Clear Sky Science · en
Novel green synthesis of polyfunctionally substituted phthalazines promoted by visible light, DFT studies and molecular docking with antimicrobial and antibiofilm potency
Shining Light on Greener Germ-Fighting Chemistry
Antibiotic resistance and stubborn microbial films that cling to medical devices are growing threats to modern medicine. This study explores a way to make new germ-fighting molecules using nothing more high-tech than a household-style white LED lamp, common lab chemicals, and air. The researchers not only created these compounds in an environmentally friendly way, but also showed that some of them can strongly slow the growth of dangerous microbes and weaken their protective biofilms.
Why New Microbe Fighters Are Needed
Microbes such as Pseudomonas aeruginosa and Klebsiella pneumoniae often become resistant to multiple drugs, especially when they huddle together in slimy biofilms on catheters, implants, and other devices. In a biofilm, bacteria sit inside a self-made gel that shields them from antibiotics and from the immune system. This makes infections difficult to cure and easy to relapse. Chemists have long known a family of ring-shaped molecules called phthalazines that show promise against a range of diseases, from infections to cancer. However, traditional ways to build these rings can require high heat, toxic metals, and harsh conditions that are not ideal for large-scale or sustainable drug discovery.
Making Complex Rings with Gentle Light
In this work, the team developed a “green” route to a series of ten new phthalazine-based molecules. Instead of metal catalysts and high temperatures, they mixed two types of starting materials in ethanol, added a small amount of a simple organic base, and then shone white LED light on the mixture while it sat in open air at room temperature. Under these mild conditions, the reaction that had refused to proceed with normal heating suddenly went almost to completion, giving the phthalazine products in impressive 90–93% yields. Control experiments, including the addition of a radical-trapping additive, showed that the light triggers a chain of short-lived radical particles that ultimately knit the carbon and nitrogen atoms into the new ring system. A proposed step-by-step pathway explains how light excites one partner, how radicals join, and how the final stable ring emerges.
Testing the New Molecules Against Tough Microbes
The researchers then asked whether these freshly made compounds could tackle real microbial foes. Using clinical isolates of bacteria and a yeast that are known to resist multiple drugs, they measured both the zones of growth inhibition on agar plates and the lowest concentration needed to stop visible growth in liquid culture. Two members of the series, labeled 3g and 3j, stood out. Compound 3g inhibited P. aeruginosa at just over 3 micrograms per milliliter and K. pneumoniae at 12.5 micrograms per milliliter, while 3j also showed strong effects, especially against P. aeruginosa. Although a standard antibiotic, ciprofloxacin, was still more potent, the new molecules reached a performance level that makes them interesting as lead structures for further refinement.
Breaking Down the Microbial Shield
Stopping free-floating microbes is only half the battle; breaking their protective films is equally important. The team used a staining test in small plates to see how well the compounds could prevent biofilms from forming. Again, 3g and 3j were best in class: 3g reduced biofilm formation by 81% for P. aeruginosa and by 65–60% for other tested microbes, while 3j cut biofilms by up to 75% for some strains. Several other compounds showed moderate effects, and one was essentially inactive, which helped the authors begin to link small changes in molecular structure to gains or losses in activity. For example, rings decorated with strongly electron-pulling groups such as chlorine or nitro tended to give better antimicrobial and antibiofilm power than those with electron-donating groups like methyl.
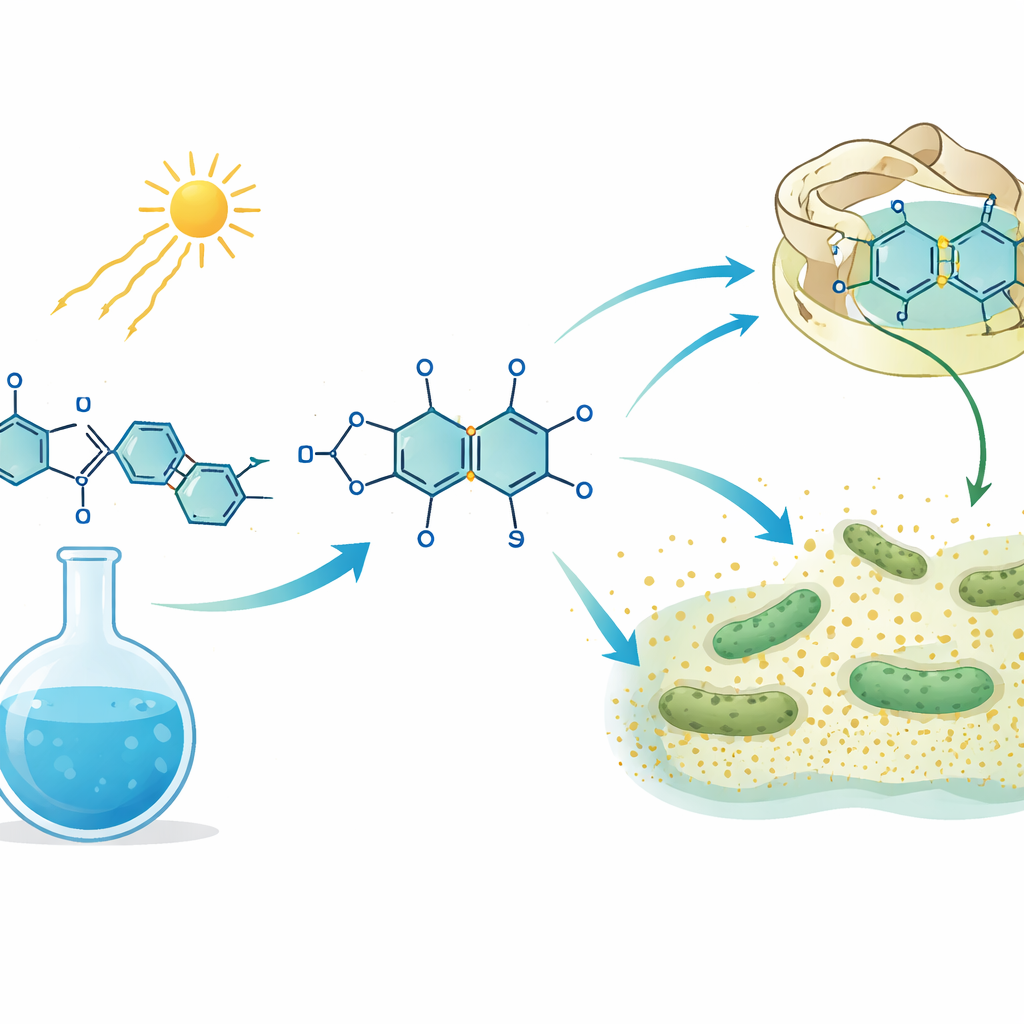
Peering Inside the Molecules and Their Targets
To understand why 3g and 3j worked so well, the team turned to computer modeling. They optimized the shapes and electron distributions of the molecules using quantum chemistry methods, mapped where positive and negative charge tends to sit, and calculated how easily electrons can move within each structure. These features, such as a relatively small gap between key energy levels, hint that 3j in particular can interact strongly with biological targets. Docking simulations then placed the molecules into the pockets of two proteins linked to infection and sterol metabolism. Both 3g and 3j formed tight, stable complexes with these proteins, making multiple hydrogen bonds and snug hydrophobic contacts, with 3j showing slightly stronger predicted binding. This digital view supports the laboratory findings and suggests ways chemists might further tune the molecules.
From Lab Light to Future Medicines
Together, the results show that simple white-light illumination can drive an efficient, metal-free route to complex phthalazine structures using mild conditions and common solvents. Among the products, compounds 3g and 3j emerge as especially promising candidates, combining solid antimicrobial strength with the ability to weaken stubborn biofilms and form stable interactions with target proteins in simulations. While much work remains before any drug can reach patients, this study offers a blueprint for marrying greener synthetic chemistry with the urgent search for new treatments against resistant, biofilm-forming pathogens.
Citation: Mekheimer, R.A., Khalifa, B.A., Hashem, Z.S. et al. Novel green synthesis of polyfunctionally substituted phthalazines promoted by visible light, DFT studies and molecular docking with antimicrobial and antibiofilm potency. Sci Rep 16, 14275 (2026). https://doi.org/10.1038/s41598-026-47154-w
Keywords: antibiotic resistance, biofilms, green chemistry, visible light synthesis, phthalazine derivatives