Clear Sky Science · en
Early cAMP signaling orchestrates single-cell synchronicity throughout Dictyostelium development
How Cells Keep Time Together
Many living things grow in a remarkably coordinated way: segments of a fish spine appear one after another, the units of a fly’s eye line up like tiles, and even single-celled organisms can move and change shape in step. This article explores how a soil-dwelling amoeba, Dictyostelium discoideum, keeps thousands of its cells marching through development in sync. Understanding this natural "cell choreography" helps explain how tissues form correctly—and what might go wrong when timing falls apart.
A Social Amoeba with a Talent for Teamwork
Dictyostelium lives most of its life as individual amoebae that crawl around eating bacteria. When food runs out, these loners suddenly become highly social. They gather into visible clumps, build finger-like structures called slugs, and finally form slender fruiting bodies that lift spores into the air. All of this unfolds in about a day, and different groups of cells spread across a dish often look almost identical at each stage. The question the researchers asked is: how do so many separate cells manage to change their internal state and outer form in such a synchronized way?
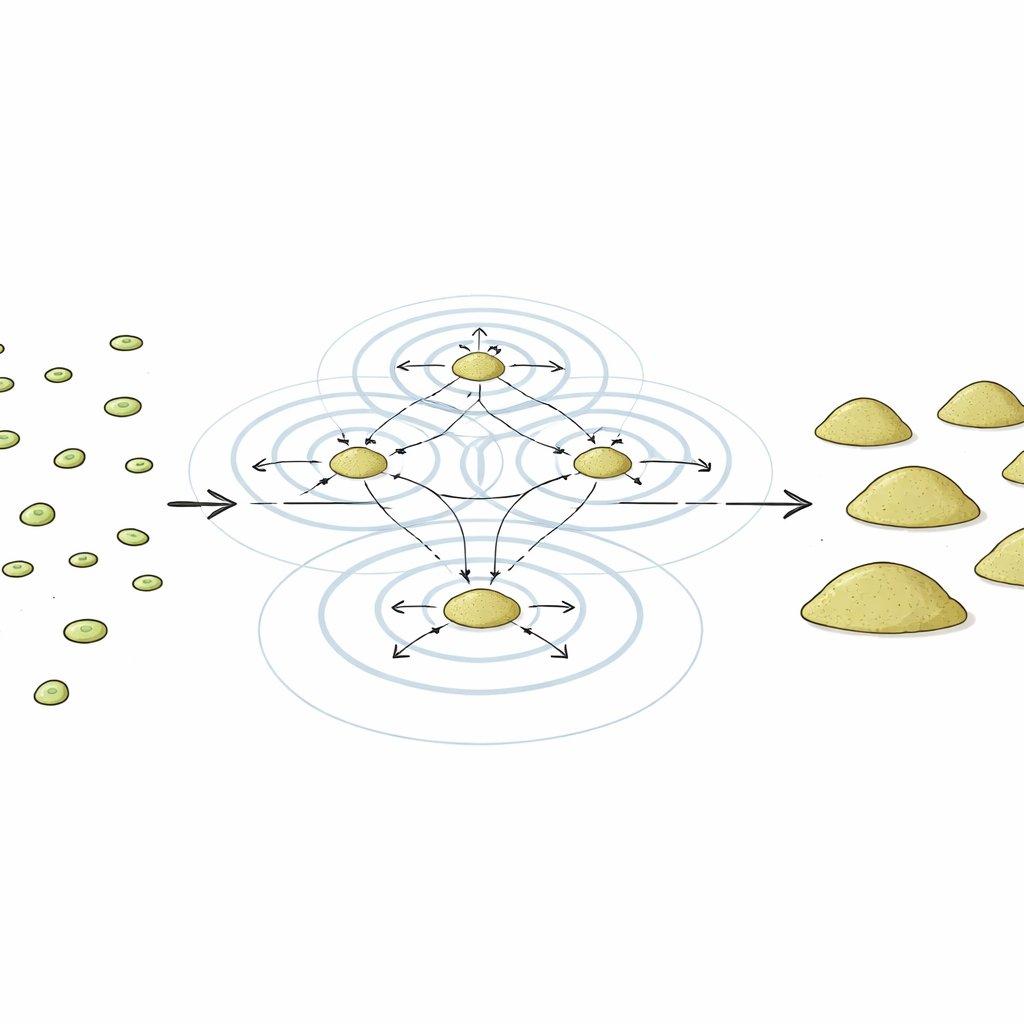
A Chemical Pulse that Sets the Pace
Earlier work showed that starving amoebae send each other rhythmic bursts of a small signaling molecule called cAMP. Every few minutes, waves of cAMP ripple through the cell population, guiding cells to move and start forming multicellular clumps. The authors proposed that these early cAMP pulses do more than just tell cells where to go—they may also act like a metronome that keeps the internal programs of thousands of cells in time, so that they activate and silence genes together as development proceeds.
Reading Cell States One Cell at a Time
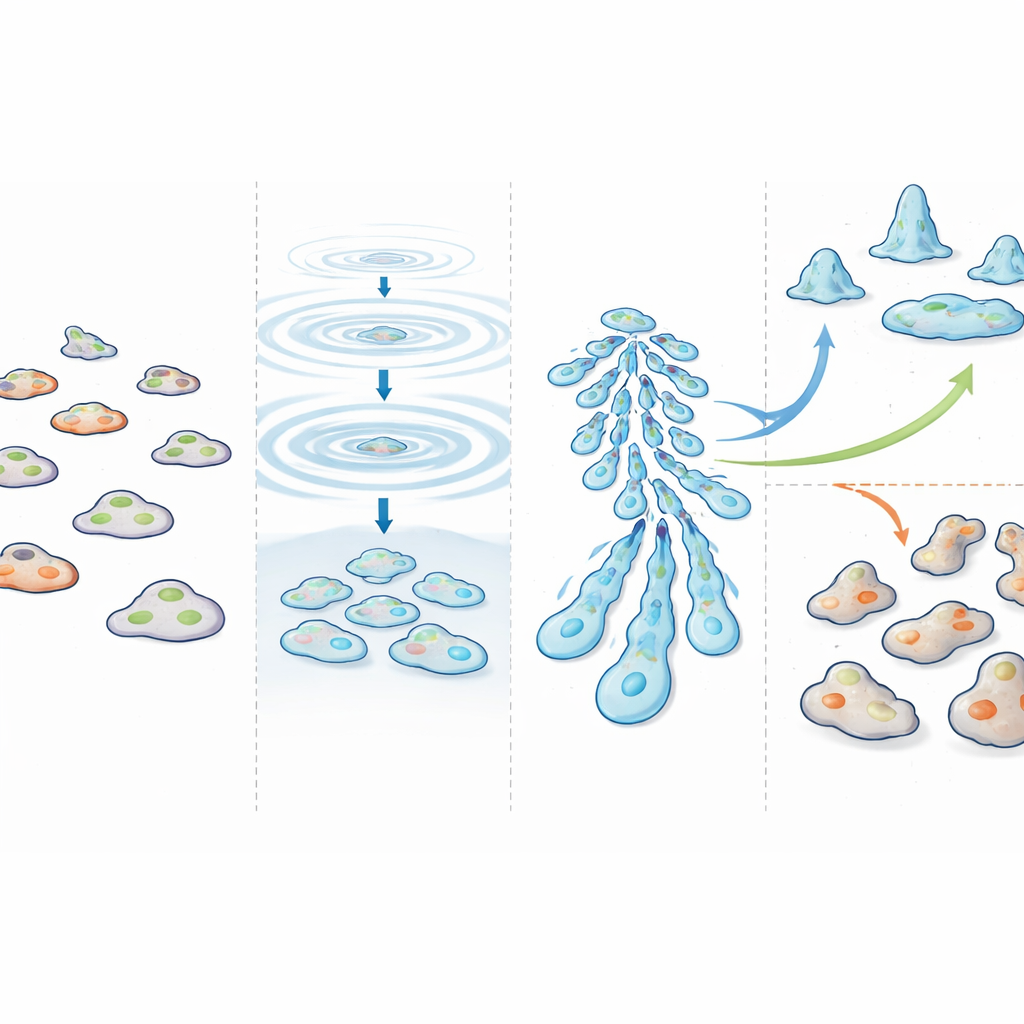
To test this idea, the team turned to single-cell RNA sequencing, a technology that reads which genes are active in thousands of individual cells at once. They grew three versions of Dictyostelium: a normal strain; a mutant unable to make cAMP pulses; and a double mutant that cannot pulse but is forced to develop anyway by boosting a key regulatory enzyme. At several time points over 20 hours, they captured cells and measured their RNA profiles. By comparing how similar or different these profiles were across cells, they could calculate a numerical score for "synchronicity"—how alike the cells’ internal states were at each moment of development.
When the Metronome Works—and When It Doesn’t
In normal cells, synchronicity initially dipped right after starvation, reflecting the shock of changing conditions. Then, between four and eight hours, as cAMP pulses appeared and cells began to gather, synchronicity rose sharply and stayed high through later stages. Even as cells split into two main fates—spore-forming and stalk-forming types—cells within each group remained tightly coordinated. In striking contrast, cells that could not make cAMP pulses never formed proper multicellular structures and showed only weak, unstable synchronicity over time. The double mutant, which can develop without pulses, did reach advanced shapes, but its cells drifted out of sync: at any given time point, they were scattered across many developmental states, and neighboring aggregates often sat at visibly different stages.
Zooming In on Cell Types and Developmental Paths
Using computational maps of the single-cell data, the authors traced how normal cells moved from early solitary stages to late multicellular forms. They could clearly see the branching into future spore and stalk cells and confirmed that spore precursors form a more uniform group than the more varied stalk precursors. Remarkably, in the double mutant lacking cAMP pulses, cells still chose the same two main fates and followed a broadly similar path—just not at the same time. This shows that cAMP pulses are not needed to decide what each cell will become, but are crucial for making sure many cells reach those fates together.
Why This Matters for Multicellular Life
The study concludes that early waves of cAMP act as a master timing signal that aligns both the inner gene activity and the outer shapes of Dictyostelium cells. Once this early clock has done its job, development can unfold in a mostly synchronized way, helped along by other, more local signals between cells. Although this mechanism is specific to social amoebae, the broader principle—using rhythmic chemical signals to keep cells on the same schedule—resembles timing systems in animal embryos. By showing that single-cell RNA sequencing can quantify synchronicity over time, this work also provides a blueprint for probing how timing is controlled in more complex organisms, and what might happen when that timing breaks down.
Citation: Katoh-Kurasawa, M., Trnovec, L., Lehmann, P. et al. Early cAMP signaling orchestrates single-cell synchronicity throughout Dictyostelium development. Commun Biol 9, 543 (2026). https://doi.org/10.1038/s42003-026-09806-5
Keywords: cell synchronicity, Dictyostelium development, cAMP signaling, single-cell RNA sequencing, multicellular coordination