Clear Sky Science · en
Synthesis and characterization of 2-hydroxy-3-methoxyphenyl imino chitosan as a novel adsorbent for effective removal of methyl orange
Why cleaning colored water matters
Many everyday products—from brightly dyed clothes to printed paper—leave behind colorful chemical residues in water. These synthetic dyes can persist for a long time, harming aquatic life and potentially affecting human health even at very low levels. This study explores a new, plant- and shell-derived material that can grab and remove one such dye, called methyl orange, from water quickly and efficiently, offering a more environmentally friendly way to treat wastewater from textile and related industries.
A natural helper from seafood waste
The starting point for this work is chitosan, a substance obtained from chitin, the structural material found in the shells of shrimp, crabs and other crustaceans. Chitosan is inexpensive, biodegradable and non-toxic, and it already has a knack for attracting pollutants. However, ordinary chitosan does not always bind dyes strongly enough or remain stable under real-world treatment conditions. The researchers set out to upgrade this natural material by attaching an additional small organic component to its structure, creating new binding sites designed to capture dye molecules more effectively.
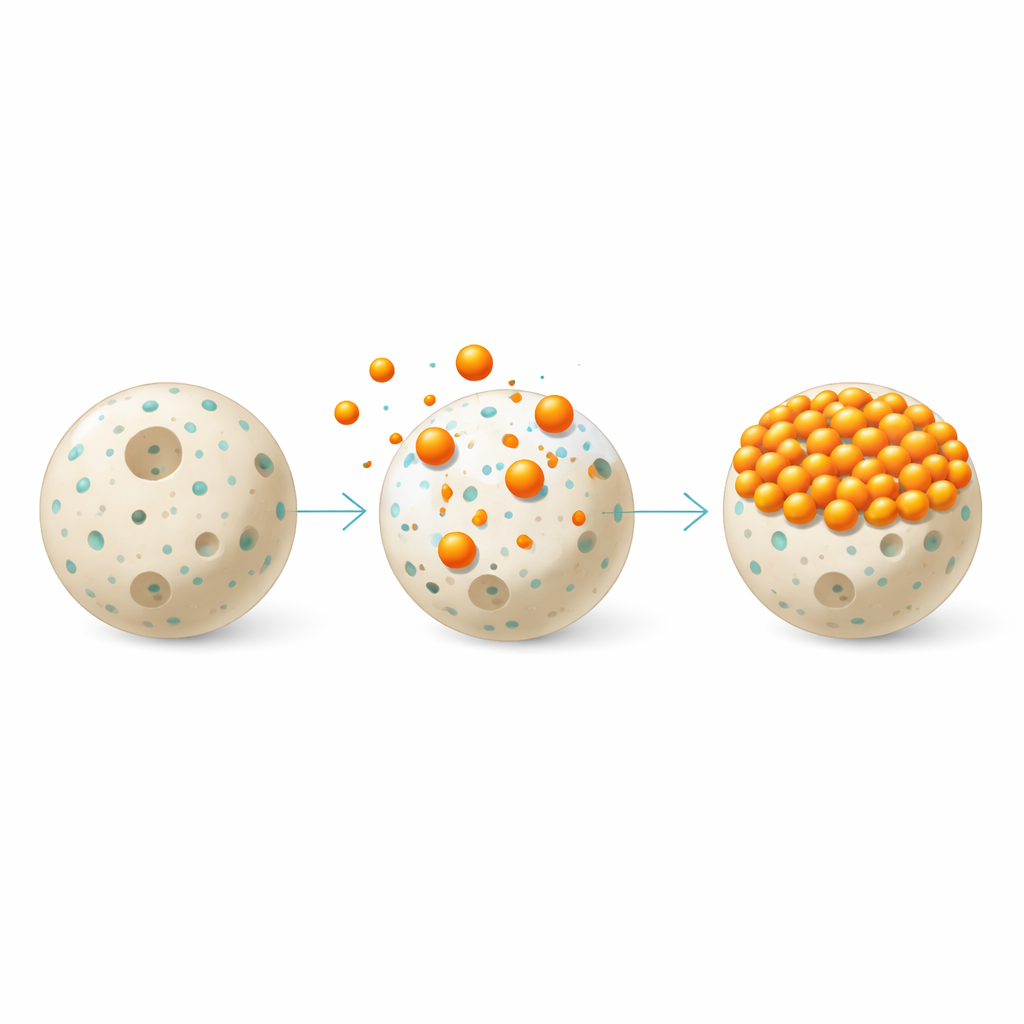
Building smarter cleaning beads
To make the improved material, the team first formed chitosan into small beads using a mild acid and a sodium hydroxide bath, yielding spherical particles that can be handled and separated easily from water. They then reacted these beads with a compound called 2-hydroxy-3-methoxybenzaldehyde using microwave heating. This step created new chemical links, known as imine bonds, between the chitosan and the added molecule, producing what they call 2-hydroxy-3-methoxyphenyl imino chitosan. Microscopy images showed that the modified beads had a rougher, more porous surface than the original smooth chitosan beads, and surface area measurements revealed that the available area for binding increased about fivefold, from 8.6 to 42.8 square meters per gram.
How the beads trap dye molecules
The team probed the structure and behavior of the new beads with several techniques. Infrared spectroscopy confirmed that the intended chemical bonds had formed and that the original free amine groups on chitosan had been largely converted into the new imine structures. X-ray measurements showed that the material became more amorphous—that is, less ordered—after modification and dye uptake, which is typical when flexible polymer chains are chemically altered. When methyl orange was allowed to contact the beads, changes in the infrared signals indicated that several types of interactions were at work: electrical attraction between positively charged sites on the bead surface and negatively charged groups on the dye, hydrogen bonding, and stacking between flat aromatic rings on both the dye and the added organic groups. Together, these forces explain why the dye sticks so strongly to the modified chitosan.
Finding the best conditions for cleanup
The researchers systematically varied the water’s acidity (pH), contact time, dye concentration, temperature and bead dosage to understand and optimize performance. The beads worked best in mildly acidic water around pH 4, where their surface carries positive charges that attract the negatively charged dye. Under these conditions, most of the dye was removed within about 20 minutes, with little improvement at longer times, showing that the process is fast. Raising the temperature reduced the amount of dye captured, indicating that the binding releases heat and is less favorable at higher temperatures. Mathematical models of the data suggested that dye molecules form a single, tightly packed layer on uniform sites on the bead surface and that the rate-limiting step involves chemical bonding rather than simple physical sticking.
Performance, reusability and real-world promise
Compared with many other low-cost sorbents made from plant peels, eggshells, clay minerals or unmodified chitosan, the new beads stood out by a wide margin, capturing up to about 445 milligrams of methyl orange per gram of material and removing more than 98 percent of the dye under optimized conditions. Importantly, the beads could be regenerated with simple acid washing and reused at least five times while still retaining over 84 percent of their original efficiency. Overall, this study shows that smartly modified chitosan beads, derived from abundant biological waste, can act as powerful, reusable "sponges" for stubborn dyes, pointing toward greener and more affordable treatment options for polluted industrial wastewater.
Citation: Khan, R., Zoreen, S., Khan, A. et al. Synthesis and characterization of 2-hydroxy-3-methoxyphenyl imino chitosan as a novel adsorbent for effective removal of methyl orange. Sci Rep 16, 14402 (2026). https://doi.org/10.1038/s41598-026-50311-w
Keywords: wastewater treatment, dye removal, chitosan beads, biosorbent, methyl orange