Clear Sky Science · en
UHRF1 regulates AR ubiquitination to promote the loss of AR signaling and enzalutamide resistance in progression of prostate cancer
Why this research matters
Prostate cancer is often kept in check by drugs that shut down male hormone signals, but many tumors eventually outsmart these treatments and come back in a more dangerous form. This study uncovers a key internal "switch" that helps prostate cancer cells escape hormone-blocking drugs, and suggests a new way to make those medicines work longer and better for patients.
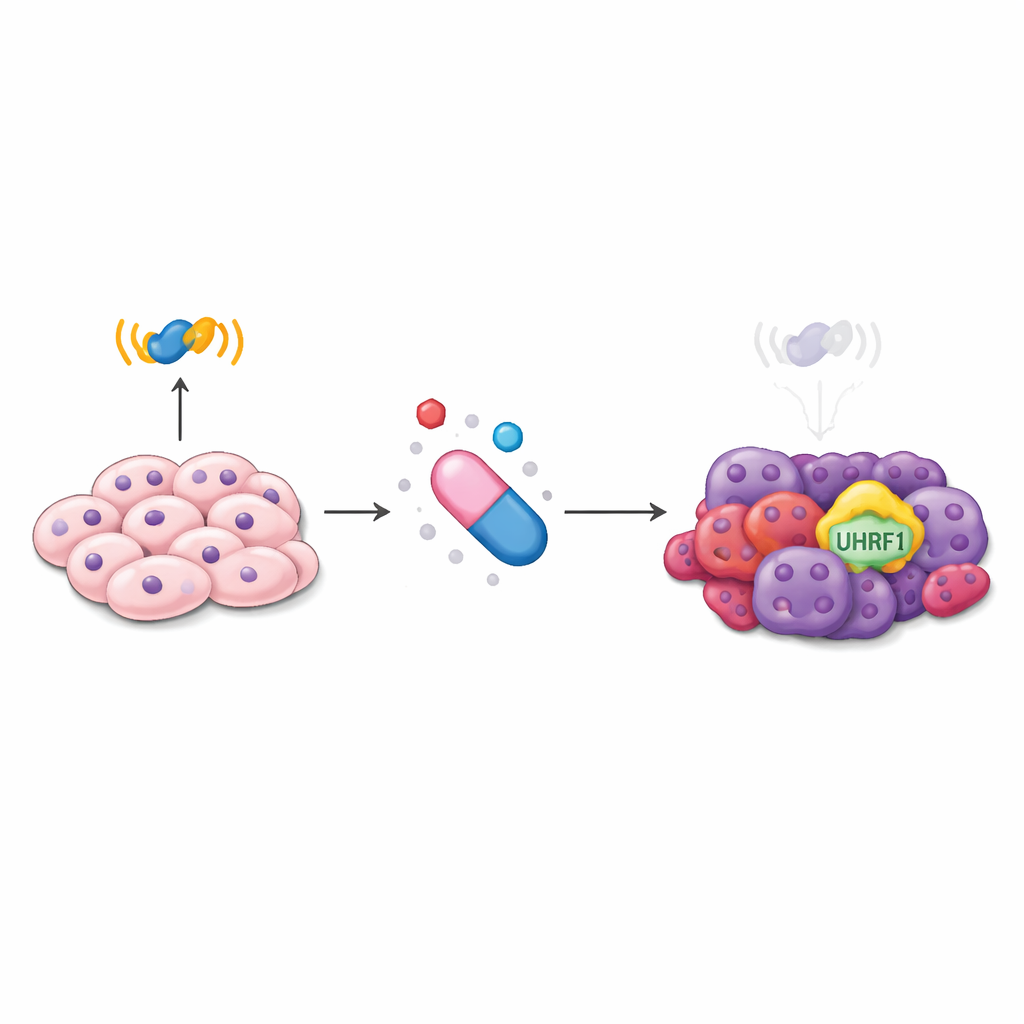
A drug that stops working
Modern prostate cancer care relies heavily on blocking the androgen receptor, a protein inside cells that senses male hormones and drives tumor growth. Drugs like enzalutamide are designed to silence this signal. Over time, however, some tumors shift away from relying on this pathway. They adopt more flexible identities, including stem cell–like and nerve-like traits, and become largely indifferent to hormone-blocking therapy. At this advanced stage, options are limited. The researchers focused on a molecule called UHRF1, known for controlling how DNA is packaged and read, to see whether it might be involved in this dangerous shape-shifting process.
A hidden helper of resistance
By examining patient data and cancer cell lines, the team found that UHRF1 levels rise as prostate cancer progresses and spreads. Tumors with higher UHRF1 tended to have worse outcomes. In datasets enriched for aggressive, nerve-like prostate cancers, UHRF1 was especially abundant and clustered with genes that mark neuroendocrine and stem-like states. In the lab, cells that had been forced to become resistant to enzalutamide showed a clear pattern: the androgen receptor protein was greatly reduced, while UHRF1 and neuroendocrine markers were increased. This suggested that UHRF1 might be part of the machinery that allows cells to abandon their hormone-dependent identity and tolerate treatment.
How UHRF1 breaks the hormone signal
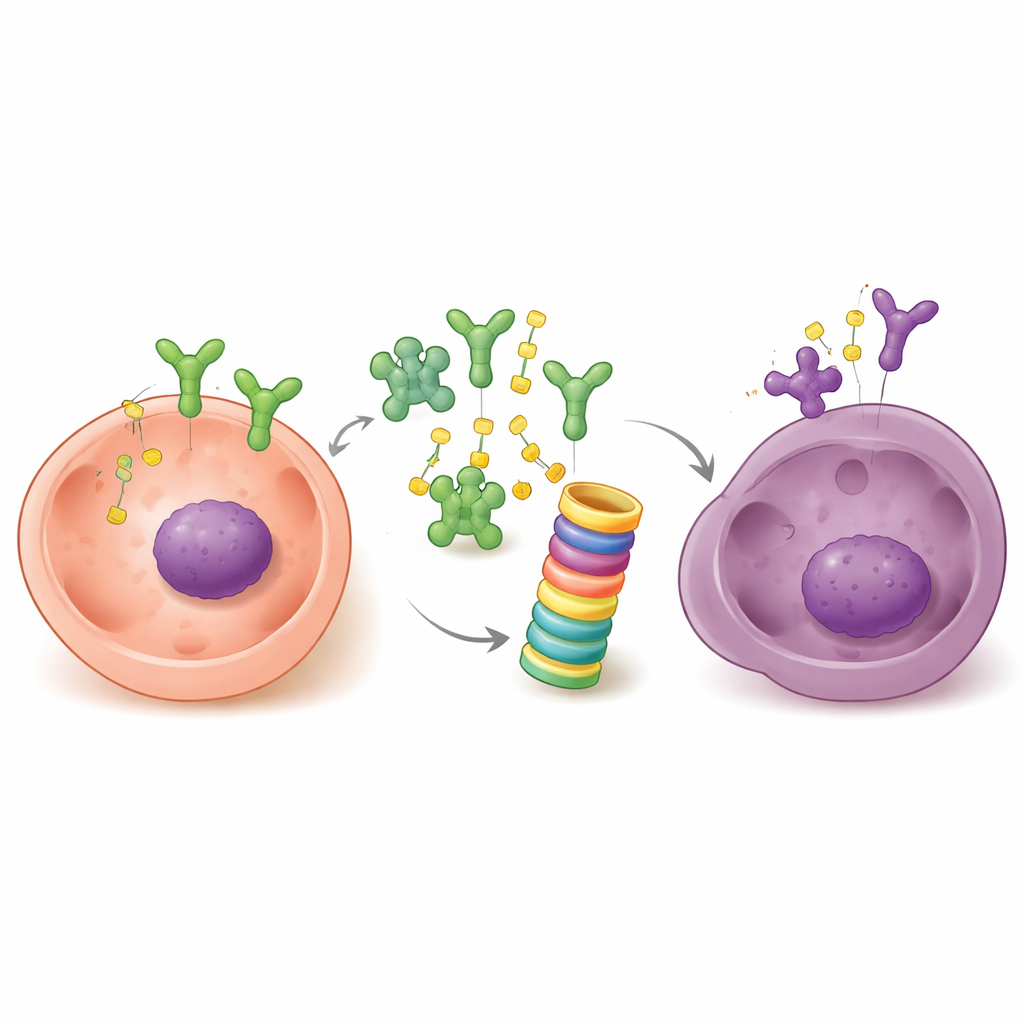
The investigators then asked how UHRF1 affects the androgen receptor itself. They discovered that reducing UHRF1 in resistant cells restored the androgen receptor protein and reactivated its downstream signaling, even though the gene’s RNA levels barely changed. This pointed to a problem of protein stability rather than gene activity. Further experiments revealed that UHRF1 physically binds to the androgen receptor and tags it with chains of a small protein used by cells as a disposal signal. This tag marks the receptor for destruction in the cell’s protein-shredding machinery. When the team disabled the part of UHRF1 responsible for this tagging activity, the androgen receptor was no longer efficiently broken down. In essence, UHRF1 works as an internal "hitman," marking the hormone receptor for removal and thereby weakening the drug’s target.
Turning resistance back into vulnerability
Because UHRF1 helps erase the hormone signal, the researchers tested whether blocking UHRF1 could make resistant tumors sensitive to enzalutamide again. In cell culture, knocking down UHRF1 slowed the growth of enzalutamide-resistant cells and, importantly, made them respond to the drug once more. The combination of UHRF1 loss and enzalutamide treatment suppressed cell proliferation much more than either approach alone, largely by curbing growth rather than triggering massive cell death. In mice carrying resistant prostate cancer grafts, tumors with reduced UHRF1 shrank dramatically under enzalutamide, while control tumors barely responded. Molecular analyses showed a partial reversal of aggressive, neuroendocrine-like features, consistent with a shift back toward a hormone-responsive state.
What this means for patients
The work paints a clear picture: as prostate cancers are pressured by hormone-blocking therapy, rising UHRF1 levels help them shed their reliance on the androgen receptor by accelerating its destruction. This pushes tumors toward a more plastic, drug-resistant identity. Interrupting UHRF1 during this transitional window can preserve or restore hormone signaling long enough for drugs like enzalutamide to keep working. While UHRF1 inhibition alone is unlikely to cure late-stage, fully transformed disease, targeting this molecule alongside standard therapy could delay or prevent the shift to an untreatable state, offering patients more time and more effective control of their cancer.
Citation: Zhang, Y., Yu, Z., Li, Y. et al. UHRF1 regulates AR ubiquitination to promote the loss of AR signaling and enzalutamide resistance in progression of prostate cancer. Cell Death Dis 17, 286 (2026). https://doi.org/10.1038/s41419-026-08511-9
Keywords: prostate cancer, androgen receptor, drug resistance, UHRF1, enzalutamide