Clear Sky Science · en
CD25 modulation enhances broadly neutralizing antibody response of SARS-CoV-2 subunit vaccine
Why tuning vaccines still matters
As the coronavirus continues to spawn new variants, many people worry that today’s vaccines may not keep up. This study explores a different way to strengthen protection: instead of constantly redesigning the vaccine to match each new strain, the researchers ask whether we can tune the body’s own immune machinery so that a standard vaccine produces antibodies that recognize a wider range of variants.
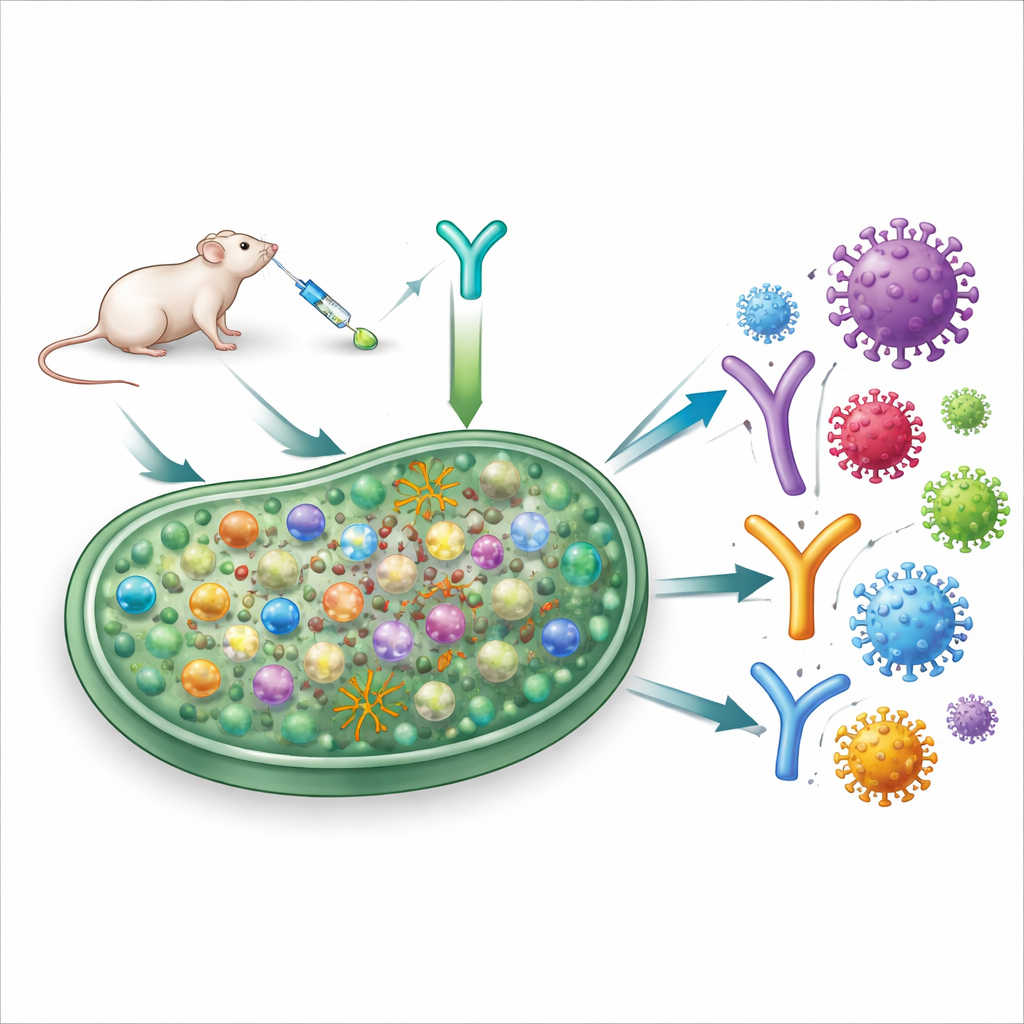
A traffic cop for immune responses
Our immune system walks a fine line between attacking germs and avoiding damage to our own tissues. Special “brake” cells help keep this balance by dialing down excessive responses. Many of these brake cells carry a molecule called CD25 on their surface. Doctors already use drugs that target CD25 in some cancer therapies, but their effects on vaccine responses are less clear. In this work, the authors combined a SARS-CoV-2 protein-based vaccine with an antibody that binds CD25 and temporarily disables these regulatory cells in mice.
Supercharging the antibody workshop
Powerful antibodies are forged in small structures inside lymph nodes known as germinal centers, where B cells repeatedly mutate and are tested for how well they bind a virus. The team found that adding the anti-CD25 antibody at the time of vaccination made these germinal centers larger and more active without simply inflating every branch of the immune system. Total B cell numbers stayed similar, but the pool of germinal center B cells expanded sharply, and within that pool, the number of B cells that specifically recognized the coronavirus spike protein rose substantially. Helper T cells that support this reaction also increased, while opposing regulatory T cells declined, tipping the balance toward a strong antibody workshop.
Stronger protection across many variants
The quantity of spike-binding antibodies in the blood is only part of the story; what really matters is how well those antibodies block infection. When the researchers tested blood from vaccinated mice against a panel of viral strains, animals that had received both the vaccine and the CD25 antibody showed much higher “neutralizing” activity than those given vaccine alone. This boost was seen not only against the original virus but also against several variants of concern, including multiple Omicron lineages that carry many spike mutations. In other words, even though overall antibody levels eventually looked similar across groups, the antibodies from the CD25-treated animals were better at stopping a broad set of viral versions.
Timing the immune boost
Because immune reactions unfold over days and weeks, the scientists asked when CD25 blocking works best. They compared giving the CD25 antibody just before vaccination, on the same day, or several days afterward. Short “windows” around the first shot turned out to be crucial. A dose on the vaccination day produced the strongest early germinal center burst and improved the range of variant recognition after later booster shots. A dose the day before vaccination more strongly enhanced virus-fighting T cells, which help clear infected cells and support long-term immunity. Remarkably, even when CD25 was blocked only during the very first immunization, the improved germinal center response left a lasting imprint that shaped better antibody quality months later.
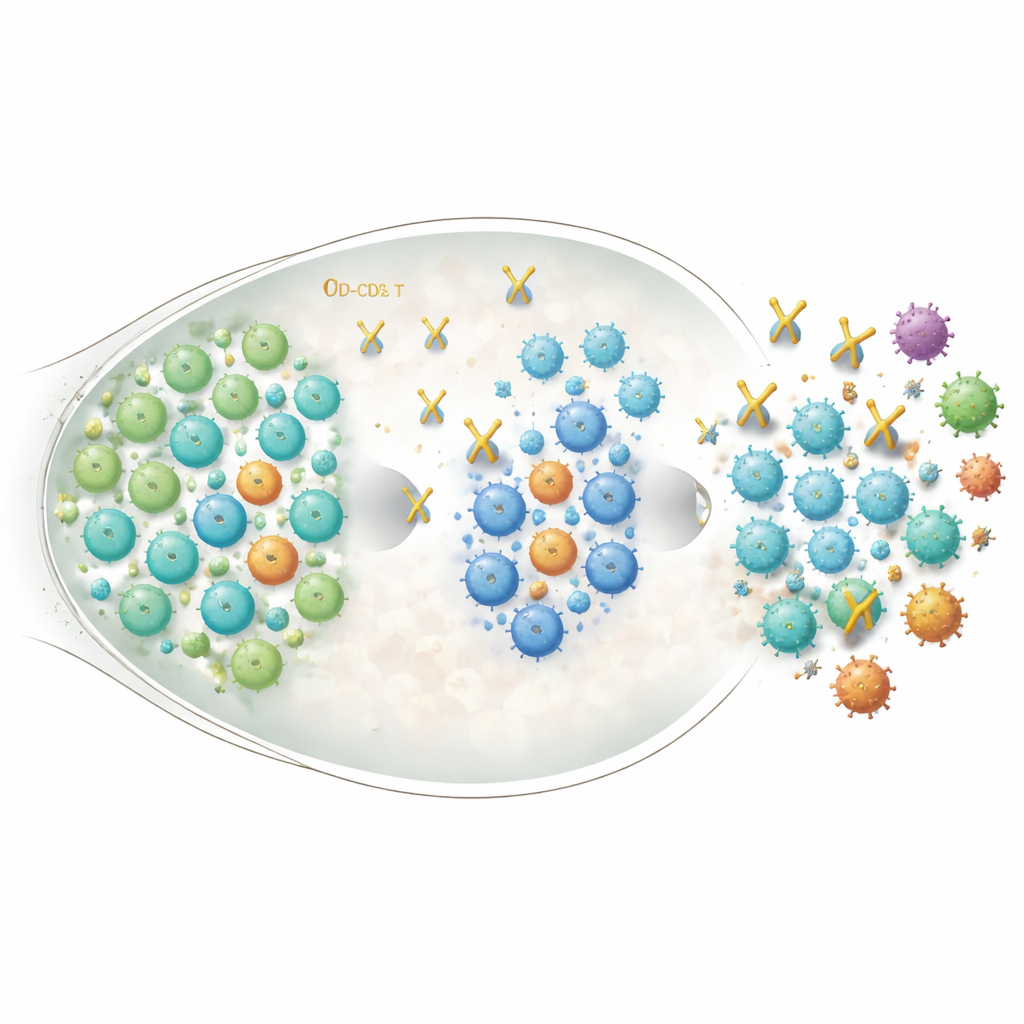
What this could mean for future vaccines
This mouse study suggests that briefly lifting some of the immune system’s brakes at the right moment can help a standard coronavirus subunit vaccine produce antibodies that are both stronger and more broadly protective, without chronically disturbing immune balance. If similar effects are confirmed in humans, carefully timed CD25-targeting treatments could become a general add-on strategy to make vaccines against rapidly changing viruses—not only SARS-CoV-2 but also HIV and others—more resilient to future variants.
Citation: Li, F., Yu, X., Zhang, C. et al. CD25 modulation enhances broadly neutralizing antibody response of SARS-CoV-2 subunit vaccine. Commun Biol 9, 447 (2026). https://doi.org/10.1038/s42003-026-09721-9
Keywords: COVID-19 vaccines, neutralizing antibodies, germinal center, regulatory T cells, vaccine adjuvant