Clear Sky Science · en
A STAT3 degrader demonstrates efficacy in venetoclax resistant acute myeloid leukemia
Why drug resistance in blood cancer matters
For people with acute myeloid leukemia (AML), a fast‑moving cancer of the blood and bone marrow, new medicines have improved early responses but not long‑term survival. One widely used drug, venetoclax, can push the disease into remission, yet many patients eventually relapse as their cancer cells learn to bypass the treatment. This study explores why that escape happens and tests a new type of drug designed to dismantle a key survival switch inside leukemia cells, opening a possible path for patients who currently have few options.
A powerful switch that drives diseased blood cells
The researchers focused on a protein called STAT3, which acts as an on–off switch for many genes that control how blood cells grow and survive. When STAT3 is abnormally active, it pushes immature blood cells to multiply and resist normal death signals. Using genetically engineered mice whose blood‑forming cells produce a constantly active form of STAT3, the team showed that STAT3 alone can drive a myeloid blood cancer that closely resembles human AML. These mice developed enlarged spleens and livers packed with immature white blood cells, along with a strong bias toward producing myeloid cells instead of healthy balanced blood. This established STAT3 as not just a bystander but a direct engine of leukemia.
How venetoclax resistance hijacks cell power plants
Venetoclax works by blocking BCL2, a protein that keeps damaged cells from dying. Over time, however, surviving leukemia cells often switch to other backup proteins and pathways. The team found that in cell lines made resistant to venetoclax, STAT3 levels rose sharply, along with its partner MCL1, another strong protector against cell death. Detailed patient data from hundreds of people treated with venetoclax‑based regimens showed that those whose leukemia cells carried higher amounts of activated STAT3 had shorter remissions and worse overall survival, regardless of age or treatment combination. In resistant cells and patient samples, a form of STAT3 that sits inside mitochondria—the cell’s energy factories—was especially increased and was linked to swollen, distorted internal folds inside mitochondria, signs of stressed and rewired energy metabolism.
Taking STAT3 apart instead of just blocking it
Because traditional STAT3 blockers have struggled in the clinic, the investigators turned to a newer strategy: targeted protein degradation. Rather than inhibiting STAT3’s activity, small “degrader” molecules physically tag STAT3 for disposal by the cell’s waste‑recycling machinery. In venetoclax‑resistant AML cell lines, two experimental degraders, KTX‑201 and KTX‑105, efficiently removed STAT3 and its activated forms without affecting related proteins. This triggered strong cell death, reduced the leukemia cells’ dependence on MCL1, and partially normalized mitochondrial structure and function. In cells from patients with AML and venetoclax‑resistant AML, the degraders wiped out STAT3 and encouraged immature cancer cells to mature into more normal red and white blood cells, while having little effect on healthy donor cells, suggesting a degree of selectivity for diseased stem and progenitor cells.
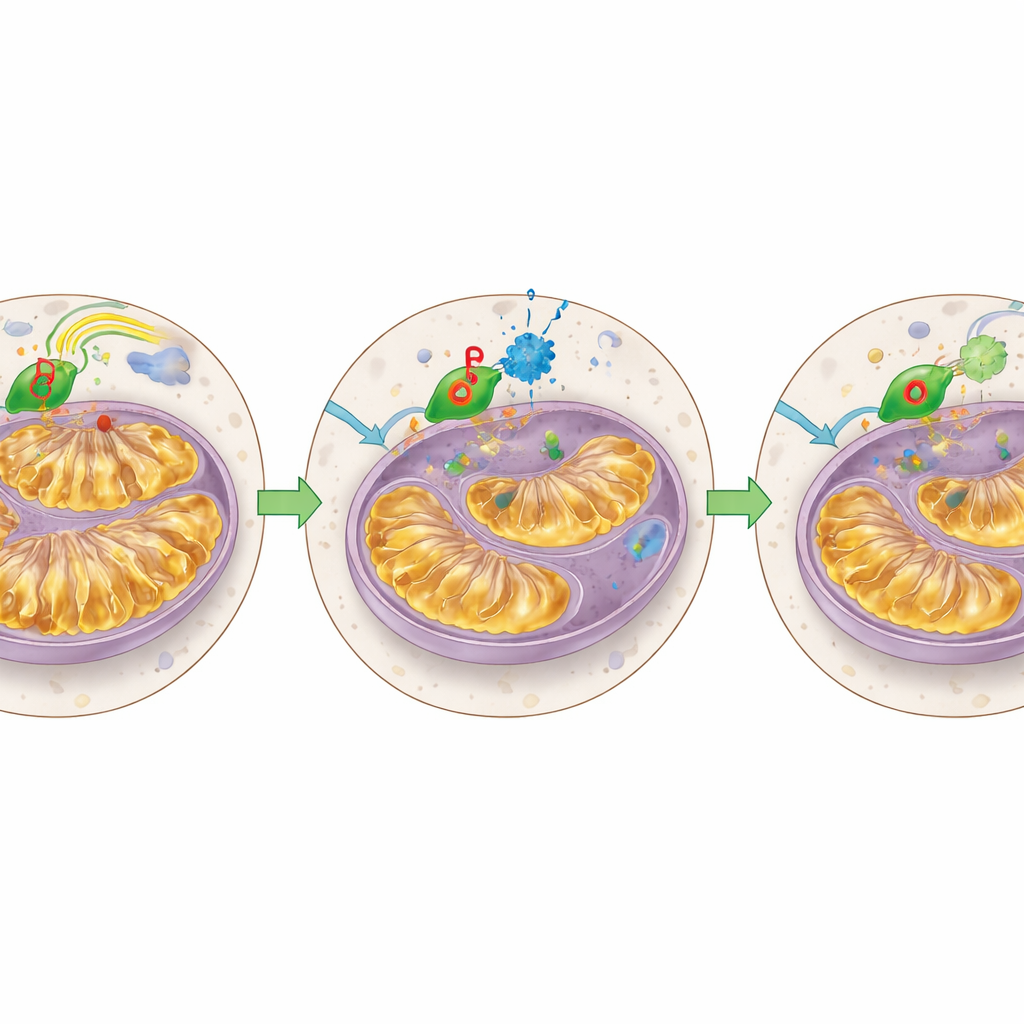
Fixing damaged mitochondria and testing in living models
Looking more closely at mitochondria, the team showed that STAT3 degraders lowered the amount of mitochondrial STAT3, reduced the abnormal expansion of internal folds, and shifted energy output back toward a more normal state. Venetoclax‑resistant cells had been running their mitochondria “hot,” with high energy production but little spare capacity to handle stress; degrader treatment cooled this down. The researchers then moved to mouse models that carried either venetoclax‑resistant leukemia cell lines or patient‑derived resistant AML cells. In these animals, a clinical‑stage degrader called KT‑333 markedly lowered STAT3 and MCL1 levels in leukemia cells, shrank the pool of immature leukemia‑initiating cells in the bone marrow, and significantly extended survival compared with untreated mice.
What this means for future AML treatment
Together, the findings suggest that venetoclax resistance in AML is closely tied to an overactive STAT3 pathway that reshapes both gene activity and mitochondrial behavior to keep leukemia cells alive. By selectively destroying STAT3, degrader drugs can cut off this escape route, restore more normal mitochondrial structure, push cancer cells toward death or maturation, and improve outcomes in preclinical models. For patients whose disease no longer responds to current therapies, STAT3 degraders such as KT‑333 may offer a new, mutation‑agnostic way to target the root survival machinery of resistant leukemia cells, and they are already entering early clinical testing.
Citation: Chakraborty, S., Morganti, C., Zaldana, K. et al. A STAT3 degrader demonstrates efficacy in venetoclax resistant acute myeloid leukemia. Leukemia 40, 717–729 (2026). https://doi.org/10.1038/s41375-026-02883-9
Keywords: acute myeloid leukemia, venetoclax resistance, STAT3 degrader, mitochondrial dysfunction, targeted protein degradation