Clear Sky Science · en
Bone adhesive with temporally-synchronized degradation for enhanced osteointegration
Glue that Helps Broken Bones Heal Better
When a bone breaks into awkward pieces, surgeons often rely on metal plates and screws to hold everything together. These devices work well but can be bulky, require second surgeries for removal, and don’t always match the way bone naturally heals. This study introduces a new type of bone “superglue” that not only holds fragments firmly in place but is designed to disappear in step with the body’s own repair process, potentially making fracture treatment gentler and more effective.
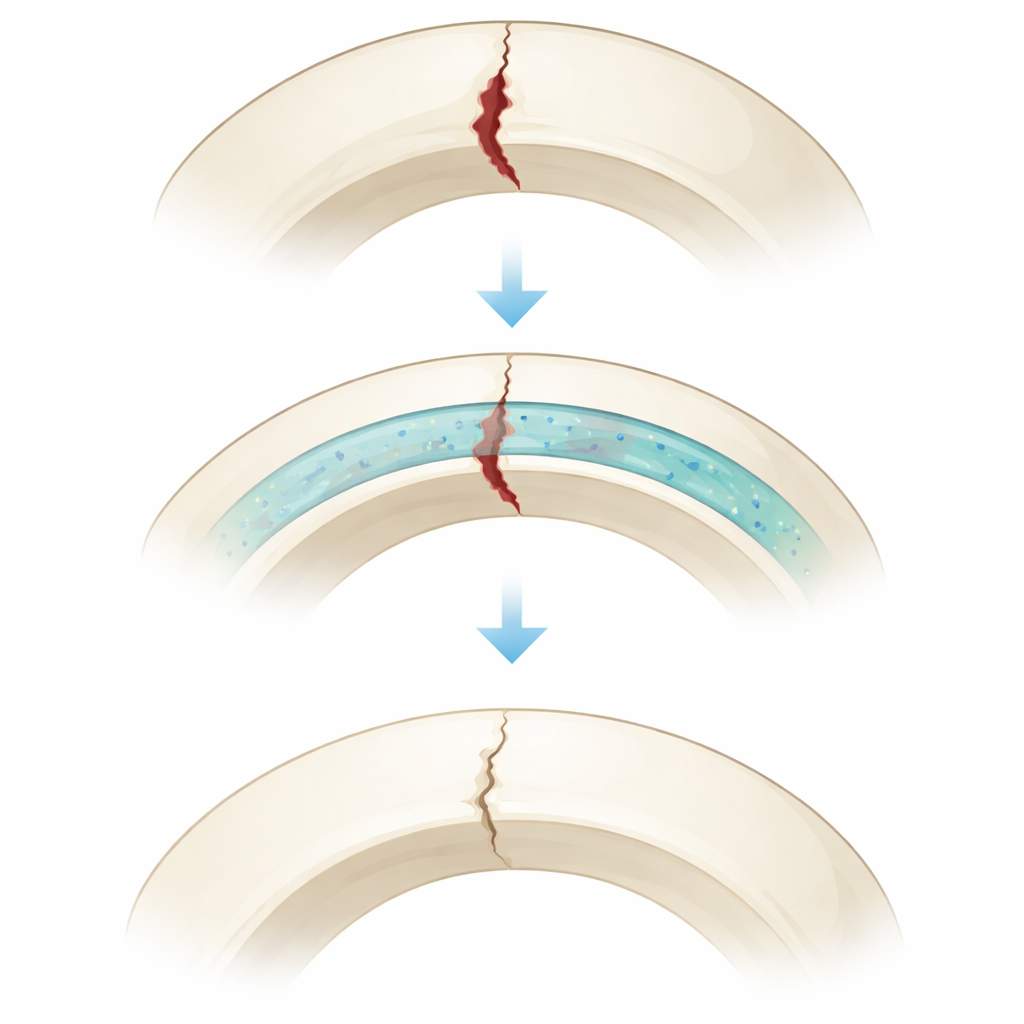
Why Bones Need a Smarter Glue
Traditional materials called bone cements or tissue glues have important drawbacks. Some stick poorly to hard bone, others are strong but take too long to set or linger in the body long after they are needed. If an adhesive disappears too quickly, the fracture can loosen; if it stays too long, it can block the growth of new bone and blood vessels. Natural bone healing passes through well-defined stages, from early clot and soft tissue formation to hard callus and finally slow reshaping of the bone. The key idea behind this work is that a truly advanced bone adhesive should match these stages: very stable at the beginning, then breaking down faster only when the bone is strong enough to take over.
Building a Bone-Friendly Superglue
The researchers created a polyurethane-based material they call TNC, made from three building blocks: a reactive connector molecule (tri-HDI), tiny particles of bone-like mineral (nano-hydroxyapatite), and type I collagen, the main protein in natural bone. Mixed together, these ingredients form a glue that starts as a viscous liquid and hardens within about ten minutes at body-like temperatures, avoiding the heat spikes that can damage tissue. By adjusting how much collagen they added, the team tuned the internal pore structure of the material. Higher collagen levels created a more open, sponge-like glue that was easier for cells and fluids to enter, while lower collagen gave higher mechanical strength. Across versions, the adhesive showed strong bonding to both dense and spongy bone, even when surfaces were wet or contaminated with blood, demonstrating performance that rivals or exceeds existing experimental bone adhesives.
Safe for Cells and Kind to the Body
Any implant must be friendly to living tissue. In lab tests, bone-forming cells grown with extracts of the TNC glue remained highly viable, and the material caused almost no damage to red blood cells. Under the microscope, cells migrated deep into the porous structure, especially in the more open, high-collagen version, suggesting that the glue can act as a scaffold for new tissue. In rats, small TNC samples placed under the skin did not harm major organs or disturb routine blood markers. Instead, host cells and connective tissue grew into the material’s pores, showing that the body tolerated the adhesive well and began to integrate it rather than wall it off.
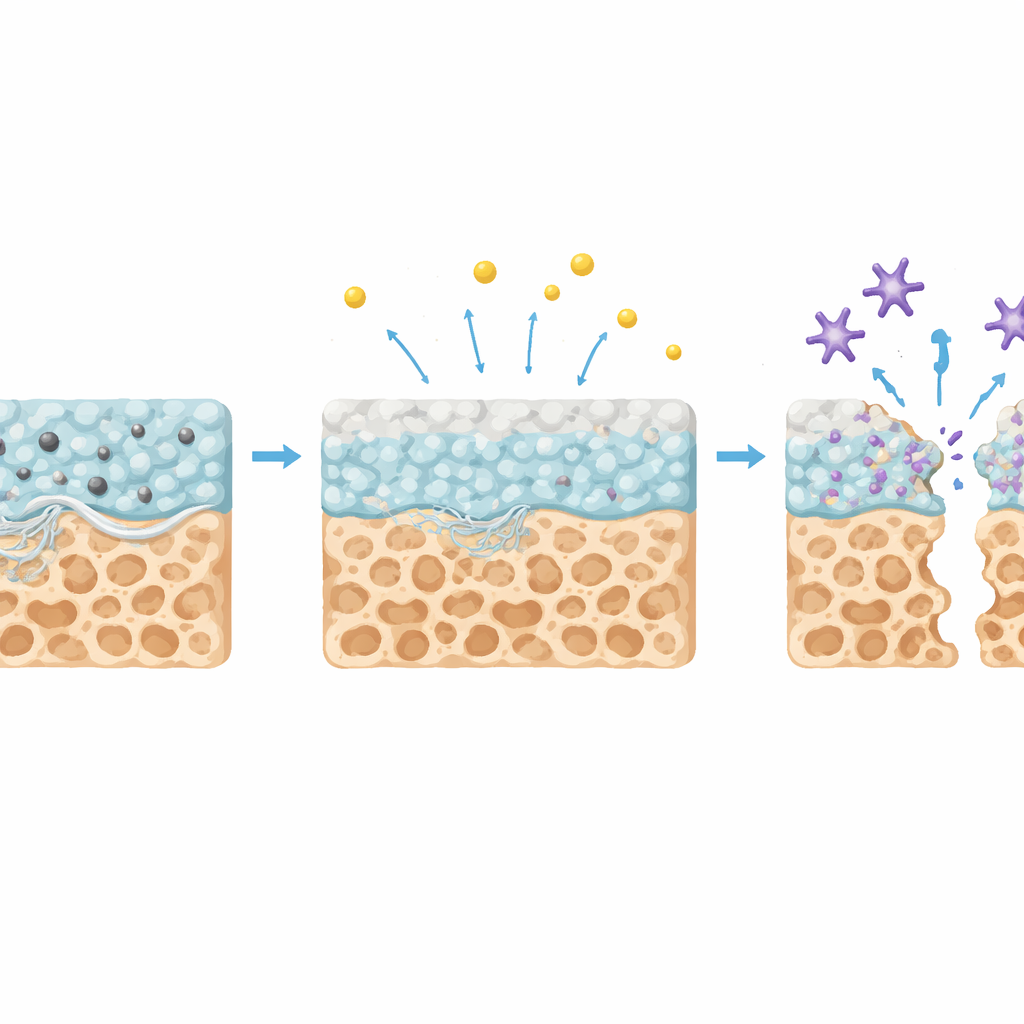
A Glue that Speeds Its Own Disappearance
The most innovative feature of TNC is how it degrades over time. During normal bone remodeling, specialized cells release an enzyme called cathepsin K that helps dissolve old bone matrix. The team showed that, in the presence of this enzyme, TNC initially breaks down slowly, then after several weeks speeds up its own degradation by forming a thin mineral layer rich in calcium and phosphate on its surface. This layer releases calcium ions, which in turn change the shape and flexibility of cathepsin K, making the enzyme more active. As a result, the glue’s breakdown rate nearly doubles in the later phase, just when natural bone remodeling is most active. At the same time, the material’s mineral content and porous, water-loving structure encourage bone-forming cells to deposit new mineral, further supporting repair.
Helping Broken Skulls Heal in Animals
To see whether this concept works in living bone, the researchers created circular skull fractures in rats and either left them untreated, fixed them with a collagen-only glue, or used the full TNC formulation with mineral particles. Without any adhesive, the bone fragments stayed misaligned and failed to heal properly. The collagen-only glue provided some stabilization but degraded too slowly, leaving much of the material in place and limiting new bone growth and blood vessel formation. In contrast, TNC held the fragments securely, built up a mineral surface layer, and then degraded in a timely way that opened space for blood vessels and fresh bone. Imaging and tissue stains showed more complete bridging of the fracture, higher bone volume and density, and strong signs of new bone and vessel formation in the TNC group, while only small remnants of the glue remained.
What This Could Mean for Future Fracture Care
In everyday terms, this study describes a bone glue that behaves like a well-timed temporary scaffold. It grabs and holds broken pieces firmly when they are fragile, then senses the body’s own remodeling signals and steps out of the way as new bone takes over. By combining strong early fixation, good biocompatibility, and a built-in trigger for faster late-stage degradation, TNC points to a new generation of “smart” materials for mending complex fractures. Although more testing in different bones and larger animals is needed, the approach suggests that, in the future, surgeons may fix difficult breaks with injectable glues that safely vanish as the patient’s own skeleton is restored.
Citation: Gu, Jt., Li, Zt., Wang, Yz. et al. Bone adhesive with temporally-synchronized degradation for enhanced osteointegration. Bone Res 14, 39 (2026). https://doi.org/10.1038/s41413-026-00522-8
Keywords: bone adhesive, fracture healing, biodegradable implant, bone regeneration, smart biomaterial