Clear Sky Science · en
Fuel to fire: developmental niche-empowered ApoEVs unlock adult hierarchical tissue regenerative potential via mitochondrial complex I-driven developmental metabolic profile
Why rebuilding adult tissues is so hard
Many organs in our bodies are made of several tightly coordinated layers, like a layer cake. Once we are grown, these layered structures are surprisingly bad at repairing themselves after serious injury. This study asks a bold question: instead of trying to patch adult tissues as they are, could we briefly switch them back into a more youthful, developmental state so they can rebuild properly? Using the tissues that support teeth as a model, the researchers present a new way to “fuel” adult cells with developmental signals packaged in tiny natural particles.
How young teeth build a perfect support system
During childhood, the tissues that hold a tooth in place form in a highly choreographed way. Bone, ligament, and a thin mineral layer on the root all grow together and interlock, giving the tooth both stability and a bit of spring. The team compared this developing setting with the situation in adult repair. In young tissue, they found a calm, anti-inflammatory environment shaped by a special type of immune cell called an M2-like macrophage. These cells help create a supportive “niche” around stem cells, encouraging strong energy use, healthy mitochondria, and a preference for efficient oxygen-based metabolism. In adults, by contrast, the environment is more inflamed, stem cells are fewer and more sluggish, and the finely layered structure usually fails to grow back.
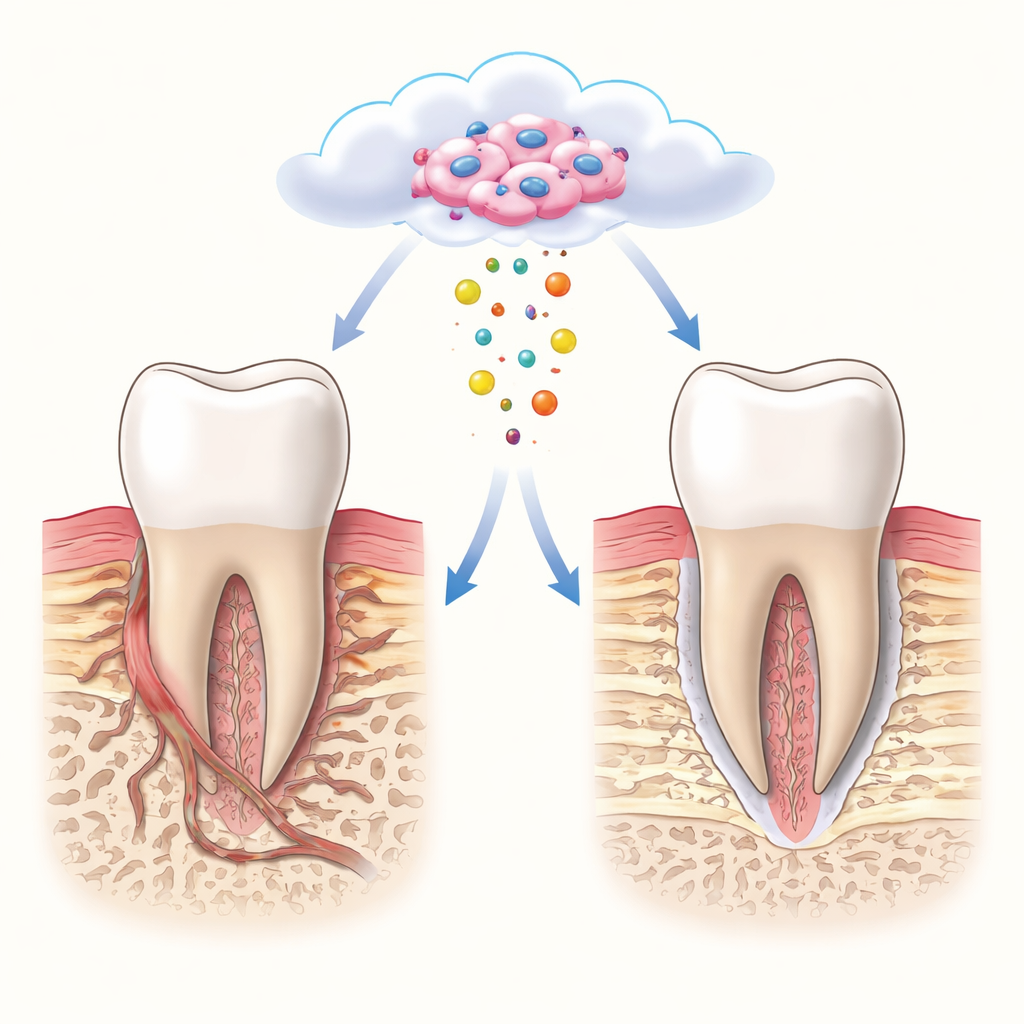
Borrowing from nature’s own clean-up system
When transplanted stem cells are placed into damaged tissue, most of them die within days. Rather than seeing this as a failure, recent work has suggested that the dying cells may still help by shedding tiny membrane-bound particles called apoptotic extracellular vesicles. These particles carry proteins and other molecules from their parent cells and can influence nearby cells without the risks of transplanting whole cells. The authors reasoned that if they first exposed stem cells from the tooth ligament to a developmental-like niche, then triggered their controlled death, the resulting vesicles would “remember” both the stem cells and their youthful environment. They called these tailored particles DevNiche-ApoEVs.
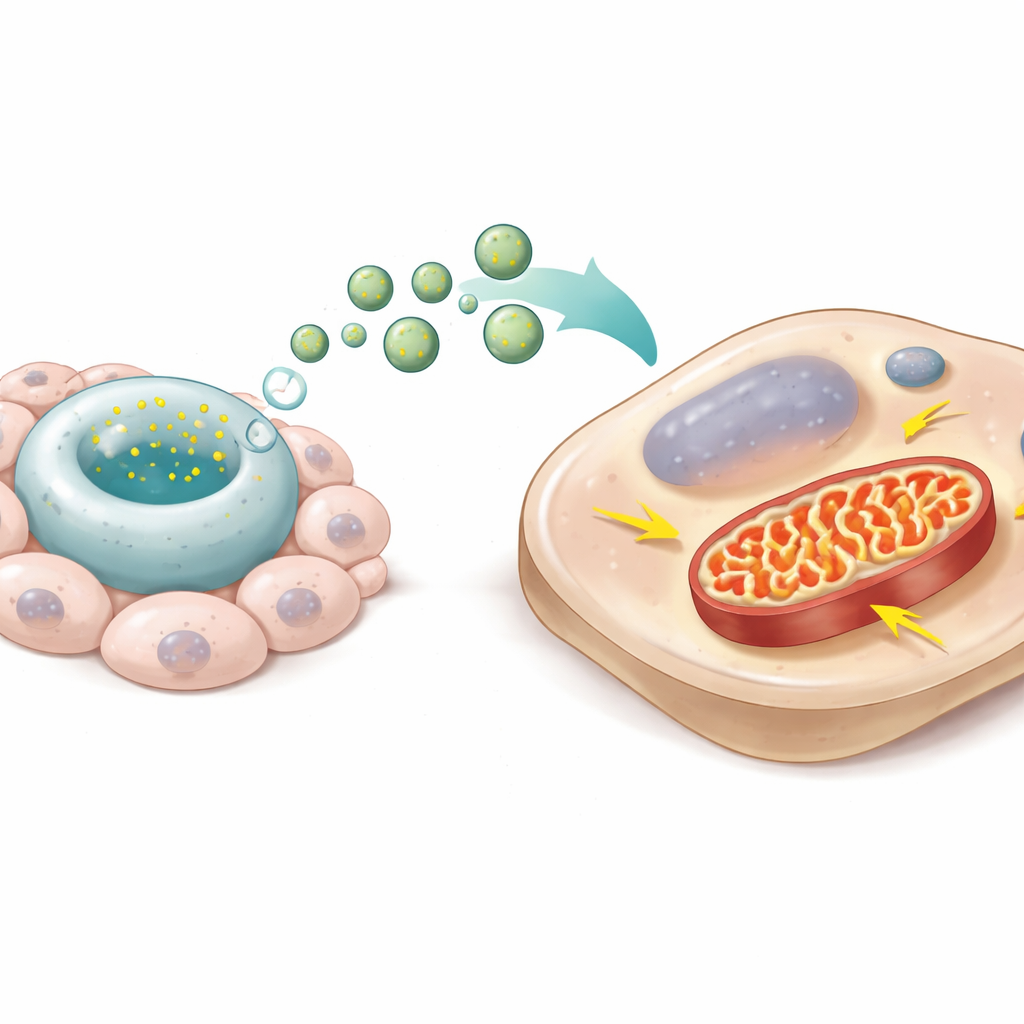
Recharging adult cells through mitochondria
The researchers examined the cargo of DevNiche-ApoEVs using advanced protein profiling. They found that these particles were rich in components of mitochondrial complex I, a key entry point for generating energy within mitochondria. When DevNiche-ApoEVs were added to adult ligament cells in the lab, the vesicles were taken up and their complex I proteins showed up inside the recipient cells. Mitochondria became more elongated with dense inner folds, produced more energy through oxygen use, and generated less damaging reactive oxygen species. Blocking complex I with a drug erased these benefits, showing that this mitochondrial machinery is central to the effect. In essence, the vesicles shifted adult cells from a low-power, stress-prone state toward a youthful, high-efficiency energy profile.
From petri dish to living jawbone
To see if this metabolic reboot could actually rebuild complex tissues, the team created precise periodontal defects in rats that removed bone, ligament, and root coating in one block. They filled the defects with a standard mineral scaffold, either alone or loaded with DevNiche-ApoEVs. Over the following weeks, animals that received DevNiche-ApoEVs grew dense, well-integrated bone, a new cementum-like layer on the root, and ligament fibers that ran in the correct direction and anchored into both root and bone. Microscopy showed signs of blood vessel growth, organized cell clustering, mineral deposition, and a calmer immune environment resembling the developmental stage. Defects treated without DevNiche-ApoEVs mostly filled with scar-like tissue and poorly oriented fibers.
What this could mean for future treatments
This study suggests that carefully engineered vesicles can act as “development in a bottle,” carrying both stem cell traits and environmental cues to wake up dormant repair programs in adult tissues. By delivering mitochondrial complex I and restoring a youthful energy pattern, DevNiche-ApoEVs helped adult cells rebuild the tooth-supporting complex in a way that closely mirrors natural formation during growth. While still at an early stage, this approach hints at future therapies where doctors might use similar vesicles to coax damaged organs toward self-directed regeneration rather than simple repair.
Citation: Zhang, Y., Xu, J., Shi, Y. et al. Fuel to fire: developmental niche-empowered ApoEVs unlock adult hierarchical tissue regenerative potential via mitochondrial complex I-driven developmental metabolic profile. Int J Oral Sci 18, 40 (2026). https://doi.org/10.1038/s41368-026-00440-9
Keywords: periodontal regeneration, stem cell niche, extracellular vesicles, mitochondrial metabolism, tissue engineering