Clear Sky Science · en
AI-based autism identification from hyperspectral imaging detection of oxidative stress in pediatric red blood cells
Why a Drop of Blood May Reveal Hidden Stress
Doctors have long suspected that many diseases quietly damage our cells years before symptoms appear, but they have lacked simple tools to see this early harm. This study explores a new way to read that hidden damage from a single drop of blood, using advanced imaging and artificial intelligence. The work focuses on children with autism, but the approach could eventually help monitor a wide range of conditions linked to cellular “wear and tear.” 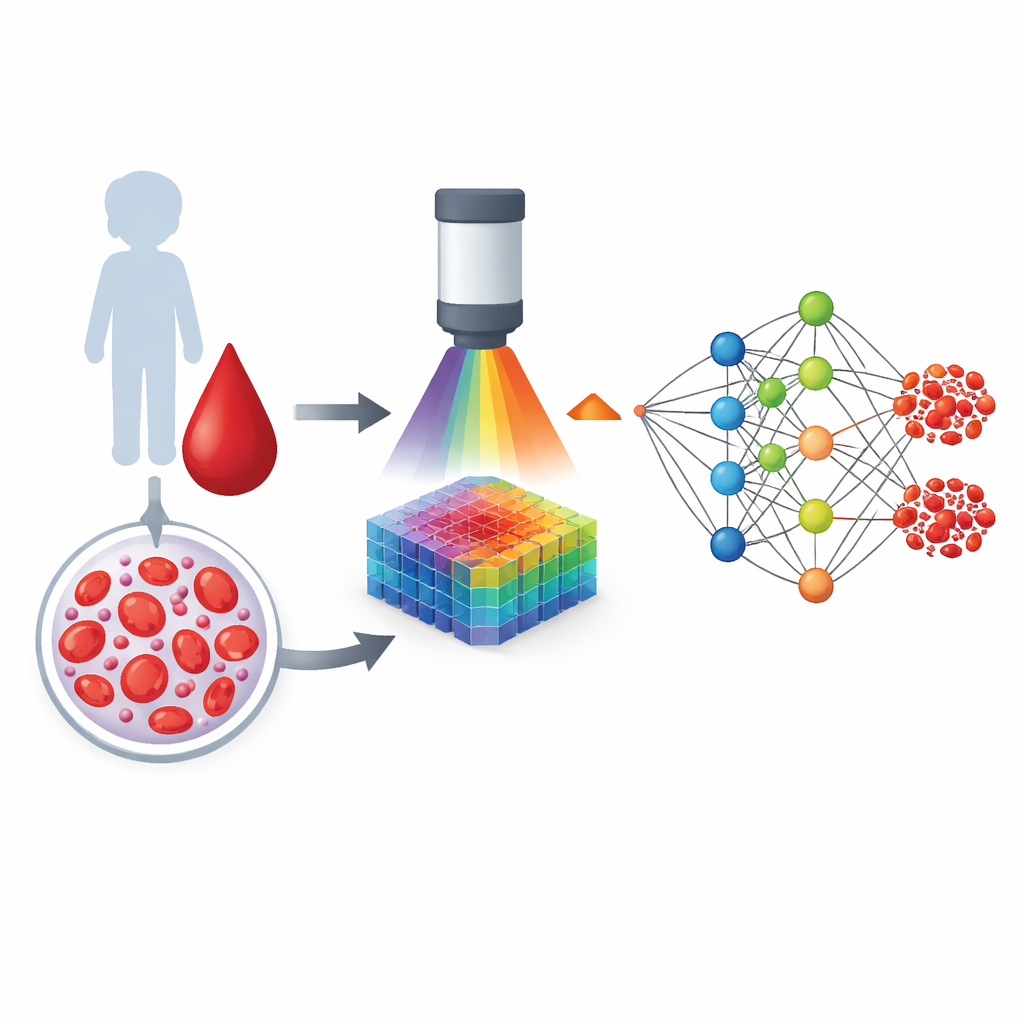
A New Way to Look at Blood Cells
Red blood cells do more than carry oxygen; their delicate outer membrane also records the chemical stresses a person has faced. When the body is exposed to oxidative stress—an imbalance between harmful reactive molecules and the body’s defenses—certain fragile fats in this membrane are among the first targets. Instead of looking for one specific chemical, the researchers used hyperspectral imaging, a technique that captures how each tiny patch of a cell scatters light across many colors, far beyond what our eyes can see. Each pixel becomes a kind of color fingerprint that reflects both the molecules present and how they are arranged in the membrane.
Building a Map of Healthy and Stressed Cells
To understand what oxidative stress does to red blood cells, the team first created a controlled laboratory model. They collected small blood samples from healthy adults and gently exposed some of the samples to hydrogen peroxide, a well-known oxidizing agent. Using hyperspectral dark-field microscopy, they recorded detailed light-scattering patterns from many individual cells and then grouped these patterns into eight recurring “spectral signatures.” In parallel, they chemically analyzed the membrane fats and confirmed that oxidation reduced sensitive polyunsaturated fats and increased sturdier saturated fats. Specific spectral signatures shifted in a consistent way when this chemical remodeling occurred, showing that the imaging method could “see” structural membrane changes without touching the cells or adding any dyes.
Applying the Method to Children with Autism
With this reference in hand, the researchers turned to blood samples from 27 boys with autism spectrum disorder and 31 neurotypical boys of similar age and body size. They imaged red blood cells from each child using the same hyperspectral protocol. Several of the spectral signatures in children with autism differed from those in neurotypical children, and the direction of these changes closely mirrored what had been seen in the oxidized adult samples. One particular signature, associated with membranes that had lost vulnerable fats and gained more rigid ones, stood out as especially altered. This pointed to a common pattern: the red blood cells of many children with autism appeared to carry the optical hallmarks of heightened oxidative stress. 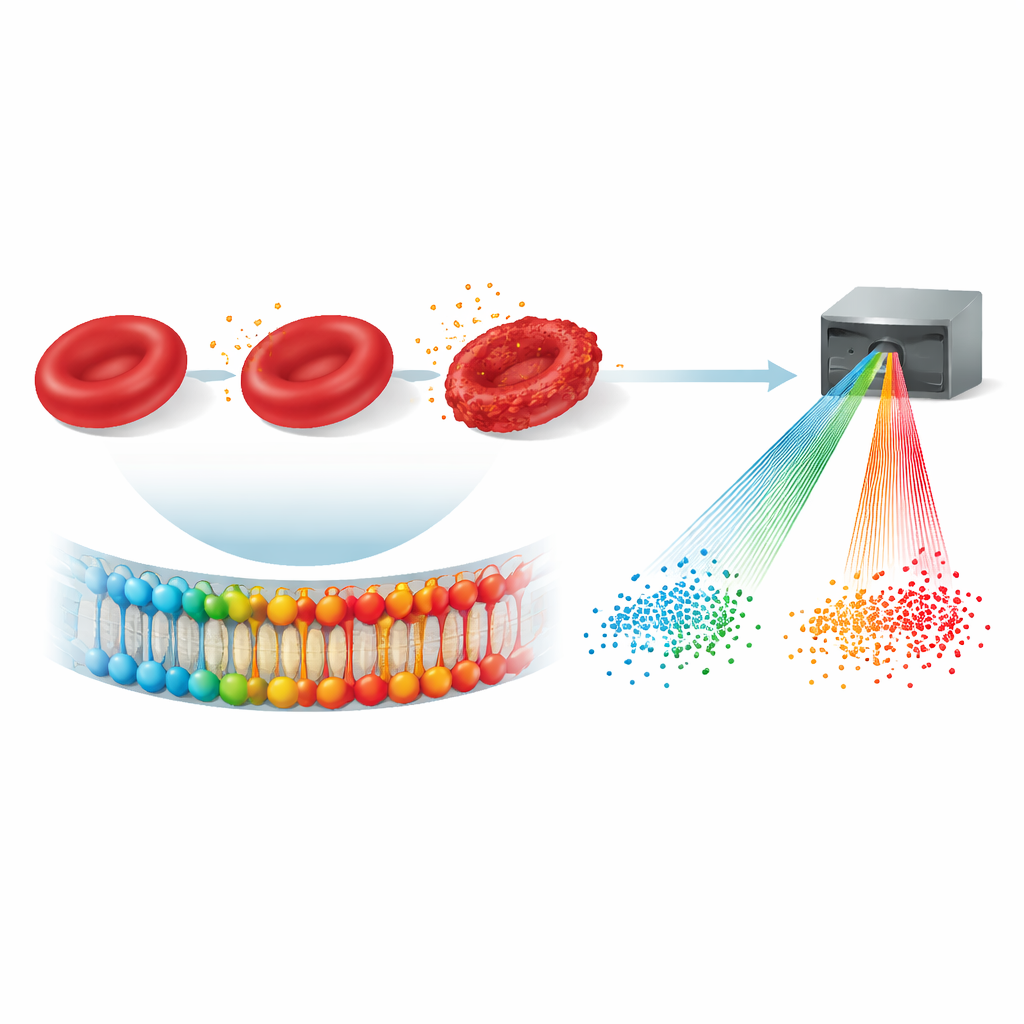
Letting Artificial Intelligence Read the Patterns
Because the hyperspectral data are complex, the team used artificial neural networks—computer models inspired by brain circuits—to learn how combinations of spectral signatures relate to autism versus typical development. After carefully splitting the data into training and testing sets and trimming away unhelpful inputs, the system was able to sort the children’s samples into “autism” and “neurotypical” groups with about 93 percent overall accuracy, with both high sensitivity and specificity. A second AI tool, designed to reveal hidden relationships rather than make predictions, highlighted one key spectral signature as most strongly linked to the autism group, reinforcing the idea that a particular pattern of membrane change is central.
What This Could Mean for Future Care
The authors emphasize that this is not yet a diagnostic test for autism, nor should it be used to label individual children. Instead, the study shows that red blood cell membranes carry a readable record of oxidative stress, and that hyperspectral imaging combined with AI can extract this information quickly from only two microliters of blood, without invasive procedures or added chemicals. In the long run, similar approaches could help doctors track how much hidden stress a person’s cells are under, guide personalized choices about nutrition and treatment, and monitor whether therapies are restoring healthier cell membranes before serious disease takes hold.
Citation: Vartian, R., Sansone, A., Batani, G. et al. AI-based autism identification from hyperspectral imaging detection of oxidative stress in pediatric red blood cells. Commun Med 6, 266 (2026). https://doi.org/10.1038/s43856-026-01581-y
Keywords: oxidative stress, red blood cells, hyperspectral imaging, autism, artificial intelligence