Clear Sky Science · en
CHK1 is an integral regulator of DNA replication in human cells
Why keeping DNA copying on track matters
Every time a cell in our body divides, it must copy its entire DNA—billions of chemical “letters”—without introducing dangerous mistakes. If this copying process goes wrong, the result can be broken chromosomes, mutations, and ultimately diseases such as cancer. This study focuses on a protein called CHK1, a molecular traffic controller that helps keep DNA copying orderly. By switching CHK1 off with unusual precision, the researchers reveal just how central this guardian is for the life and health of human cells.
A molecular traffic controller for DNA copying
DNA replication begins at many starting points along the chromosomes and progresses through structures called replication forks, where the double helix is opened and duplicated. CHK1 belongs to a signaling pathway that watches over these forks, slowing them down or pausing the cell cycle when problems arise. Previous work mostly examined CHK1 during extreme stress, such as after chemotherapy or radiation, often using drugs that hit other targets as well. As a result, it was unclear what CHK1 does during everyday, “normal” DNA copying in otherwise unstressed cells.
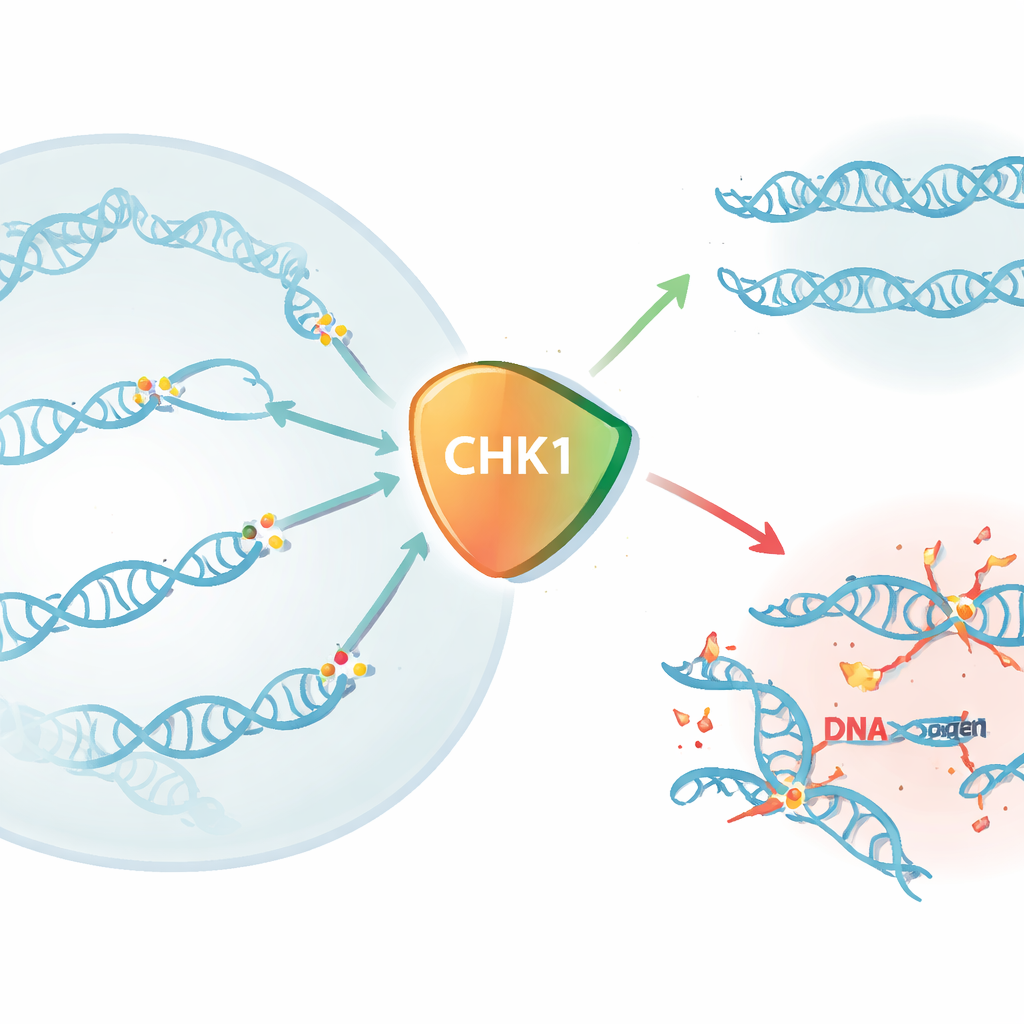
Rapid removal of CHK1 shows its importance
To answer this, the authors used the dTAG system, a genetic trick that lets them tag the CHK1 protein so it can be destroyed within minutes by adding a small molecule to the cell culture. In human cell lines, more than three-quarters of CHK1 disappeared within 15 minutes, and almost all of it was gone by half an hour. When CHK1 was acutely removed in this way, cells rapidly lost the ability to form colonies and showed a steep drop in viability within 16 hours, with almost complete cell death by 48 hours—often within a single cell cycle. Only reintroducing a fully functional CHK1 molecule, with its enzymatic activity and key regulatory regions intact, could rescue the cells, proving that CHK1’s kinase activity and its activation by an upstream protein called ATR are both essential for survival.
When CHK1 is gone, DNA forks crash
Once CHK1 was depleted, markers of DNA damage and replication stress surged. The researchers observed more DNA strand breaks using comet assays and increased activation of proteins that coat exposed single-stranded DNA. At the same time, cells stalled in the DNA-copying (S) phase: they could start replication, but their replication forks slowed, collapsed, and failed to finish the job. These cells then could not enter mitosis, the stage where chromosomes are separated into daughter cells. Pharmacological CHK1 inhibitors produced very similar patterns, and the effects appeared in both cancer-derived and non-transformed human cells, underscoring that CHK1 is not just a backup system for stressed cells but a core requirement for normal DNA replication.
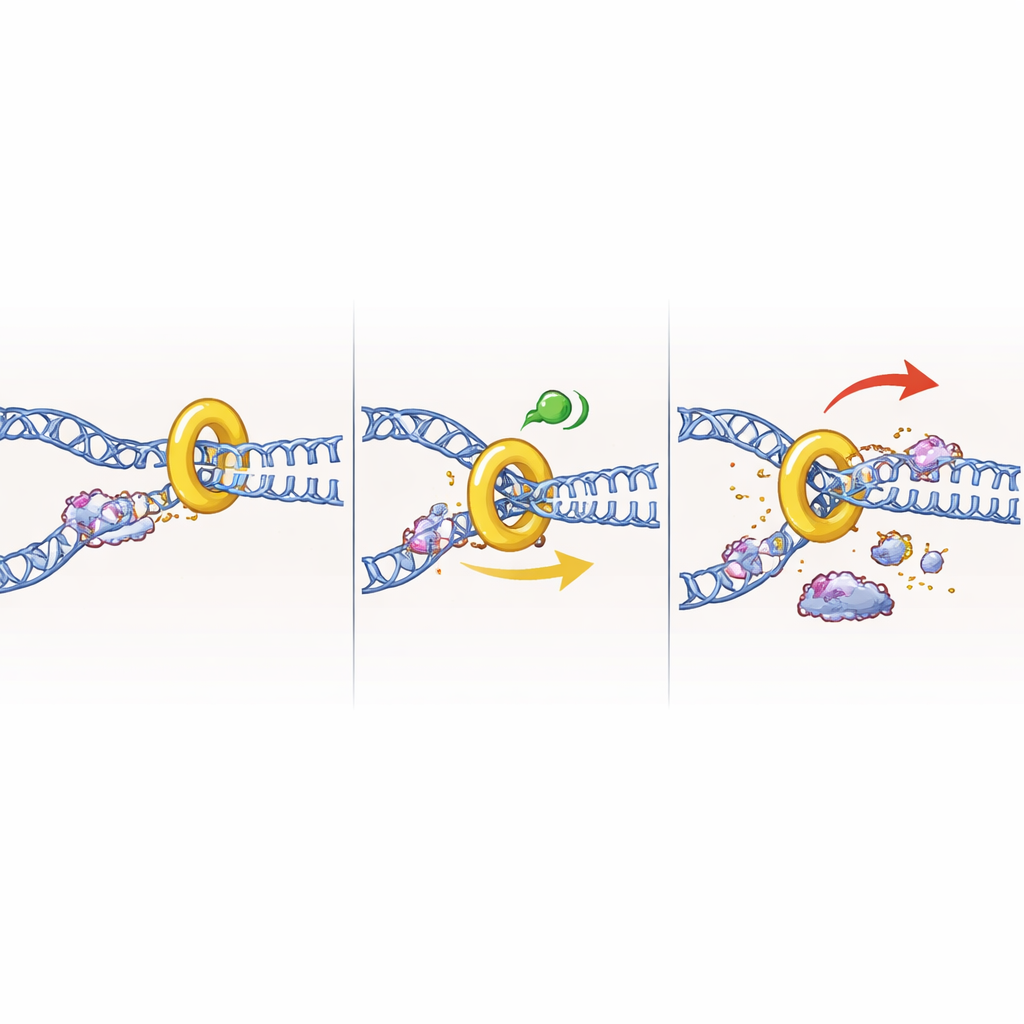
An unexpected role before DNA copying even starts
Perhaps the most surprising finding emerged when the team blocked cells at the boundary between the pre-replication (G1) phase and S phase, a point where bulk DNA synthesis has not yet begun. Intuitively, one might expect that losing CHK1 here would be harmless, since replication forks have not yet formed. Instead, removing CHK1 in these G1/S-arrested cells still triggered strong DNA damage signals and loss of viability, even though the cells were prevented from entering S phase. Follow-up experiments suggested that CHK1 normally restrains premature activation of replication origins and unwinding of DNA by the helicase machinery. When CHK1 was absent, this machinery appeared to initiate inappropriate unwinding, creating stretches of exposed, fragile DNA that led to breaks. Inhibiting other enzymes that kick-start origin firing partially rescued the damage, supporting this model.
What this means for health and therapy
Together, the work paints CHK1 as an integral, constant manager of DNA replication rather than a protein that steps in only when things go wrong. CHK1 acts before DNA copying starts, to prevent reckless origin activation, and continues to operate during S phase, matching the speed of the DNA-unwinding helicase to the copying enzymes so that forks move safely. When CHK1 is removed, this coordination fails, replication forks crash, DNA breaks accumulate, and cells die quickly. For a lay reader, the key message is that CHK1 helps ensure that each new copy of the genome is produced carefully and completely. This central role helps explain why CHK1 has become a promising target in cancer therapy—blocking it can push already stressed tumor cells over the edge—but also why such treatments must be used thoughtfully, given CHK1’s fundamental importance to the survival of normal dividing cells.
Citation: Li, S., Zhu, D., Tang, M. et al. CHK1 is an integral regulator of DNA replication in human cells. Cell Death Dis 17, 375 (2026). https://doi.org/10.1038/s41419-026-08624-1
Keywords: DNA replication, cell cycle, genome stability, checkpoint kinase, replication stress