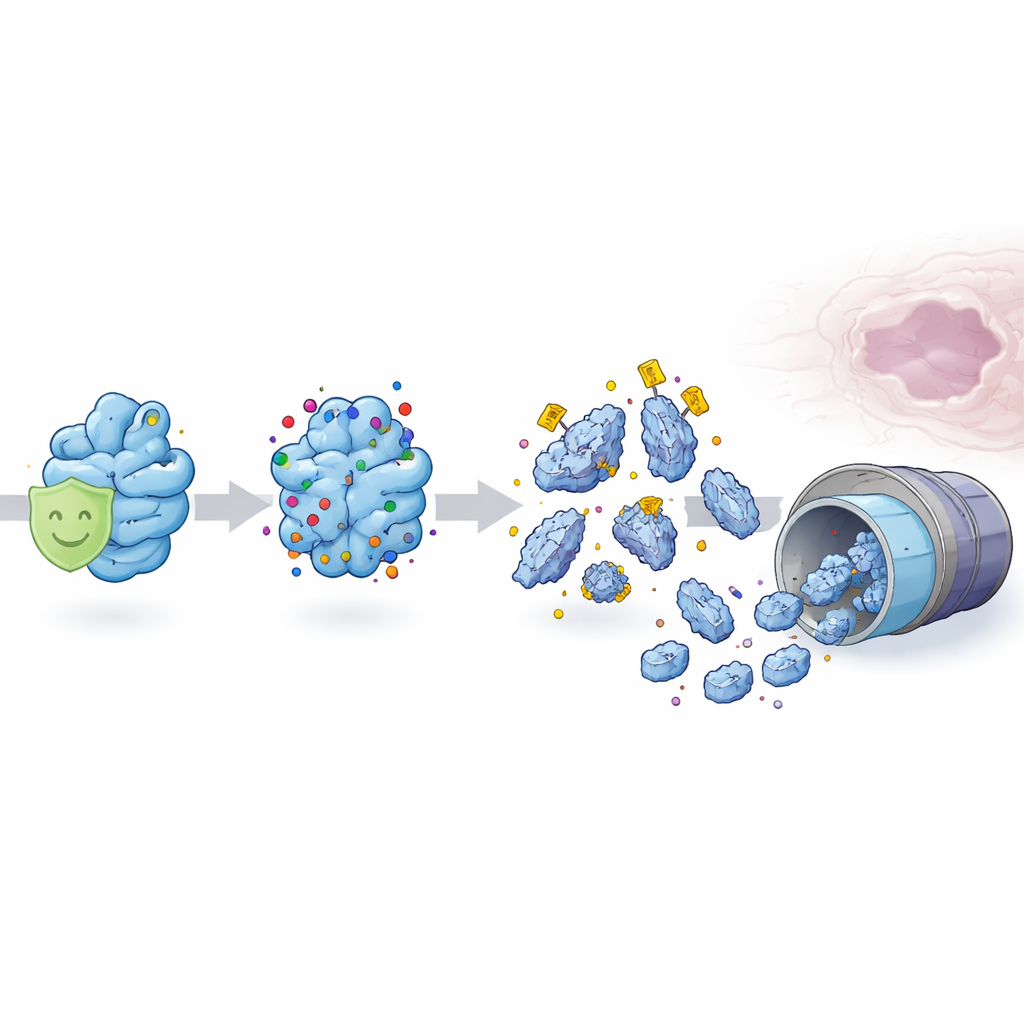
Testing Molecular “Decoys” to Rescue the Palate
To see whether they could counteract this damage, the scientists engineered two versions of MSX1: one that could not be acetylated at the key site and one that mimicked constant acetylation. In cell experiments, the non-acetylatable version stayed stable and strongly protected cells from dying, while the acetylation-mimic made things worse. When they delivered extra SIRT1, normal MSX1, or the acetylation-resistant MSX1 into pregnant mice exposed to retinoic acid, the severity of cleft palate was reduced and fewer cells in the palate underwent cell death. These tests showed that controlling this single chemical mark on MSX1 can meaningfully shift the course of palate development.
What This Could Mean for Future Care
For non-specialists, the key message is that this research identifies a precise molecular on–off switch that links environmental exposures to a common birth defect. Rather than changing what MSX1 does as a gene switch, acetylation controls how long the MSX1 protein survives, silently removing its protective role when tagging goes too far. By boosting SIRT1 activity or designing drugs that keep MSX1 from being over-tagged, it may one day be possible to lower cleft palate risk in pregnancies known to be high risk. The work also suggests that measuring modified MSX1 in prenatal samples could help identify vulnerable fetuses earlier, opening the door to more targeted monitoring and, eventually, preventive therapies.
Citation: Meng, L., You, J., Zhang, Z. et al. Acetylation-triggered degradation of MSX1 impairs palatal development. Cell Death Discov. 12, 156 (2026). https://doi.org/10.1038/s41420-026-03018-w
Keywords: cleft palate, embryonic development, protein acetylation, MSX1, SIRT1