Clear Sky Science · en
Corrosion inhibition of ductile iron in hydrochloric acid using 5-amino-1,3,4-thiadiazole-2-thiol: electrochemical and computational studies
Why protecting everyday metals matters
From water pipes and city valves to car parts and farm equipment, many common structures rely on ductile iron, a strong and affordable form of cast iron. Yet when these metal parts meet acidic liquids, such as cleaning solutions or industrial acids, they can slowly dissolve, weaken, and eventually fail. This study explores how a small organic molecule, called 5-ATT, can form a protective film on ductile iron in hydrochloric acid, sharply slowing down this hidden damage and helping extend the life of critical infrastructure.
How acid quietly eats away at ductile iron
In harsh chloride and acid environments, positively charged hydrogen and negatively charged chloride ions attack exposed metal atoms on the iron surface. Because ductile iron has a special microstructure, with graphite nodules embedded in a metallic matrix, it can develop tiny local batteries that speed up corrosion in certain spots. When researchers placed polished ductile iron samples in hydrochloric acid, they measured steady weight loss and a high corrosion rate, showing how quickly the surface can roughen and thin if left unprotected.
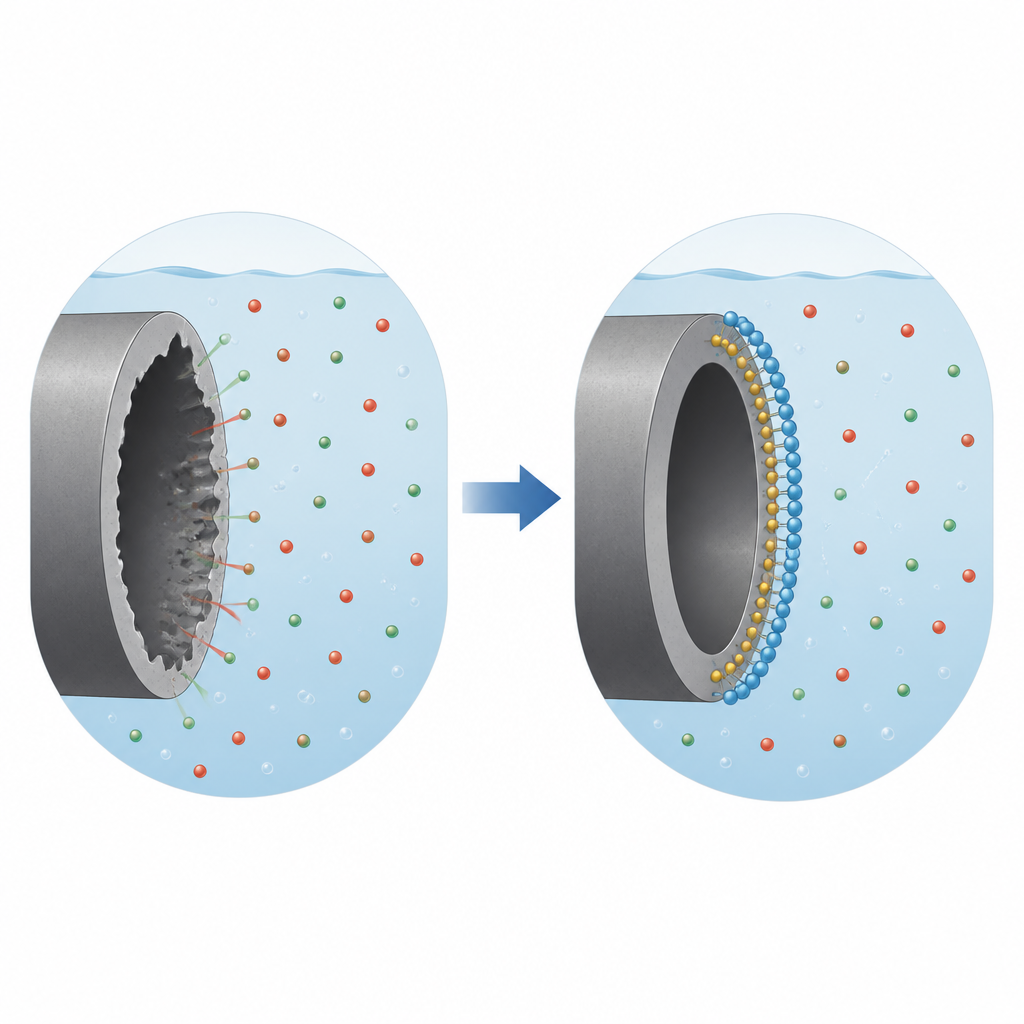
A small molecule that builds a protective shield
The team tested 5-ATT, an organic compound rich in nitrogen and sulfur atoms, as an additive in the acid solution. As they increased the concentration of 5-ATT, the iron lost less mass and the calculated corrosion rate dropped sharply, with protection reaching around 70–80 percent at the highest doses. Electrochemical tests, which track tiny electric currents linked to metal dissolution, showed that the corrosion current fell and the resistance to charge transfer rose as more 5-ATT was added. Together, these results indicate that 5-ATT molecules spread across the iron surface and act like a thin shield that blocks aggressive ions from reaching the metal.
Probing how the shield forms and holds
To see this shield directly, the researchers examined iron surfaces with and without 5-ATT using electron microscopy. In plain acid, the metal looked rough, cracked, and scarred by local attack. When 5-ATT was present, the surface appeared smoother and more uniform, with fewer visible defects. Chemical analysis of the surface detected carbon, nitrogen, and sulfur from the inhibitor, confirming that 5-ATT molecules were indeed clinging to the metal. By analyzing how well different mathematical models fit the coverage of 5-ATT, the team concluded that the molecules do not form a perfectly tidy single layer. Instead, they occupy multiple sites, interact with each other, and displace water in a more complex, real-world manner.
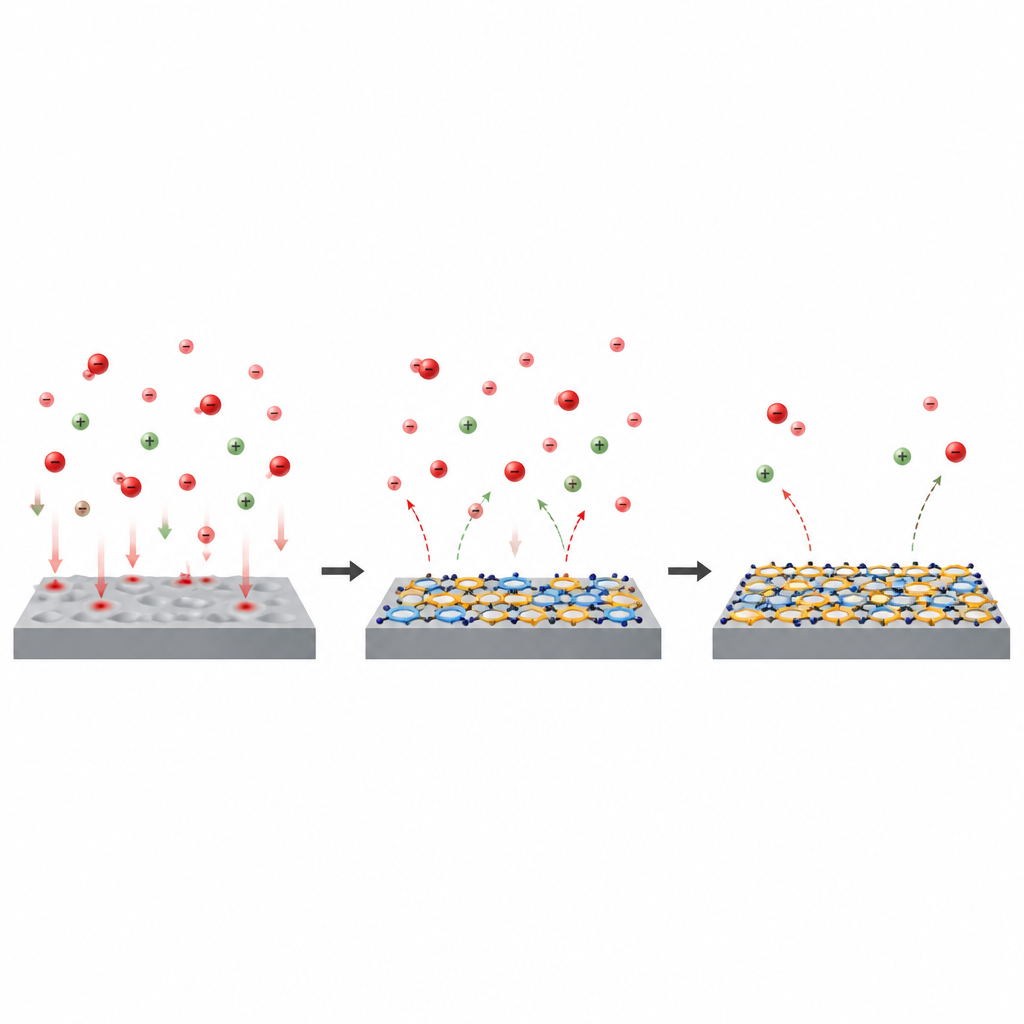
Looking inside the molecule with digital tools
Beyond lab measurements, the study used quantum chemical calculations and computer simulations to understand why 5-ATT works so well. The molecule can exist in two closely related forms, called thiol and thione, which differ in how a hydrogen atom and a double bond are arranged. Calculations showed that both forms have regions of concentrated electron density on their sulfur and nitrogen atoms, making these spots ideal for bonding to iron atoms. Simulations of the molecule lying on an iron surface in an acidic, water-rich environment revealed that 5-ATT tends to adopt a nearly flat posture, maximizing contact with the metal and with nearby ions. In water and acid, the thiol form in particular interacts strongly, helping to stabilize a compact protective film that pushes corrosive species away.
What this means for real-world metal protection
Put simply, this work shows that a carefully chosen small molecule can act like a smart, self-assembled raincoat for ductile iron in acid. 5-ATT adsorbs spontaneously onto the metal, using both physical attraction and chemical bonding to build a tight, multi-point barrier. This barrier slows both the loss of iron atoms and the reaction that releases hydrogen gas, cutting the corrosion rate by up to about four-fifths under the tested conditions. By linking detailed lab measurements with computer models of how 5-ATT sticks to iron at the atomic scale, the study provides a clear picture of how such inhibitors can be designed and improved to protect pipes, fittings, and other iron-based components that work in acidic environments.
Citation: Helmy, M., El-Zomrawy, A.A., Mogoda, A.S. et al. Corrosion inhibition of ductile iron in hydrochloric acid using 5-amino-1,3,4-thiadiazole-2-thiol: electrochemical and computational studies. Sci Rep 16, 14740 (2026). https://doi.org/10.1038/s41598-026-51250-2
Keywords: corrosion, ductile iron, hydrochloric acid, corrosion inhibitor, surface film