Clear Sky Science · en
Knockdown of Ddx3x in mPFC induces autistic-like phenotype in mice via altered synaptic plasticity
When Gene Changes Shape Social Behavior
Why do some gene changes lead to differences in social behavior, learning, and sensory responses that we call autism? This study zooms in on one such gene, DDX3X, and asks a simple but powerful question: what happens in the brain when this gene does not work properly? By studying brain cells in a dish and carefully engineered mice, the researchers trace a path from a tiny molecular fault to changes in brain wiring and, finally, to autism-like behaviors.
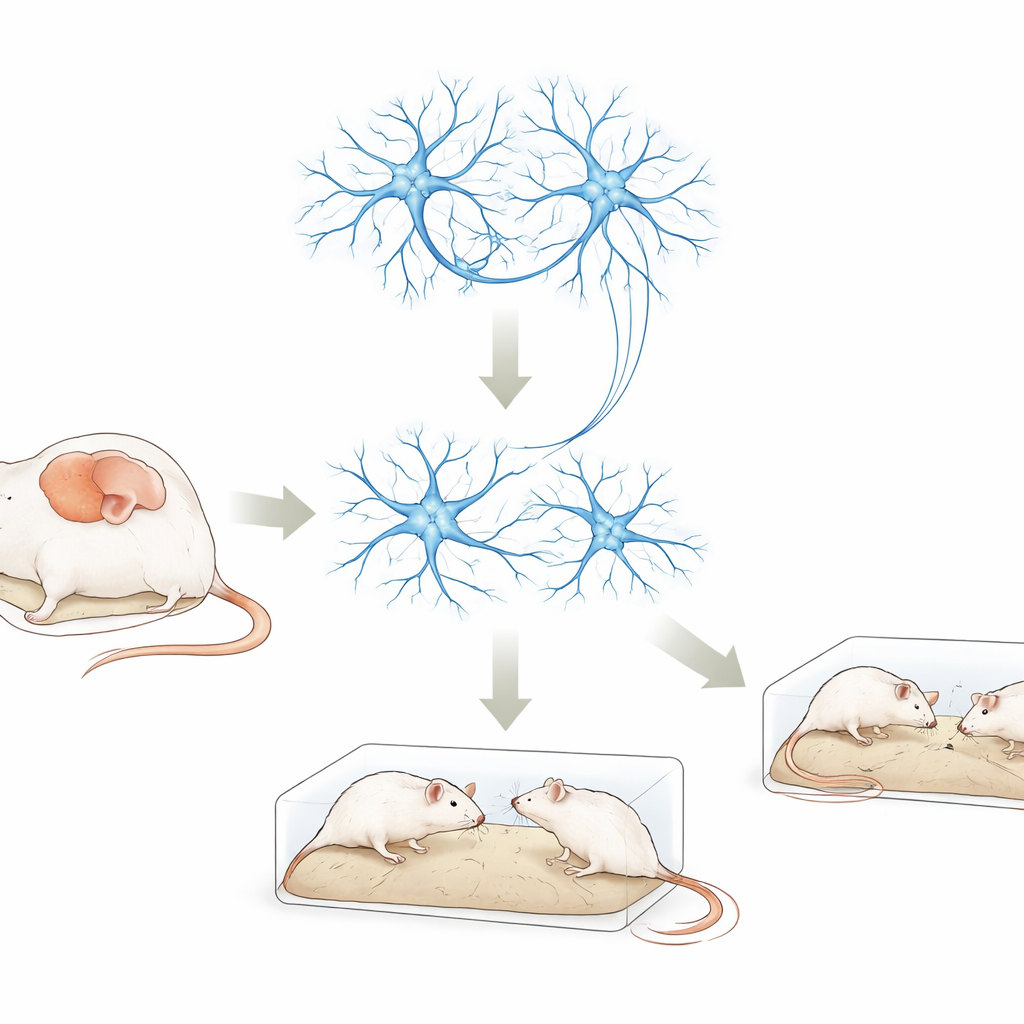
A Key Brain Gene Under the Microscope
DDX3X is a gene that helps cells handle RNA, the working copy of our genetic code used to build proteins. Variations in DDX3X are increasingly found in people with autism, intellectual disability, and developmental delay. Yet it has been unclear how these changes disturb brain function. In this work, scientists reduced the activity of the mouse version of the gene, called Ddx3x, in two settings: cultured nerve-like cells and a specific brain area in living mice, the medial prefrontal cortex. This region is heavily involved in social interaction, decision making, and memory—abilities often affected in autism.
From Slower-Growing Cells to Social Changes in Mice
When the team lowered Ddx3x levels in nerve-like cells grown in the lab, the cells grew more slowly and formed connections less readily than normal cells. In mice, they used a harmless virus as a delivery tool to reduce Ddx3x only in neurons of the medial prefrontal cortex. These mice were then put through a battery of behavioral tests. Compared with control animals, Ddx3x-knockdown mice moved normally and were not unusually anxious in an open field, but they showed weaker interest in an unfamiliar mouse, were less drawn to a new social partner, and performed worse on a memory-heavy maze task when the delay was longer. They also displayed more repetitive digging and burying behavior—paralleling two core features of autism: social difficulties and repetitive routines.
Hidden Shifts in the Brain’s Protein Landscape
To see what was changing inside cells, the researchers conducted large-scale protein surveys in both the cultured cells and the mouse medial prefrontal cortex. They found hundreds of proteins whose levels shifted when Ddx3x was reduced. Many of the most affected proteins were involved in how synapses—the junctions where nerve cells communicate—adapt and strengthen over time, a property known as synaptic plasticity. Pathways linked to learning-related strengthening of connections, chemical messaging by glutamate and GABA (the brain’s main excitatory and inhibitory signals), and hormone and energy metabolism all showed signs of being dialed down. In both cells and brain tissue, they also saw overlapping disruptions in systems that fold new proteins correctly and tag worn-out proteins for removal, suggesting a broad breakdown in protein quality control.
Weakened Connections Between Nerve Cells
Looking more closely at the structure of synapses, the team used high-resolution microscopes to examine neurons in the affected brain region. They found that mice with reduced Ddx3x had fewer dendritic spines—the tiny protrusions on nerve cell branches where excitatory synapses sit—especially the mature, mushroom-shaped spines that are crucial for stable, strong connections. The postsynaptic density, a protein-rich zone that anchors receptors and signaling molecules, was thinner and smaller. Electrical recordings from brain slices showed that excitatory signals arrived less frequently, even though their individual strength was unchanged. Together, these findings point to a brain network that is wired with fewer and weaker communication points, rather than one that is entirely silent.
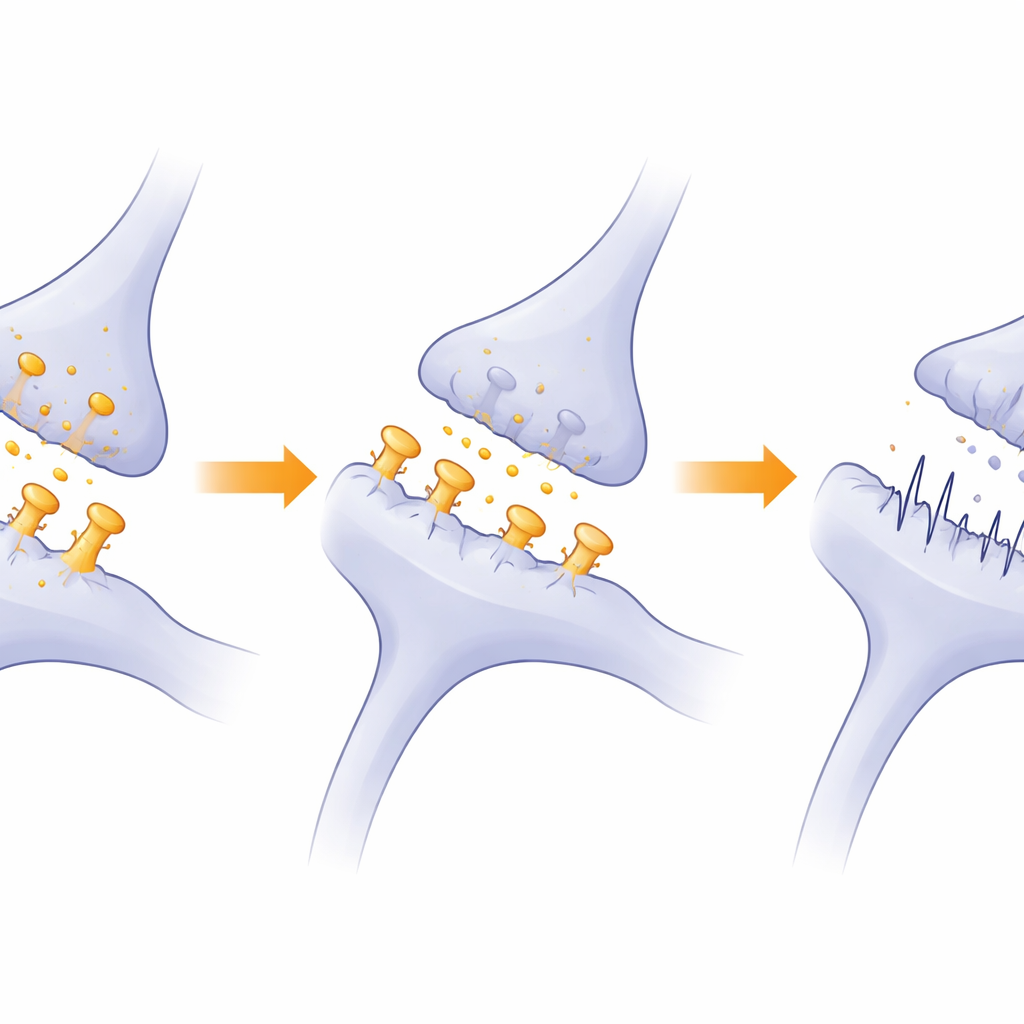
How Disrupted Protein Balance May Fuel Autism Traits
Beyond individual synapses, the study highlights a deeper problem: the machinery that keeps synaptic proteins in balance appears to be suppressed when Ddx3x is low. Systems that fold fresh proteins and those that break down damaged ones—especially the ubiquitin–proteasome pathway—were less active. Because synapses rely on constant renewal and fine-tuning of their protein components, this slowdown can gradually erode synaptic strength and flexibility. The authors propose that this combination of fewer mature spines, dampened signaling, and impaired protein housekeeping in the medial prefrontal cortex helps explain the social and cognitive changes seen in their mice. In simple terms, when DDX3X falters, brain circuits that support social behavior and learning cannot build or maintain the connections they need, nudging development toward an autism-like trajectory.
Citation: Zhuang, H., Cao, X., Tang, X. et al. Knockdown of Ddx3x in mPFC induces autistic-like phenotype in mice via altered synaptic plasticity. Transl Psychiatry 16, 216 (2026). https://doi.org/10.1038/s41398-026-03945-3
Keywords: DDX3X, autism spectrum disorder, synaptic plasticity, medial prefrontal cortex, ubiquitin proteasome system