Clear Sky Science · en
Multicellular origins of murine ovarian inflammaging
Why the Aging Ovary Matters
People are waiting longer than ever to have children, but our ovaries have not kept pace with this social shift. As women age, fertility drops and the risks of miscarriage and complications rise. Scientists know that the ovary becomes more fibrotic, more inflamed, and less able to support healthy eggs, but the fine‑scale reasons have been hard to pin down. This study uses powerful new genetic mapping tools to build a cell‑by‑cell atlas of the aging mouse ovary, revealing how immune cells move in, how communication between cells changes, and how this slow, smoldering inflammation—called “inflammaging”—emerges.
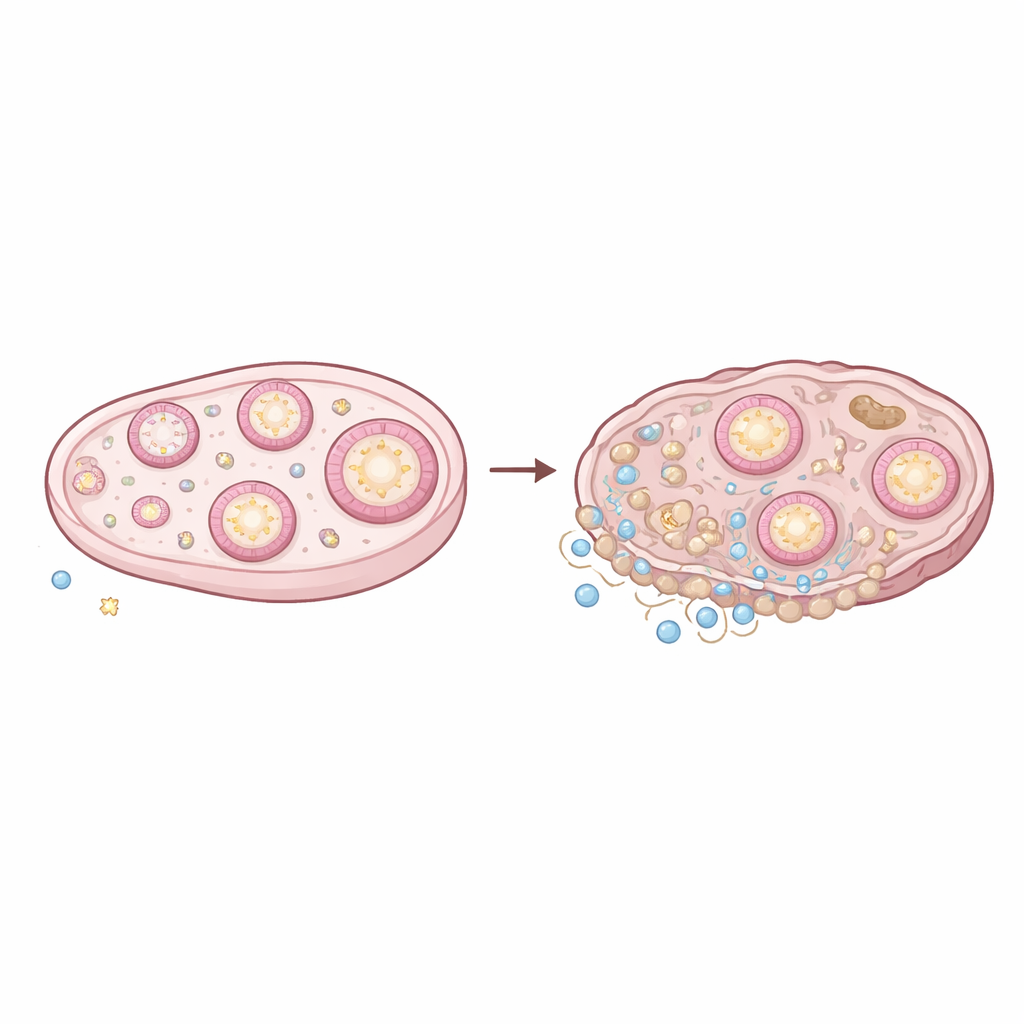
A Close Look at the Aging Ovary
The ovary is a constantly remodeling organ: follicles grow and die, ovulation creates a monthly “wound,” and temporary structures called corpora lutea appear and then regress. To see how this dynamic tissue changes with age, the researchers analyzed thousands of individual cells from young (3‑month) and old (14‑month) mouse ovaries using single‑cell RNA sequencing, which reads out which genes are switched on in each cell. They then overlaid this information with spatial transcriptomics, a technique that maps gene activity back onto the physical tissue. Together, these methods produced a detailed landscape of cell types, their locations, and how they talk to one another in young versus aged ovaries.
Immune Cells Move In and Turn Up the Heat
One of the clearest age‑related changes was a surge in immune cells. While the total number of macrophages (tissue‑resident scavenger cells) did not skyrocket, their makeup shifted toward more inflammatory and fibrosis‑promoting types. The team identified aging‑specific scar‑associated macrophages and a separate group marked by an activating receptor (Cd300e), both known in other organs to drive chronic inflammation and scarring. Lymphocytes—T cells and B cells—expanded even more dramatically. Subsets of T cells associated with chronic inflammatory diseases, as well as innate lymphoid cells that can trigger fibrosis when dysregulated, became far more common. These immune cells sent stronger signals through well‑known inflammatory pathways, including tumor necrosis factor (TNF) and chemokine networks that attract and activate more immune cells, setting up a self‑reinforcing loop.
Shifts in the Ovary’s Supporting Cells
Eggs do not age in isolation; they rely on surrounding granulosa cells, which form the main body of the follicle. By separating granulosa cells into distinct subtypes corresponding to different follicle stages, and then mapping where they sit in the tissue, the researchers showed that aging changes both their composition and behavior. Old ovaries contained relatively fewer early‑stage granulosa cells and more late‑stage and degenerating (atretic) granulosa cells, mirroring the loss of the follicle pool. Importantly, gene activity linked to healthy follicle growth—such as signals involved in hormone production, lipid handling, and early development—declined with age. At the same time, outer granulosa cells near the boundary with the ovarian stroma ramped up messages that attract and activate immune cells, especially macrophages.

A Web of Crosstalk That Favors Fibrosis
By tracing which cell types send and receive specific molecular signals, the authors uncovered a dense web of age‑dependent communication. Stromal fibroblasts and granulosa cells sent more signals that recruit macrophages and guide them toward pro‑fibrotic, scar‑forming states. In turn, aging‑associated macrophages increased output of factors that stimulate fibroblasts to lay down collagen and other matrix proteins, making the ovary stiffer and more fibrotic. Lymphocytes and macrophages also exchanged signals that promote their survival, movement, and inflammatory activity, suggesting a positive feedback loop. B cells and T cells, for example, strengthened pathways that enhance antigen presentation and immune activation. Altogether, this network analysis shows how a once finely tuned environment for supporting eggs gradually becomes dominated by chronic inflammation and scar tissue.
What This Means for Reproductive Health
In everyday terms, this work suggests that the aging ovary slowly shifts from being an egg‑nurturing organ to an immune‑centered, inflamed tissue. Granulosa cells that once focused on feeding and coordinating the egg begin to act more like beacons for immune cells. Specialized macrophages and T cells, in turn, keep inflammation and fibrosis going, making it harder for follicles to develop and ovulate normally and potentially lowering egg quality. Although the study was done in mice, many of the same cell types and pathways are found in humans, hinting that targeting these inflammatory and fibrotic circuits—rather than the eggs alone—could open new strategies to extend reproductive health and ease the broader health impacts of menopause.
Citation: Galligos, A., Varberg, J.M., Yueh, WT. et al. Multicellular origins of murine ovarian inflammaging. Commun Biol 9, 593 (2026). https://doi.org/10.1038/s42003-026-09826-1
Keywords: ovarian aging, inflammaging, immune cells, granulosa cells, fertility decline