Clear Sky Science · en
Molecular imprinting in double-network-hydrogel-based organic electrochemical transistor channel for glucose sensing
Smarter Sensors for Everyday Sugar Monitoring
Glucose, the simple sugar that fuels our bodies, is also a vital health signal. People with diabetes and prediabetes increasingly rely on small sensors to track their glucose levels without constant finger‑pricks. This study explores a new kind of soft, electrically active material that can be built directly into tiny electronic switches, aiming to make future glucose monitors more sensitive, more selective for glucose over other sugars, and easier to wear on the skin.
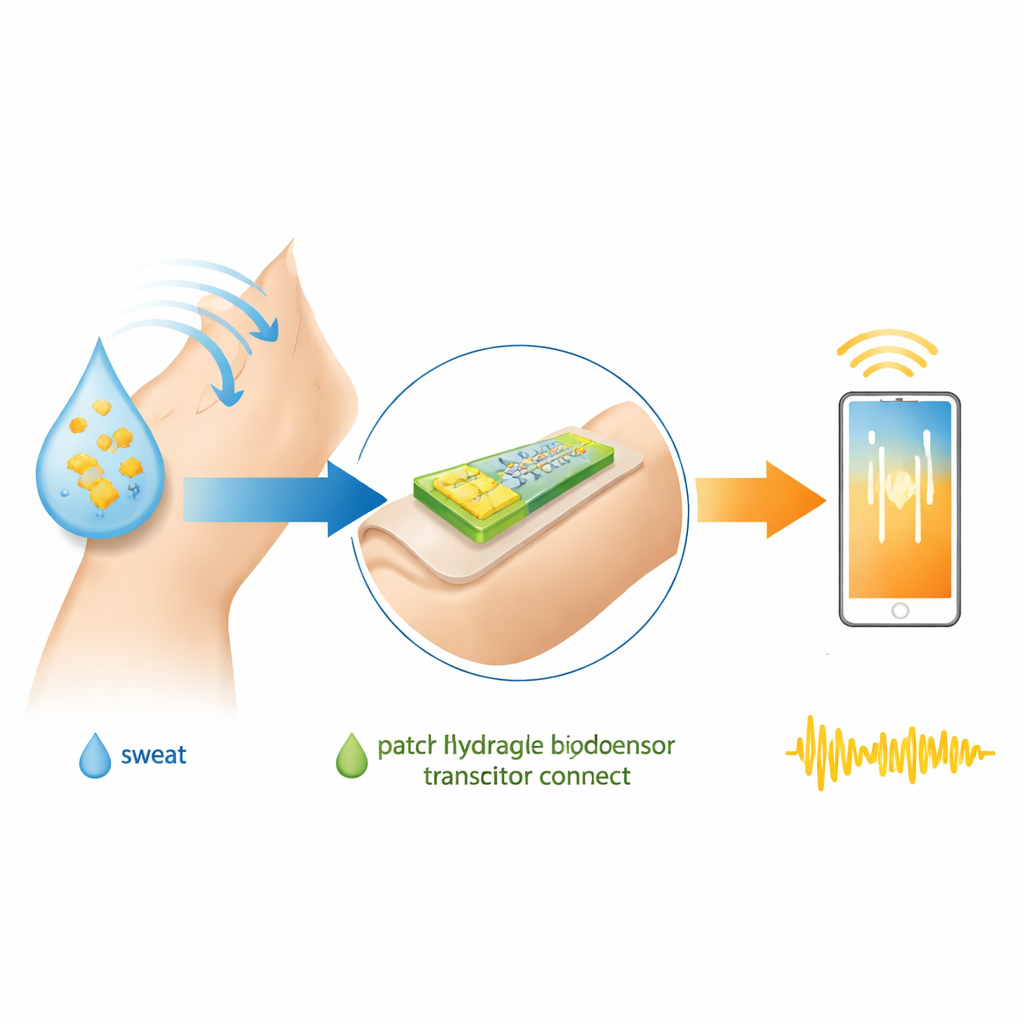
A Soft Electric Switch That Likes Water
The researchers focus on organic electrochemical transistors, a class of electronic components that work well in salty, watery environments like blood, sweat, or saliva. Unlike standard silicon chips insulated from liquids, these transistors use a soft, conductive polymer channel that allows ions from a surrounding solution to move in and out. That motion changes how easily electrical current flows, turning biological activity at the surface into a readable electronic signal. A popular material for this channel is PEDOT:PSS, a flexible, biocompatible polymer that can be formed into a hydrogel—a water‑rich, jellylike solid.
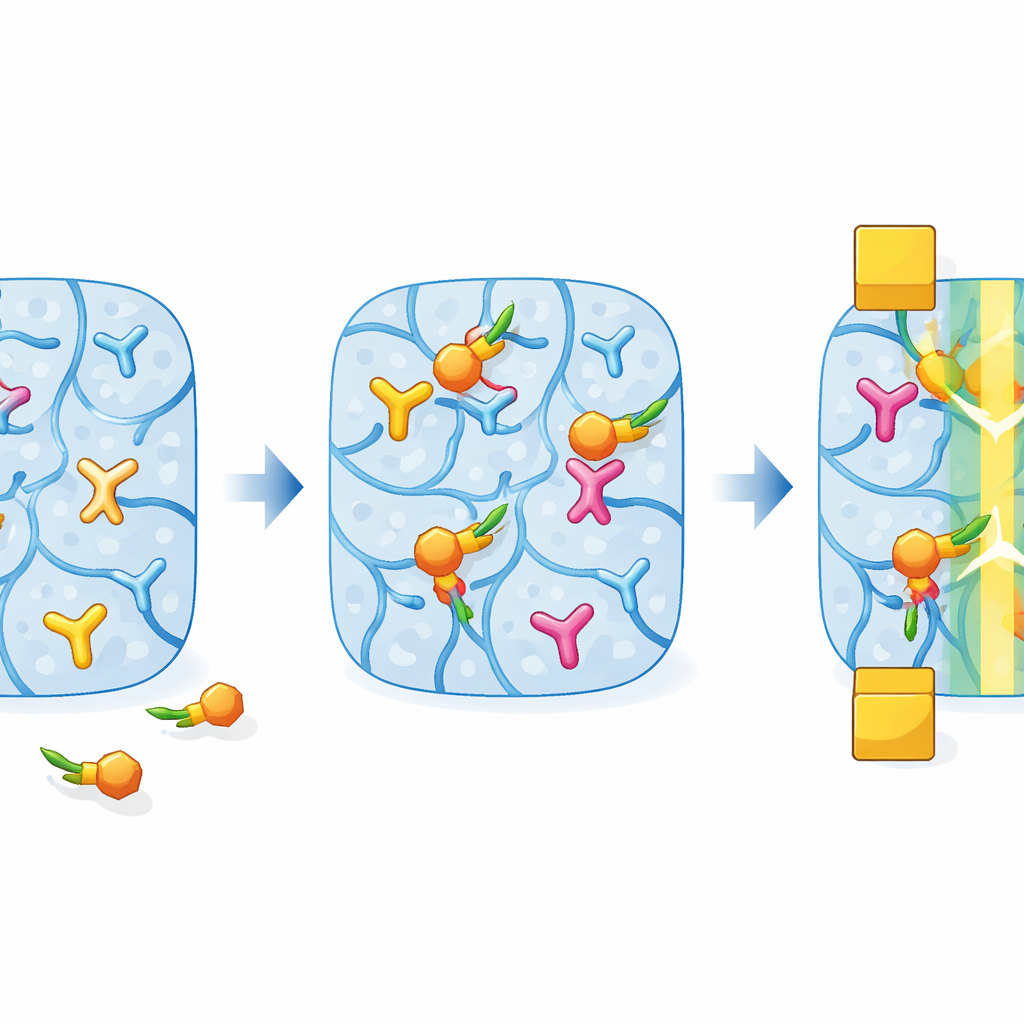
Teaching a Gel to Recognize Glucose
To make the soft channel respond specifically to glucose, the team borrows a concept called molecular imprinting. During fabrication, they mix into the gel a “receptor” building block that likes to bind molecules containing pairs of oxygen‑bearing groups, such as sugars. At the same time, they add real glucose as a temporary guest. As the second polymer network forms and crosslinks inside the PEDOT:PSS gel, it wraps around these glucose guests, creating tiny cavities that match glucose in size and bonding pattern. Afterward, the glucose is washed out with an acidic solution, leaving behind a sponge filled with glucose‑shaped pockets that are primed to grab glucose when it appears again.
From Binding Events to Stronger Electrical Signals
When glucose‑containing liquid contacts this double‑network hydrogel channel, glucose slips into the imprinted pockets and reacts with the receptor groups, changing their electrical charge. These local chemical changes alter how easily the PEDOT:PSS backbone can exchange electrons and ions, which in turn changes the current flowing through the transistor. The authors first ensure the gel is formed uniformly inside narrow tubes so it adheres well to gold electrodes and behaves consistently. They then compare channels made with and without the glucose template, measuring how the transistor’s current response grows as glucose concentration increases over a wide range. By fitting the data to a model that describes how molecules occupy binding sites, they estimate an effective “binding strength” between glucose and the gel.
Sharper Vision for Glucose Among Other Sugars
The molecularly imprinted channels show a clear advantage. Their apparent binding constant for glucose is about ten times higher than that of non‑imprinted gels, meaning glucose is captured more strongly and efficiently. As a result, the transistor’s response to small changes in glucose becomes steeper at low concentrations, and the minimum concentration it can reliably pick up drops into the sub‑micromolar range—well below typical glucose levels found in human sweat. Importantly, the gel also becomes more selective: in the imprinted material, glucose is favored over a similar sugar, fructose, by roughly two orders of magnitude compared with the same chemistry in simple solution. The transistor’s own amplification properties help magnify these chemical differences into robust electrical signals without needing separate amplifying circuits.
Why This Matters for Future Wearable Health Devices
To a lay reader, the central message is that the authors have taken a squishy, water‑loving electronic material and taught it to “remember” glucose, then wired that memory directly into a tiny switch. This combination of molecular imprinting inside a conductive hydrogel, used as the active channel of an organic electrochemical transistor, yields stronger binding to glucose, better discrimination against other sugars, and a lower detection limit in a range suitable for sweat‑based monitoring. While challenges remain—such as dealing with interfering substances in real body fluids and ensuring long‑term stability—the work points toward thin, flexible patches that could one day continuously track sugar levels by reading subtle electrical changes in a smart gel pressed gently against the skin.
Citation: Kawamura, M., Tseng, A.C. & Sakata, T. Molecular imprinting in double-network-hydrogel-based organic electrochemical transistor channel for glucose sensing. npj Biosensing 3, 24 (2026). https://doi.org/10.1038/s44328-026-00093-y
Keywords: glucose sensor, wearable biosensor, organic electrochemical transistor, molecular imprinting, hydrogel electronics