Clear Sky Science · en
A fistful of iron: ferritin as a vulnerability point of the brain cancers
Why iron in the brain matters
Iron is essential for healthy brain function, helping nerve cells make energy and communicate. But too much free iron can act like a spark in dry grass, fueling damaging chemical reactions. This article explains how brain cancers, including glioblastoma and medulloblastoma, become dependent on iron and use a protein called ferritin as a kind of safety vault. By understanding this iron addiction and the role of ferritin, researchers hope to uncover new ways to weaken stubborn tumor cells that resist current treatments.
Iron hunger in brain tumors
Brain tumor cells, especially a small but powerful group known as cancer stem cells, need far more iron than normal brain cells. They pull iron from the blood, hold on to it, and store large amounts inside. This iron helps them grow, divide, and survive stressful conditions such as low oxygen or exposure to drugs and radiation. However, iron is a double-edged sword. When it reacts with certain oxygen byproducts, it generates highly reactive molecules that can tear apart cell membranes and trigger a special kind of cell death driven by fat oxidation, called ferroptosis. Tumor cells work hard to avoid this fate by tightly controlling how much iron is free and how much is locked away.
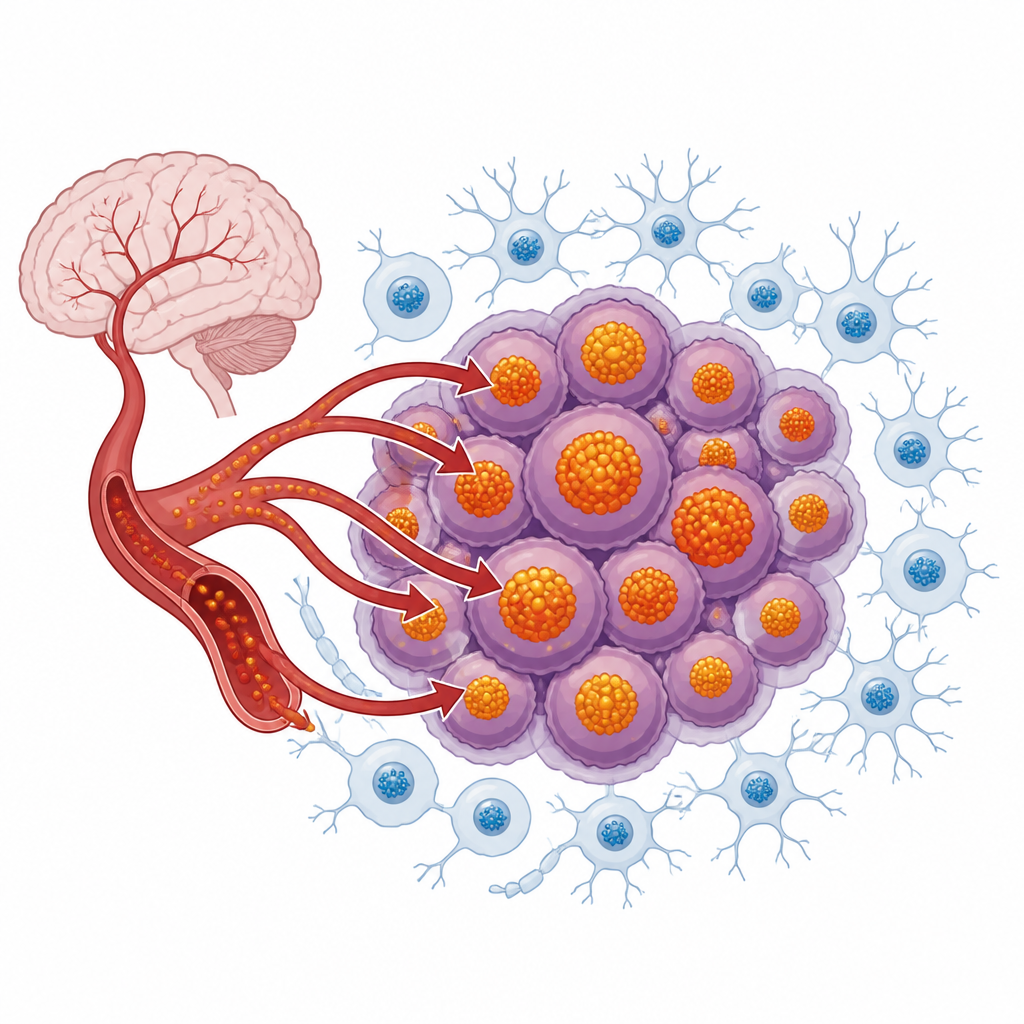
Ferritin as the iron vault
Ferritin is a hollow, spherical protein that acts as the main storage vault for iron inside cells. Each ferritin particle can safely hold thousands of iron atoms in a nonreactive form, preventing harmful chemical reactions. In the brain, ferritin is important for normal development, memory, and protection against degeneration. Brain tumors take this normal protective system and push it to the extreme. Many glioblastomas and medulloblastomas show high levels of ferritin, and these levels often rise with tumor grade and poorer outcomes. Cancer stem cells in these tumors appear to rely especially heavily on ferritin to buffer their large iron stores and shield themselves from iron-driven damage.
Iron, stem cells, and treatment resistance
Cancer stem cells are thought to drive relapse because they can self-renew, adapt, and survive therapies that kill most other tumor cells. The review shows that these cells remodel iron handling at many steps: they increase import, reduce export, and boost storage through ferritin. This extra iron does more than feed growth. It also supports changes in DNA packaging and gene activity that reinforce stem-like traits and therapy resistance. In some tumor types, disturbing ferritin can push cells toward more aggressive behavior, highlighting how context matters. In brain cancers, though, evidence suggests that removing ferritin’s buffering power can leave iron-hungry stem cells exposed to toxic reactions they can no longer contain.
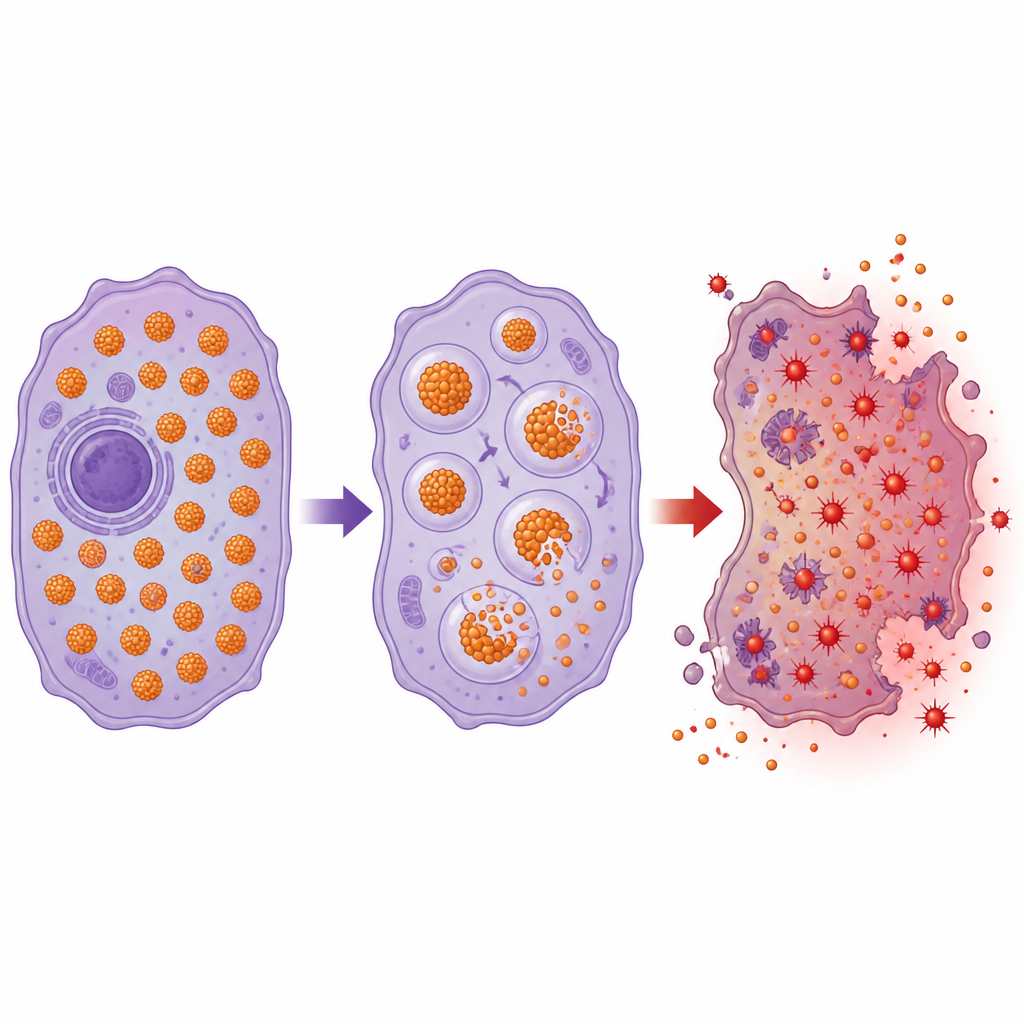
Turning iron from helper to hazard
Because brain cancers are so dependent on iron, researchers are exploring ways to flip iron from a helper to a hazard by targeting ferritin. One emerging strategy is ferritinophagy, a process in which ferritin is deliberately broken down, suddenly releasing its stored iron. This surge of accessible iron can drive the reactions that damage cell membranes and push cells toward iron-dependent death. Experimental compounds, as well as high-dose vitamin C, can trigger ferritin breakdown in lab models, and may also make tumors more sensitive to radiation and chemotherapy, both of which increase damaging oxygen byproducts. The challenge is to reach brain tumors through the blood–brain barrier and to injure cancer cells without harming healthy neurons and support cells.
What this could mean for patients
The article concludes that ferritin sits at a critical crossroads in brain cancer: it keeps iron safe enough for tumor cells to thrive, but that same safety system could be turned against them. By carefully disrupting ferritin in iron-addicted brain tumors, doctors may someday be able to weaken cancer stem cells, enhance the impact of existing treatments, and reduce the chance of relapse. To get there, scientists must better understand how ferritin behaves in different brain tumor types, how to deliver ferritin-targeting drugs into the brain, and how to avoid damaging normal tissue. If these hurdles can be overcome, ferritin-focused therapies could add a powerful new tool to the treatment of aggressive brain cancers.
Citation: Segui, F., Parks, S.K., Vucetic, M. et al. A fistful of iron: ferritin as a vulnerability point of the brain cancers. Cell Death Dis 17, 451 (2026). https://doi.org/10.1038/s41419-026-08564-w
Keywords: brain cancer, ferritin, iron metabolism, cancer stem cells, ferroptosis