Clear Sky Science · en
Chemical approaches to controlled carbon monoxide release: a focus on organic molecular donors
From Deadly Fume to Helpful Signal
Carbon monoxide is best known as a silent killer from faulty heaters and car exhaust, but inside our bodies the same gas is made in tiny amounts and quietly helps protect cells. This review article explores how chemists are learning to package carbon monoxide into smart "donor" molecules that can carry and release it like a medicine. By turning a dangerous gas into a controllable drug, researchers hope to treat problems such as inflammation, organ damage after stroke or heart attack, and stubborn infections—without risking poisoning.
Why the Body Uses a Poison on Purpose
Carbon monoxide is a simple gas made of one carbon and one oxygen atom, yet it has a split personality. At high levels it latches onto the blood’s oxygen carrier and suffocates tissues. At very low levels, however, our own cells produce it as a messenger, much like the better-known gas nitric oxide. In this gentle range it can calm inflammation, reduce cell death, and relax blood vessels. The difficulty is that breathing the gas or bubbling it into liquids gives almost no control over dose, place, or timing. What is needed instead is a way to smuggle carbon monoxide safely through the body and let it go only where and when it is useful.
Pills and Prodrugs: Bottling a Gas
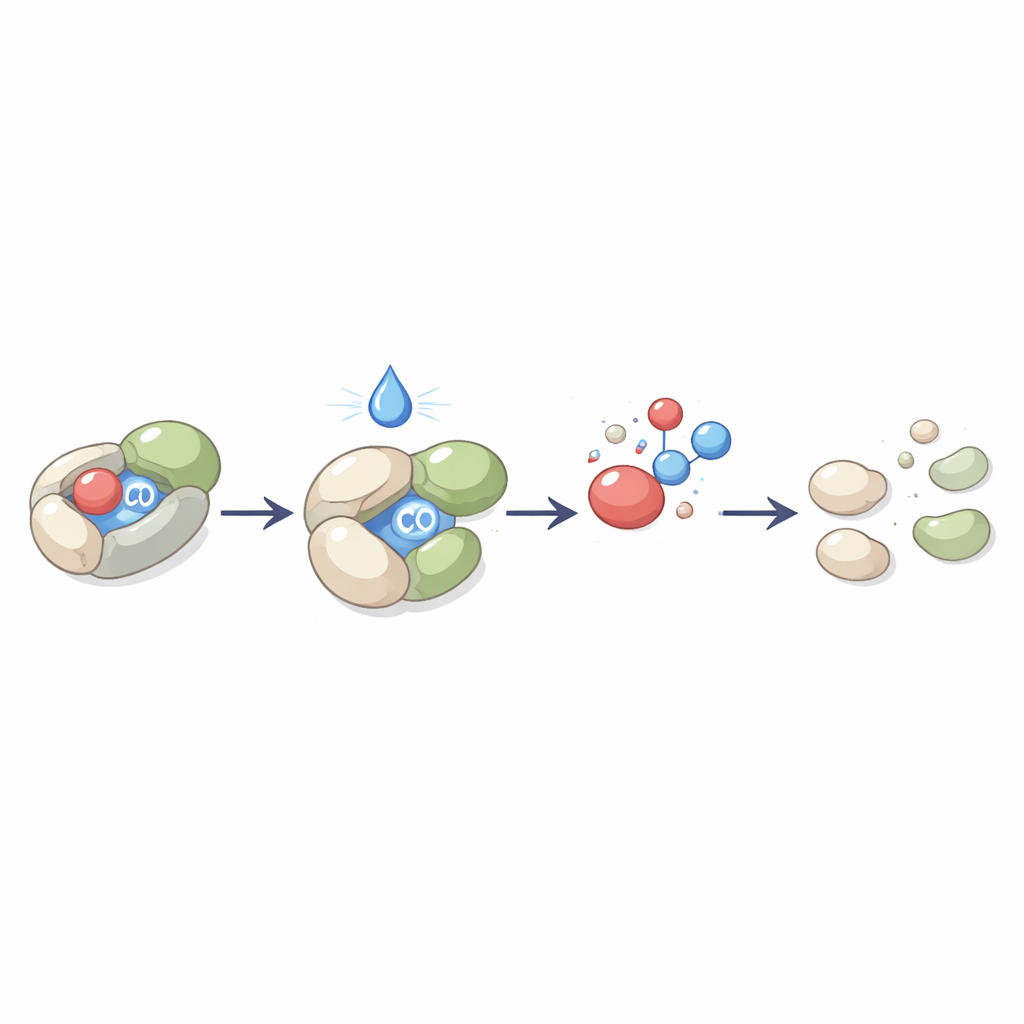
To solve this problem, chemists design carbon monoxide–releasing molecules, or CORMs. Early versions were based on metals that bind CO strongly, but worries over metal build-up and side effects have pushed the field toward metal-free, purely organic designs. These organic donors hold CO inside a larger carbon-based framework that falls apart under selected conditions, freeing the gas. The review groups them into three main shapes: open-chain molecules, single rings, and more rigid bicyclic or bridged structures. Across these families, a unifying idea is to keep CO in a “spring-loaded” state that is stable during storage yet ready to release when nudged by the right trigger such as water, light, heat, or chemical changes in diseased tissue.
Playing with Shape, Strain, and Light
Open-chain donors include modified acids, boron- and silicon-containing fragments, and special aldehydes. Clever tweaks make them more stable in the bottle but easy to activate in the body—for example, by enzymes or mild acidity—to give a steady, controllable puff of CO along with mostly harmless leftovers. Ring-shaped donors add another layer of control by building CO into strained three-, four-, five-, or six-membered loops. The strain in the ring acts like a bent spring: once triggered, the ring snaps open and ejects CO. Smaller rings tend to release quickly, while larger ones can be built to respond to signals such as reactive oxygen species found in inflamed or stressed tissues. Many designs are paired with fluorescent dyes, so that the same molecule both releases CO and lights up, allowing doctors and scientists to watch when and where the gas appears.
Smart Switches and Logic Gates in Medicine
The most advanced systems behave almost like tiny computers. Some bridged donors use "click" reactions to assemble a highly strained structure inside specific locations, such as particular cell compartments or material surfaces, and this structure then self-destructs to release CO. Others respond only when two conditions are met, such as the presence of a disease-related chemical plus light, forming an "AND" gate that sharply improves selectivity. Visible or near-infrared light, ultrasound, or built-in sensitivity to oxidants and enzymes can all serve as on–off switches. These sophisticated designs open the door to future therapies in which a pill, patch, or injectable material quietly carries CO through the body and releases it only inside diseased tissue, while simultaneously reporting its activity through a change in color or glow.
Where This Research Could Lead
In simple terms, the article concludes that turning carbon monoxide into a safe medicine depends on chemistry that can cage, aim, and precisely uncage this gas. Organic CO donors now offer a wide menu of options—fast or slow release, light or chemistry triggers, and built-in imaging—bringing the idea of "CO in a pill" much closer. The remaining hurdles are to balance stability with responsiveness, fully understand and tame any byproducts, and make these complex molecules practical to manufacture and deliver. If these challenges can be met, a gas once feared only as a household hazard may become a powerful, targeted tool against inflammation, organ injury, and certain infections.
Citation: Mu, W., Chen, X., Zhang, Z. et al. Chemical approaches to controlled carbon monoxide release: a focus on organic molecular donors. npj Soft Matter 2, 9 (2026). https://doi.org/10.1038/s44431-026-00021-0
Keywords: carbon monoxide therapy, CO-releasing molecules, organic prodrugs, stimuli-responsive drug delivery, photoactivated medicine