Clear Sky Science · en
Efficient extraction of tetravalent actinide from nitric acid feeds using tri-N-octyl methyl ammonium N-dodecyl sulphate functionalized task-specific ionic liquid
Cleaning Up Nuclear Power
Nuclear energy can generate huge amounts of electricity without releasing greenhouse gases, but it leaves behind highly radioactive leftovers. Some of the metals in this waste, such as plutonium and uranium, are both dangerous and valuable. This study explores a new, more sustainable liquid that can pull specific metals out of harsh radioactive solutions, helping to recycle useful materials while reducing the long‑term burden of nuclear waste.
A Custom-Made Cleaning Liquid
The researchers designed a special “task-specific” ionic liquid—a salt that is liquid at room temperature—built from large organic ions. Unlike common solvents made from volatile organic chemicals, ionic liquids hardly evaporate and can be tuned chemically for particular jobs. Here, the team attached a detergent-like group called dodecyl sulfate to a bulky ammonium ion, creating a thick, straw‑yellow fluid. This detergent tail is similar to the cleaning agents used in shampoos and soaps, but in this case it is anchored permanently to the ionic liquid, turning the whole molecule into a powerful extractor for metal ions dissolved in acidic water.
How It Grabs Nuclear Metals
Spent nuclear fuel is typically dissolved in nitric acid, forming a cocktail of different metal ions and molecular forms. The new ionic liquid was tested on three key actinide elements: plutonium in a four‑plus state, uranium in a six‑plus state, and americium in a three‑plus state. By carefully adjusting the acid strength, the authors showed that plutonium changes form—from simple charged ions to more complex nitrate clusters—and these changes strongly affect how it moves into the ionic liquid. Around medium to high acid levels, the liquid forms stable complexes with plutonium-containing units, pulling them into the ionic phase, while leaving most americium behind and taking up only moderate amounts of uranium.
Picking Favorites: Plutonium Over Uranium and Americium
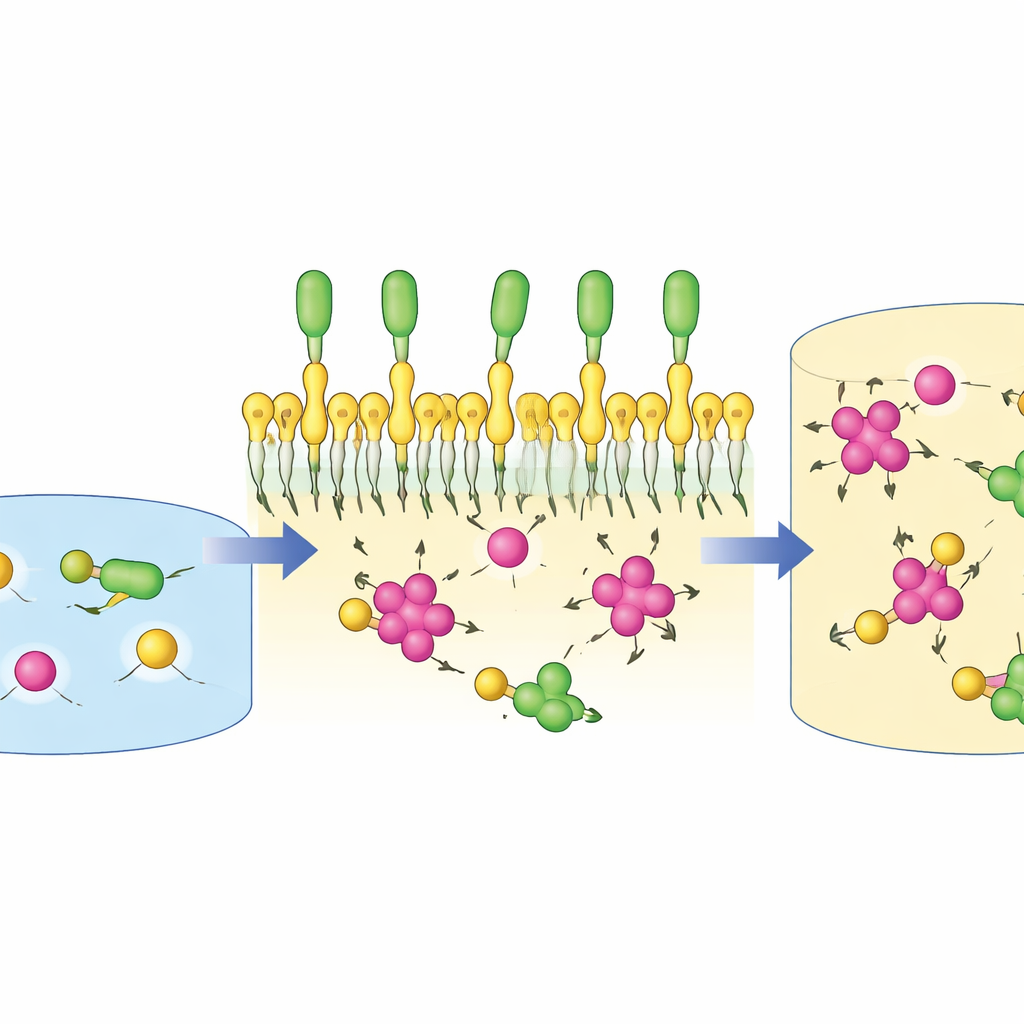
A central result is just how selective the liquid is. Under optimized conditions, plutonium was extracted thousands of times more strongly than uranium, and up to one hundred thousand times more strongly than americium. This far exceeds many conventional solvent systems used in nuclear reprocessing today. The authors traced this behavior to the way the ionic liquid’s charged head groups and sulfate-based tails wrap around different plutonium nitrate complexes, creating very favorable binding arrangements. Uranium, which forms different shapes in solution, fits less snugly, and americium hardly interacts at all, so they remain mostly in the watery phase. This natural “preference” allows plutonium to be separated from other actinides in a relatively simple liquid–liquid process.
Slow but Strong, and Reusable
The new liquid is thick, which slows down how fast metals can diffuse through it, so about an hour is needed for each extraction step—longer than for lighter, conventional solvents. However, the binding itself is strongly heat‑releasing, meaning that once plutonium or uranium is captured, the complexes are quite stable. The team also explored how to get the metals back out of the ionic liquid, an important step for recycling. Simple acid changes were not enough, but mild solutions of oxalic acid or sodium carbonate, applied in several contacts, could strip nearly all of the loaded plutonium and uranium. The ionic liquid could then be reused for at least five extraction–stripping cycles with only minor loss of performance.
Standing Up to Radiation—and Its Limits
Because nuclear waste streams are intensely radioactive, the solvent itself must withstand bombardment by high‑energy particles. The authors exposed the ionic liquid to very large doses of gamma rays and found that, although its extraction power gradually declined—by about one‑third to nearly one‑half for uranium at the highest dose—the main molecular framework remained intact according to infrared and NMR measurements. Some breakdown into smaller amines, hydrocarbons, and sulfate fragments is expected, but the material still functioned reasonably well even after harsh treatment.
Why This Matters for Nuclear Waste
To a non‑specialist, the key message is that the researchers have created a highly selective, relatively robust “designer solvent” that can pull plutonium out of acidic nuclear waste more efficiently than many current technologies, while using a liquid that does not evaporate or burn easily. By favoring plutonium over uranium and americium, and by working under conditions similar to real high‑level waste, this ionic liquid could help future recycling schemes recover strategic materials and reduce the long‑term radioactivity that must be stored. Challenges remain—especially speeding up the process and improving resistance to radiation—but the work points toward cleaner, more sustainable chemistry for managing nuclear energy’s toughest leftovers.
Citation: Chowta, S.D., Sengupta, A. & Mohapatra, P.K. Efficient extraction of tetravalent actinide from nitric acid feeds using tri-N-octyl methyl ammonium N-dodecyl sulphate functionalized task-specific ionic liquid. npj Mater. Sustain. 4, 14 (2026). https://doi.org/10.1038/s44296-025-00094-4
Keywords: ionic liquids, nuclear waste, plutonium separation, solvent extraction, actinide chemistry