Clear Sky Science · en
Phagocytosis by retinal pigment epithelium and microglia does not affect vision restoration by P3HT nanoparticles in Retinitis pigmentosa
New Hope for Inherited Blindness
Retinitis pigmentosa is a leading cause of inherited blindness, yet most people affected are told that nothing can be done once light‑sensing cells in the eye are gone. This study explores an emerging alternative: tiny injectable particles that act like a “liquid prosthetic retina.” The work tests whether these nanoparticles can still restore vision even when the eye’s own cleaning cells are healthy and actively swallowing debris—an important step toward making the technology relevant for real patients.
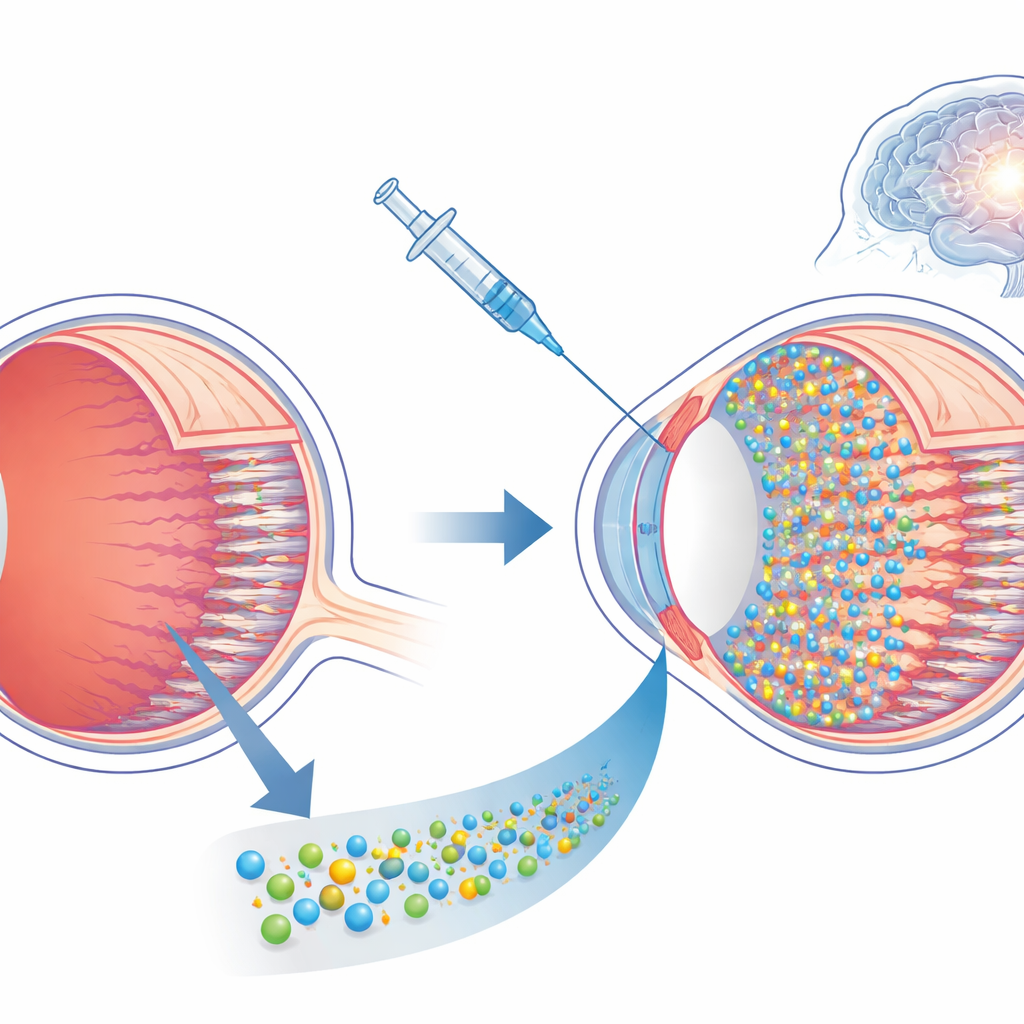
When the Eye’s Camera Fails
In a healthy eye, rod and cone cells at the back of the retina convert light into electrical signals that travel through inner retinal circuits to the brain. In retinitis pigmentosa, inherited gene faults gradually kill these photoreceptors, leaving people first night‑blind, then tunnel‑visioned, and eventually completely blind. Many experimental treatments try to fix or replace the dying cells, but they often depend on knowing the exact gene defect and acting very early in life. Once the rods and cones are gone, most of these options no longer work, and attention turns to prosthetic devices that bypass missing cells and stimulate the remaining retinal network directly.
A Liquid Retinal Prosthesis
The researchers focus on nanoparticles made from a light‑sensitive plastic called P3HT. When these particles sit near nerve cells, flashes of light cause tiny electrical changes on their surface that can nudge nearby neurons into firing. Earlier work showed that injecting P3HT nanoparticles under the retina of a particular rat strain with defective cleaning cells could restore vision‑like responses for many months. But because those rats cannot efficiently clear debris, it was unclear whether the apparent success depended on this defect: would normal cleaning cells in the eye simply swallow and remove the particles in more typical forms of the disease?
Putting Nanoparticles to the Test
To answer this, the team used rd10 mice, a widely used model of retinitis pigmentosa in which rods die early due to a mutation in a rod‑specific enzyme, while the retinal pigment epithelium (a support layer) and microglia (immune‑like cells) remain functional. The scientists waited until the disease was in its final stage—rods completely lost, cones reduced to scattered stumps, and inner retinal circuits extensively rewired—so that any recovery could be attributed to the nanoparticles rather than surviving photoreceptors. They injected a tiny volume of P3HT nanoparticle suspension under the retina and followed the animals for up to four months, comparing them with untreated mice and with animals given inert glass particles of similar size.
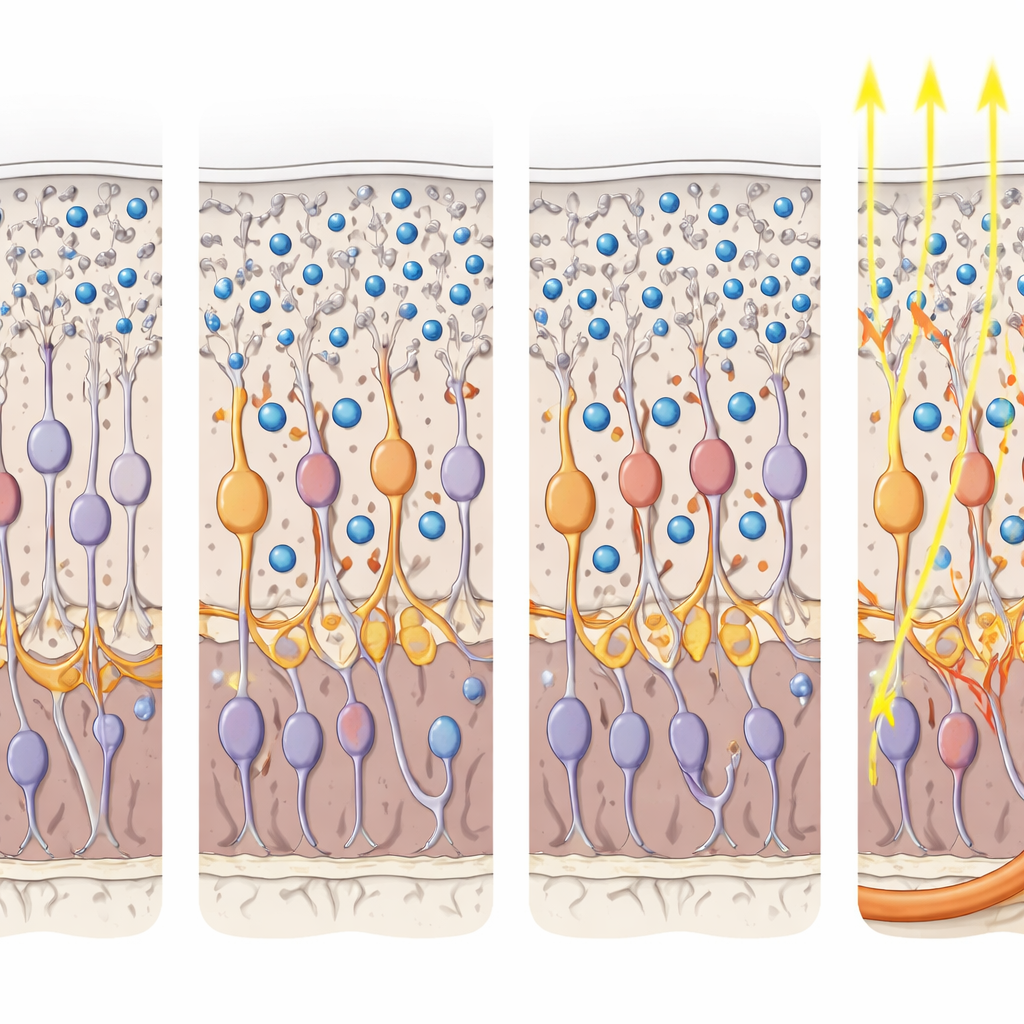
Surviving the Eye’s Cleanup Crew
High‑resolution imaging showed that a single injection spread P3HT nanoparticles across roughly 80 percent of the retinal surface. Most particles remained in the outer retina, nestled among the processes of second‑order neurons, while only about 30 percent were taken up by the pigment epithelium and less than 5 percent by microglia. Importantly, this partial engulfment did not damage these support and immune cells or provoke extra inflammation; their shape and density were similar with and without nanoparticles. In short, even in an eye with active cellular “vacuum cleaners,” a large, stable population of particles persisted in the right place to influence the surviving retinal circuitry.
From Light to Behavior and Brain Activity
The critical question was whether these resident particles actually restored useful vision. Multiple tests gave a converging yes. In an optomotor test, where mice reflexively track moving stripes, P3HT‑treated rd10 mice regained responses at spatial detail levels that untreated or sham‑treated blind mice could not detect, approaching the performance of healthy animals. In a classical conditioning task, mice learned to associate brief light flashes with a mild foot shock. Only healthy mice and P3HT‑treated rd10 mice later froze in anticipation when they saw the light alone, indicating that light‑driven signals reached and were processed by higher brain centers to form implicit visual memories. Electrical recordings from the primary visual cortex confirmed this picture: after treatment, formerly blind mice showed robust, time‑locked responses to light flashes and patterned gratings, with visual acuity matching that of age‑matched normal mice, although with somewhat reduced signal strength.
What This Means for Future Therapies
Taken together, the findings show that injectable P3HT nanoparticles can restore complex visual functions—even at late, highly degenerated stages of retinitis pigmentosa—in an eye with normal cleaning activity. The particles are not rapidly cleared or overtly toxic, and they can drive behavior and brain responses that resemble those of sighted animals. For people living with advanced inherited blindness, this suggests that a gene‑agnostic, minimally invasive “liquid prosthesis” could one day complement or substitute current approaches, offering a way to reawaken dormant visual circuits long after the original light‑sensing cells have disappeared.
Citation: Mantero, G., Francia, S., Galluzzi, F. et al. Phagocytosis by retinal pigment epithelium and microglia does not affect vision restoration by P3HT nanoparticles in Retinitis pigmentosa. Cell Death Dis 17, 295 (2026). https://doi.org/10.1038/s41419-026-08510-w
Keywords: retinitis pigmentosa, retinal prosthesis, nanoparticles, vision restoration, retinal degeneration