Clear Sky Science · en
Acyl-CoA-binding protein (ACBP): a poor-prognosis biomarker in sepsis and a target for disease mitigation
Why this matters for people with severe infections
Sepsis, a life‑threatening reaction to infection, remains one of the biggest killers in intensive care units. Even with modern antibiotics and life support, many patients die and survivors often face long‑term health problems. This study identifies a small protein in the blood, called acyl‑CoA‑binding protein (ACBP), as both a warning signal of danger and an active driver of the damage seen in sepsis. It also suggests a new treatment strategy that could work alongside existing steroid therapy to save more lives.
A stress signal that turns harmful
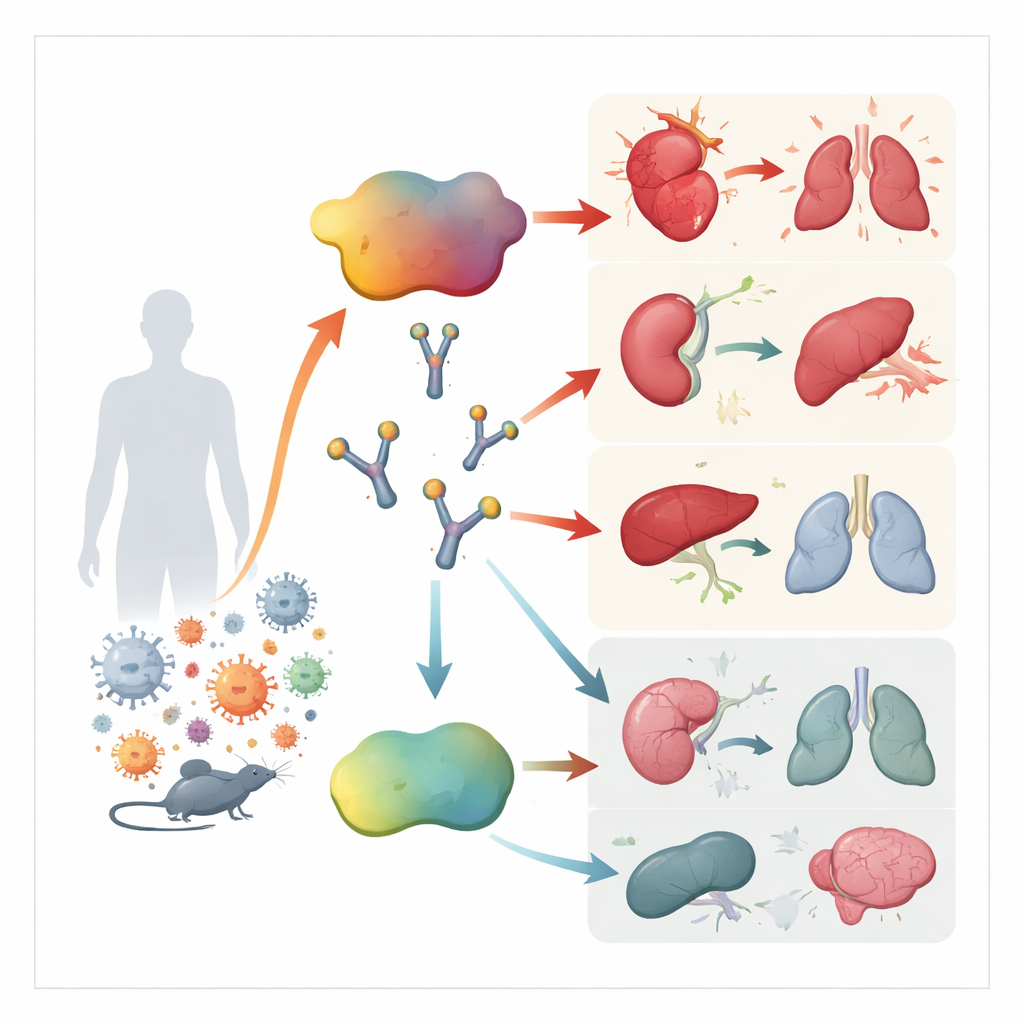
ACBP is normally involved in handling fats inside cells, but it can also be released into the bloodstream as a kind of stress hormone. Earlier work showed that high levels of this protein appear in people with inflammatory diseases, and that it can push immune cells to release more inflammatory molecules. The authors wondered whether ACBP might be part of the runaway immune reaction that characterizes sepsis, and whether blocking it could help restore balance.
A blood marker that tracks who gets sicker
The team first measured ACBP levels in two groups of hospital patients: a small “discovery” group in intensive care and a much larger “validation” group seen in the emergency department. In both groups, people with sepsis and septic shock had much higher ACBP levels than healthy volunteers or patients with non‑infectious inflammation. Within the sepsis groups, those who eventually died tended to have the highest ACBP concentrations. These levels tracked closely with established severity scores and with blood signs of heart, kidney, and liver injury. In fact, ACBP predicted death about as well as complex scoring systems used in critical care, and better than standard blood tests like C‑reactive protein and procalcitonin.
Blocking the protein protects organs in animals
To see whether ACBP was just a marker or actually part of the problem, the researchers turned to several mouse models of sepsis. They studied a sterile model driven by bacterial toxin, a live Escherichia coli infection, and a polymicrobial “dirty surgery” model that closely mimics human abdominal sepsis. In all of these, ACBP levels in the blood rose sharply after challenge. When mice were given a monoclonal antibody that neutralizes ACBP, or when the gene for ACBP was deleted, the animals were far more likely to survive. They maintained body temperature better, released fewer inflammatory cytokines, and had less evidence of heart, kidney, liver, and lung damage. Conversely, adding extra ACBP made the toxin‑induced model more deadly, underscoring its harmful role.
Helping immune cells clear bacteria more efficiently
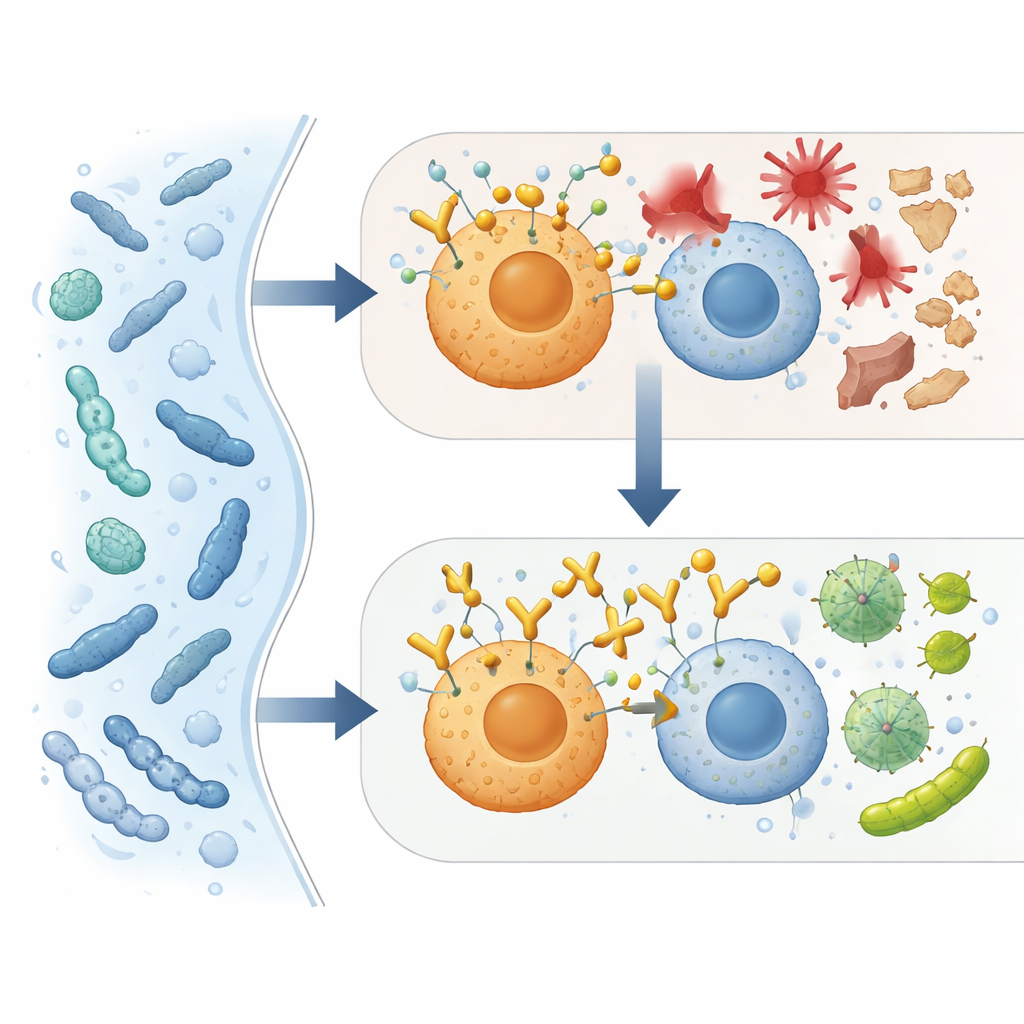
The study also explored how ACBP affects front‑line immune cells. In mice infected with E. coli or subjected to polymicrobial peritonitis, blocking ACBP reduced the number of live bacteria in blood, peritoneal fluid, and organs. When macrophages or neutrophils were depleted, this benefit largely disappeared, pointing to these cells as key players. In dishes, the antibody made neutrophils and a subset of macrophages more efficient at swallowing and killing bacteria. Detailed immune profiling in septic mice revealed that ACBP neutralization shifted myeloid cells away from strongly inflammatory states toward more tissue‑protective profiles, while broad gene‑expression and metabolite analyses showed a move back toward normal function across several organs.
Teaming up with steroids instead of working against them
Many patients with septic shock receive glucocorticoid drugs such as dexamethasone to dampen excessive inflammation, but these steroids can also raise ACBP levels and have damaging metabolic side effects. The authors tested whether blocking ACBP would clash with or complement steroid treatment in mice. In both toxin‑driven and surgical sepsis models, combining the anti‑ACBP antibody with dexamethasone generally improved survival, clinical scores, and organ function compared with either therapy alone. Importantly, the beneficial anti‑inflammatory actions of the steroid were preserved, while molecular signs of harmful metabolic rewiring were normalized by ACBP blockade. These findings suggest that ACBP inhibition could separate the short‑term benefits of steroids from their longer‑term liabilities.
What this could mean for future sepsis care
Overall, the work paints ACBP as a central amplifier of the damaging responses that turn a severe infection into deadly sepsis. High blood levels of the protein warn clinicians that a patient is at greater risk of organ failure and death, while experiments in mice show that neutralizing ACBP can both calm inflammatory overreaction and strengthen the body’s ability to clear invading microbes. Although these results still need to be confirmed in human trials, they point toward a future in which a targeted antibody against ACBP, possibly combined with standard steroids, could help interrupt the fatal spiral of septic shock and improve both survival and long‑term recovery.
Citation: Lambertucci, F., Motiño, O., Nogueira-Recalde, U. et al. Acyl-CoA-binding protein (ACBP): a poor-prognosis biomarker in sepsis and a target for disease mitigation. Sig Transduct Target Ther 11, 119 (2026). https://doi.org/10.1038/s41392-026-02670-z
Keywords: sepsis, biomarkers, immune response, monoclonal antibodies, glucocorticoids