Clear Sky Science · en
The mitigation of activity-based anorexia by obese adipose tissue transplant is abolished by neonatal AgRP neuron ablation
Why this research matters
Anorexia nervosa is one of the deadliest psychiatric illnesses, largely because patients struggle to gain and keep weight on, even with intensive care. Doctors still lack medications that reliably restore healthy weight. This study uses a powerful mouse model of anorexia-like behavior to explore an unexpected ally in the fight against dangerous weight loss: body fat itself. By asking whether fat from previously obese animals can protect against extreme slimming, the researchers uncover a surprising conversation between fat tissue and the brain that could inspire new treatments.
A lab model of self-starvation
To probe the biology of anorexia-like states, scientists often use the “activity-based anorexia” (ABA) model. In this setup, otherwise healthy rodents get access to a running wheel but can eat only during a short daily window. Many respond by running more and eating less, rapidly losing weight and eventually dropping out of the experiment when they lose a quarter of their starting body mass. This model mimics key features of human anorexia nervosa: restrictive eating, compulsive exercise, and dramatic weight loss, especially in younger females. Because the conditions are controlled, ABA lets researchers test which body systems help animals withstand or succumb to this spiral.
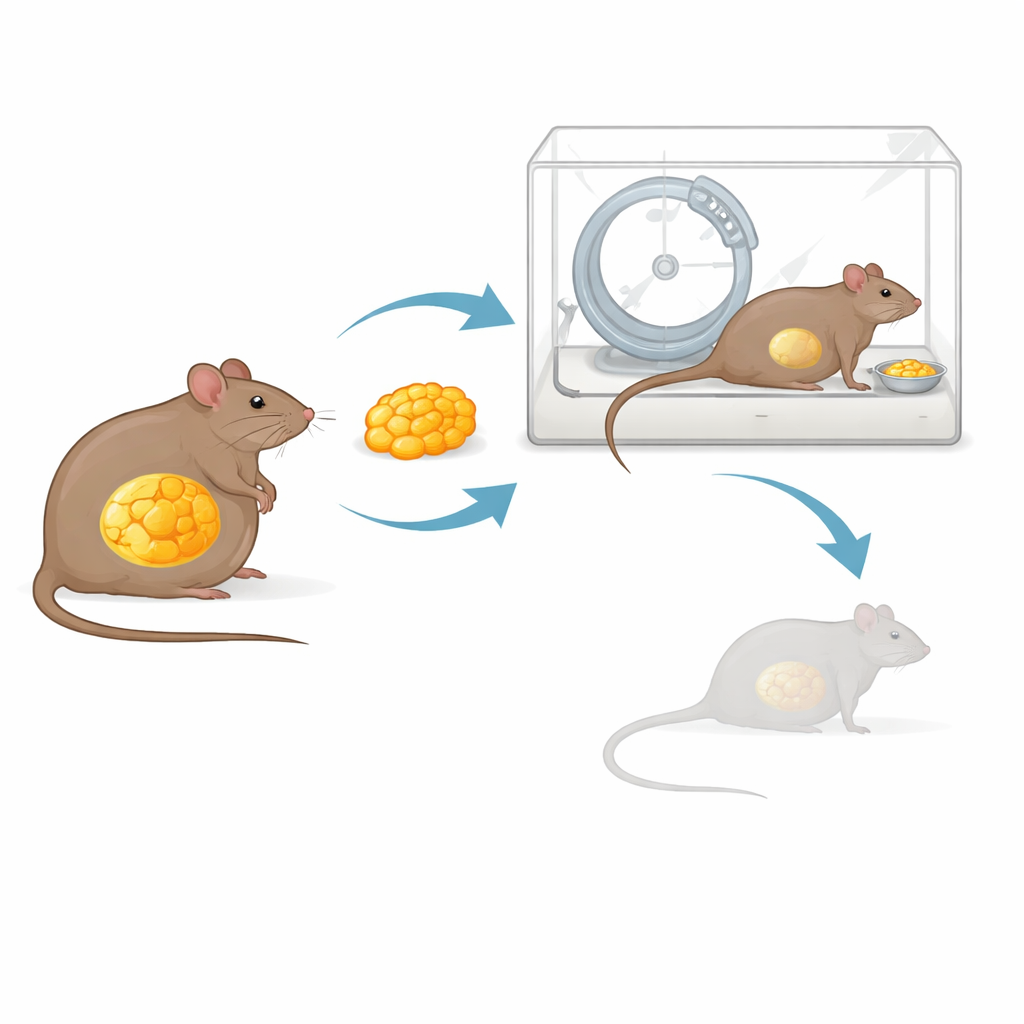
Obese fat as a protective transplant
Recent metabolic work has shown that white fat tissue is not just a passive storage depot, but an endocrine organ that remembers past obesity. Even after weight loss, fat from formerly obese animals keeps a distinct molecular “signature” and continues to send signals that favor regaining weight. The authors wondered whether that same memory could be turned to advantage—could transplanted fat from obese mice help normal-weight females resist ABA-driven wasting? They took fat from the abdomen of mice made obese on a high-fat diet and surgically attached it inside the bellies of lean recipient mice. Control animals received the same amount of fat from lean donors. After several weeks of recovery, all mice entered the ABA protocol with running wheels and limited food access.
Fat-to-brain signaling boosts survival
The results were striking. Mice that received obese fat transplants stayed in the ABA experiment longer before reaching the critical 25 percent weight-loss threshold. In other words, they “survived” better under the demanding conditions than mice given lean fat. Across two independent experiments, transplanted obese fat helped mice maintain higher body weights during restriction and showed hints of promoting greater food intake, without consistently changing how much they ran on their wheels. These patterns suggest the transplanted fat somehow encouraged the animals to conserve energy and eat more, buffering them against the self-starvation spiral, rather than simply making them less active.
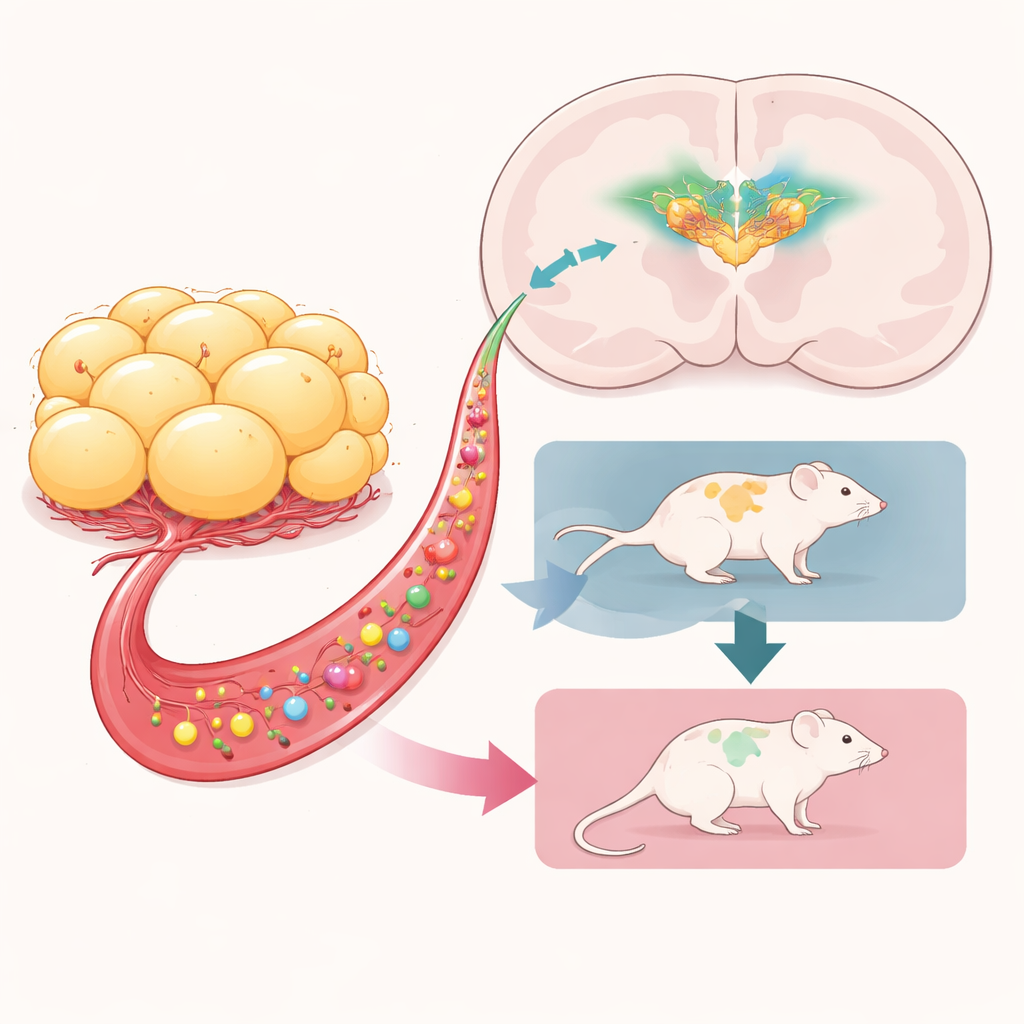
A key role for hunger-sensing brain cells
The team then asked which brain circuits were required for this protective effect. They focused on AgRP neurons, a small group of cells deep in the hypothalamus that sense energy shortage and drive food-seeking, fuel conservation, and metabolic shifts. Using a genetic trick, they selectively wiped out these neurons shortly after birth in some mice, leaving others intact. All of these animals later received either obese or lean fat transplants and were run through the same ABA protocol. When AgRP neurons were intact, obese fat again helped mice maintain weight and survive longer. But when these neurons had been ablated in infancy, the benefit vanished: mice with obese fat and missing AgRP neurons lost weight quickly and left the experiment even faster than comparable controls.
What this means for future treatment ideas
Taken together, the findings point to a direct line of communication from obese adipose tissue to AgRP neurons in the brain, which together act to protect against extreme weight loss under stress. Signals released by the transplanted fat—likely a mixture of hormones and other circulating molecules shaped by prior obesity—appear to activate these hunger-related neurons, nudging the body toward eating more and conserving energy. While surgically transplanting fat from obese donors into people is not a practical therapy, identifying the specific factors involved could inspire new drugs that safely tap into the body’s own weight-preserving circuitry. For patients with anorexia nervosa, whose lives are endangered by relentless weight loss, such fat-derived signals might someday help tip the balance back toward recovery.
Citation: Yoon, D.J., Zhang, J., Zapata, R.C. et al. The mitigation of activity-based anorexia by obese adipose tissue transplant is abolished by neonatal AgRP neuron ablation. Transl Psychiatry 16, 199 (2026). https://doi.org/10.1038/s41398-026-03970-2
Keywords: anorexia nervosa, activity-based anorexia, white adipose tissue, AgRP neurons, metabolic signaling