Clear Sky Science · en
Disinhibition of ventral tegmental area during initial punishment learning causes enduring punishment insensitivity
Why we sometimes ignore bad consequences
Most of us learn quickly to stop doing things that bring pain or trouble. Yet some people, and many animals, keep chasing rewards even when the cost is high, as in addiction or risky decision making. This study asks what happens inside a key reward center of the brain during the first moments we learn that an action has painful consequences, and how disrupting that process can leave us stubbornly insensitive to punishment long after the danger is clear.
A brain hub for weighing reward against harm
Deep in the midbrain lies the ventral tegmental area, a small region whose dopamine-releasing cells broadcast powerful “teaching signals” about rewards. These neurons burst into action when things turn out better than expected and quiet down when outcomes are worse. They are surrounded by inhibitory cells that use the chemical GABA to briefly silence dopamine output. Classic theories argue that this inhibition helps animals learn to avoid harmful actions. But researchers did not know exactly how GABA and dopamine signals in this region behave during punishment, or whether the brief silencing of dopamine cells is truly required for learning to steer clear of danger.
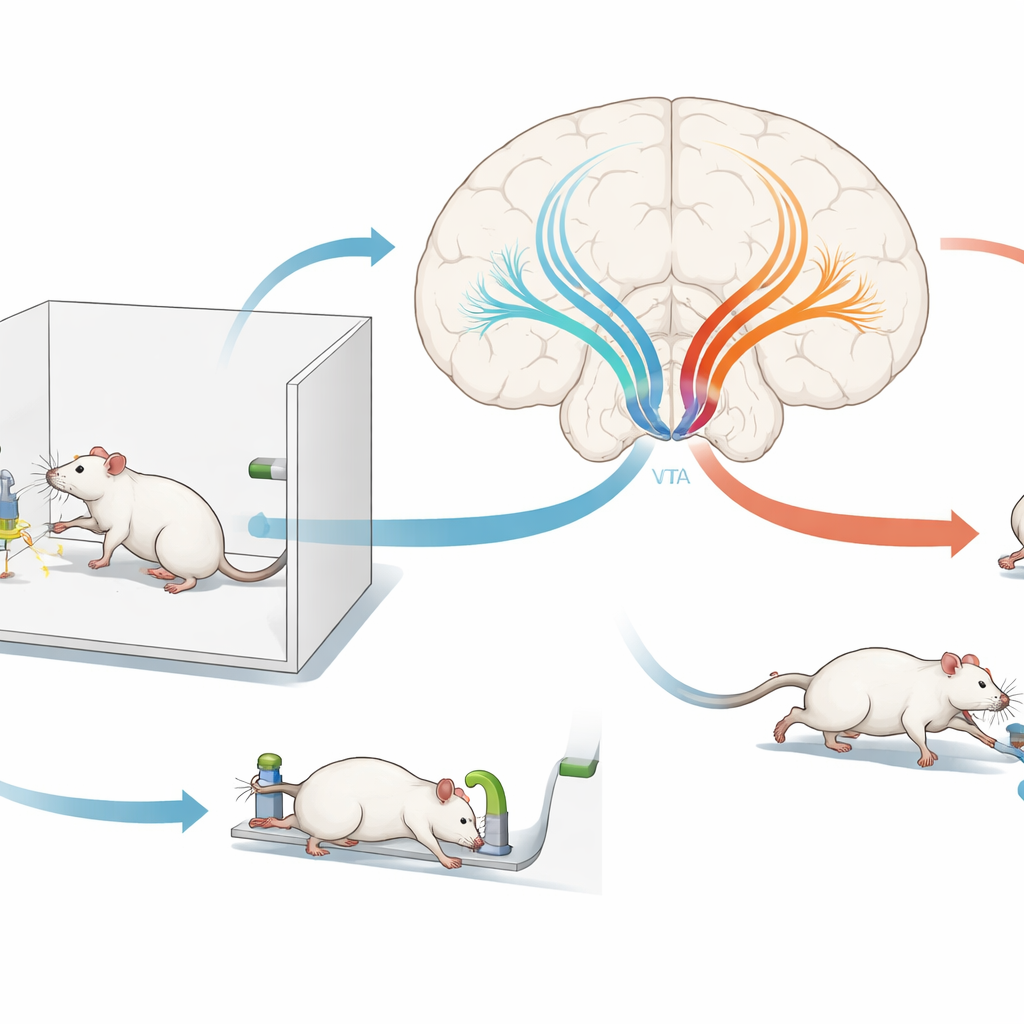
Watching punishment signals in real time
The authors trained rats to press two levers for food. Later, presses on one lever also triggered a mild electric shock, turning that action into a punished choice, while the other lever remained safe. Using fiber-optic recording tools, the team measured activity in dopamine cells and the GABA input they received as the animals experienced shocks and rewards and decided which lever to press. Both dopamine activity and GABA input briefly spiked when food or shock occurred. Dopamine cells responded more strongly to food, whereas GABA input was especially strong for shocks during the very first punishment session and then faded with experience. Around actions themselves, activity patterns shifted so that presses on the punished lever came to evoke a distinct burst of dopamine activity, while the safe lever did not. These patterns suggested that a surge of GABA-driven inhibition right when punishment is first encountered might be a crucial teaching signal.
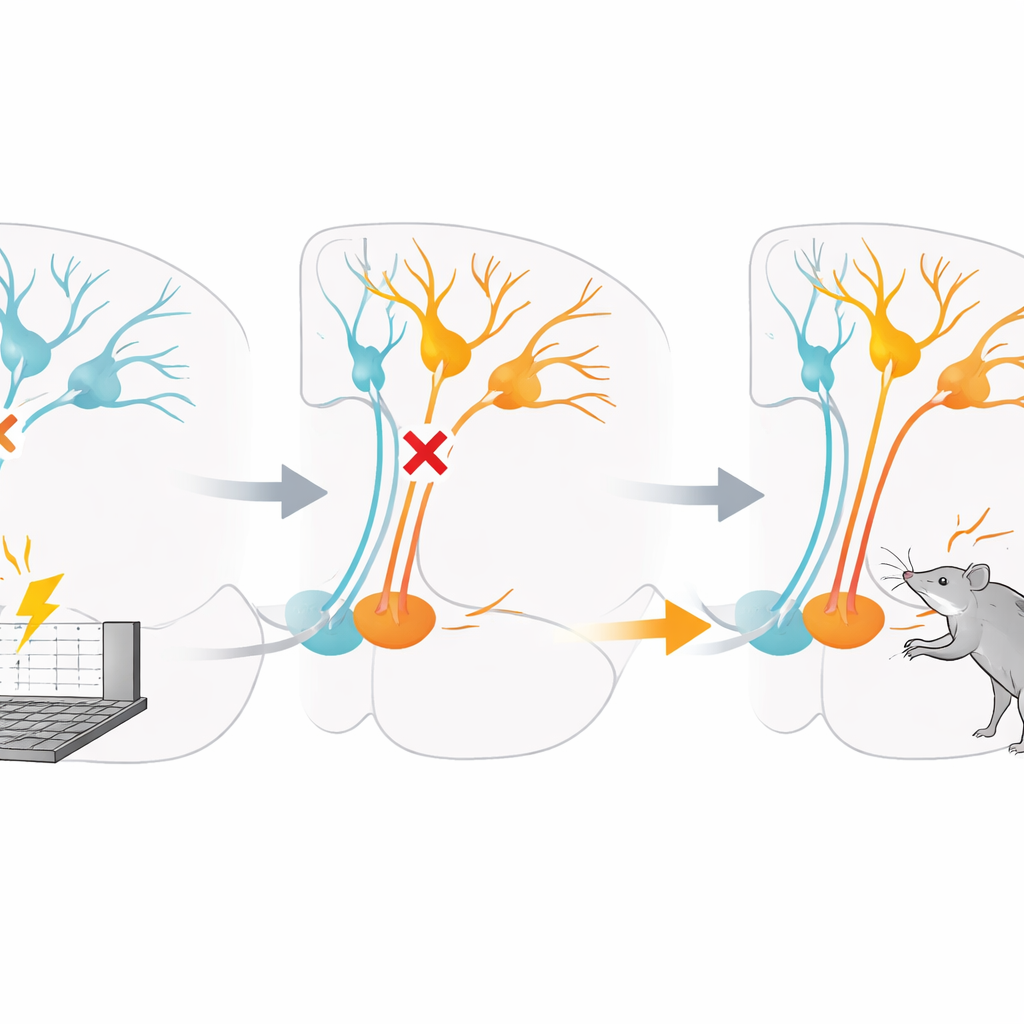
Blocking inhibition during a critical window
To test this idea, the researchers next interfered with GABA’s ability to inhibit cells in the ventral tegmental area. In one set of rats, they infused a drug directly into this region that blocked GABA type A receptors during the first two punishment sessions. In another set, they used a designer receptor technique to artificially excite dopamine neurons during the same early sessions. In both cases, the immediate effect was that rats did not reduce pressing on the punished lever as much as control animals and ended up receiving more shocks. Strikingly, once this early window had passed, restoring normal brain chemistry did not fix the problem: even on later days without any drug, these rats continued to press the punished lever more readily and to hesitate less before doing so.
Lasting changes in how danger is valued
When punishment learning was already well established, temporarily blocking inhibition or exciting dopamine cells no longer produced the same lasting damage. Manipulations at that later stage could shift overall activity levels, such as making animals more or less active, but they did not erase the learned tendency to avoid the punished lever. Additional tests showed that the treatment did not simply make rewards more attractive across the board; unpunished food-seeking was not consistently increased. Instead, the disruption seemed to specifically interfere with how the brain linked a particular action to its painful outcome during that first encounter, leaving the animals with a long-term blind spot for that danger.
What this means for real-world risky choices
To a lay reader, the takeaway is that there appears to be a brief but powerful learning window, right when we first experience that a choice has harmful consequences, during which precise inhibition in a reward hub of the brain teaches us to back off. If that inhibitory signal is blunted and dopamine activity stays high, the brain may fail to properly register the danger, producing a lasting tendency to keep making the risky choice even when it hurts. Because many addictive drugs boost dopamine or reduce its inhibition, these findings suggest one way such substances could wire the brain toward persistent, punishment-insensitive habits—and hint that protecting or restoring this early teaching signal might be key to helping people relearn to avoid harmful behaviors.
Citation: Tan, S.Y.S., Shen, M.H., Keevers, L.J. et al. Disinhibition of ventral tegmental area during initial punishment learning causes enduring punishment insensitivity. Neuropsychopharmacol. 51, 1045–1055 (2026). https://doi.org/10.1038/s41386-026-02368-4
Keywords: dopamine, punishment learning, ventral tegmental area, addiction, risk taking