Clear Sky Science · en
Investigating uncharacterised genes in Saccharomyces cerevisiae using robot scientists
Why this matters for everyday biology
We often talk about having the full genetic blueprint of an organism, but for many genes we still do not know what they actually do. This study tackles that mystery in baker’s yeast, a workhorse of both science and industry, by combining automated "robot scientists" with computer models. The researchers show how this approach can uncover the role of a previously unstudied gene involved in how yeast switches its energy source, a change that affects growth, metabolism, and ultimately how cells cope with changing environments.
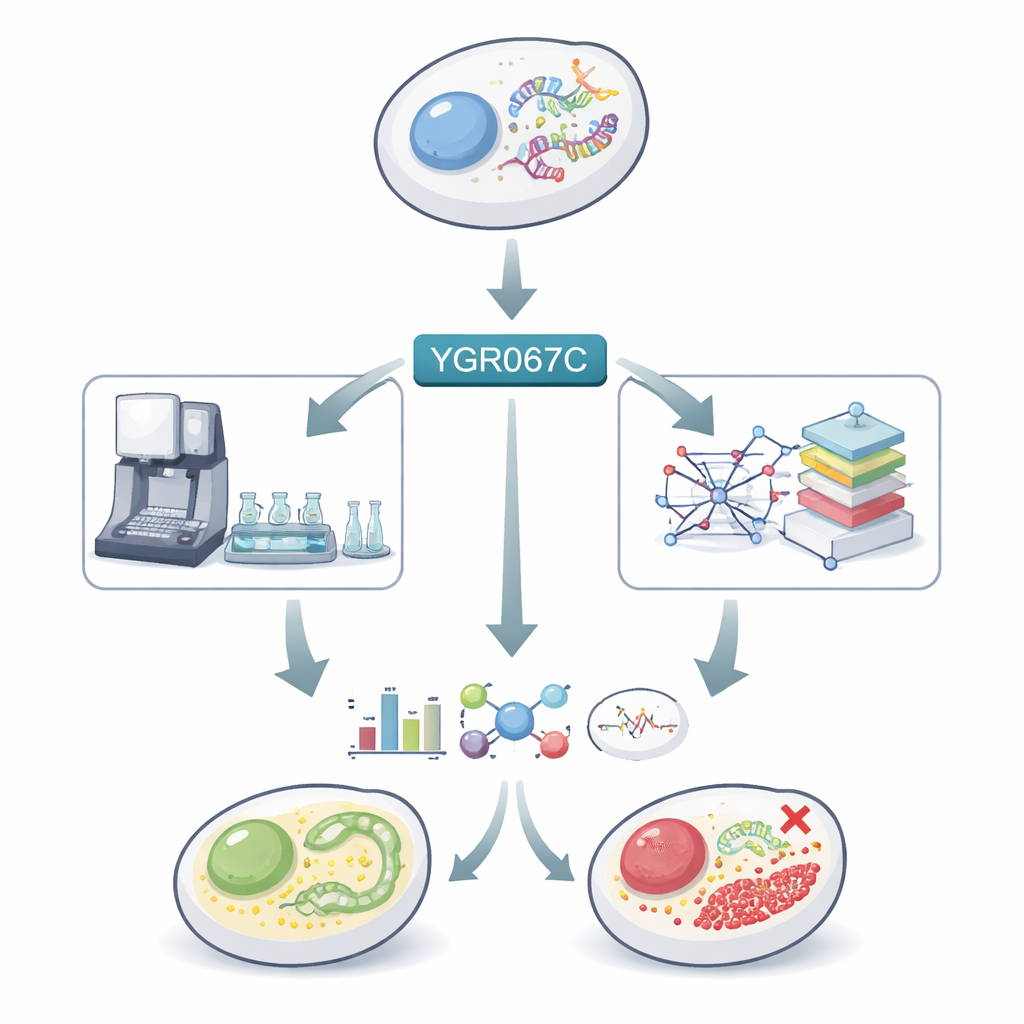
A hidden gene in a very familiar organism
Baker’s yeast has been studied for decades, yet almost 900 of its roughly 6,000 genes remain poorly understood. One of these, called YGR067C, produces a protein with a structural motif typical of gene regulators, hinting that it might control other genes. Earlier work suggested that changes in this gene helped yeast adapt to growing on unusual food sources like methanol. The authors therefore suspected that YGR067C might be involved in managing the shift from burning sugar to burning the alcohol ethanol, a major metabolic “gear change” that yeast undergoes when glucose runs out.
Letting robots and models drive the experiments
To explore this idea, the team used an automated laboratory platform nicknamed Eve. Eve grew normal yeast and a strain in which YGR067C was deleted in tiny wells filled with nutrient medium containing only a small amount of glucose, forcing the cultures through the classic two-phase growth pattern: first a sugar-burning phase, then an ethanol-burning phase. The robot carefully tracked growth and collected samples at chosen times. These samples were then analyzed at three levels: which genes were active (transcriptomics), which small molecules were present (metabolomics), and how fast and dense the cultures grew. In parallel, the researchers used two types of computer models of yeast metabolism to predict what should happen when the gene’s function is disrupted, not only in the pathways directly linked to respiration but also in the wider network of reactions across the cell.
What happens when the gene is removed
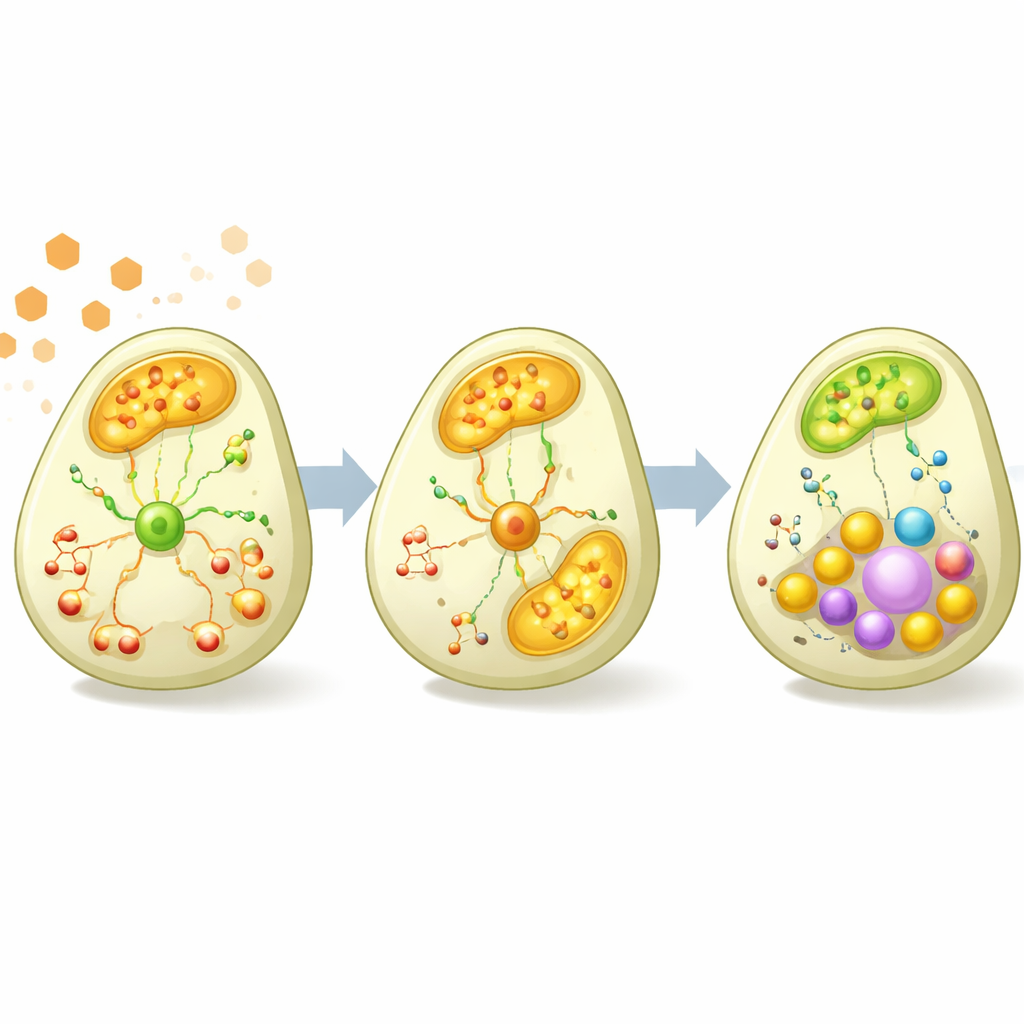
The data revealed that deleting YGR067C changes both growth and internal chemistry. The mutant strain grew slightly faster and reached a higher cell density than the normal strain under the tested conditions, suggesting that less energy was being invested in certain costly cellular machinery. During the sugar-burning phase, many genes involved in key energy pathways inside mitochondria – such as the citric acid cycle, oxidative phosphorylation, and the glyoxylate cycle – were less active in the mutant. At the same time, there were hints that sugar breakdown itself was more active, and two components of a cellular “proton pump” that helps maintain acidity were more strongly expressed, consistent with higher production of acidic byproducts from fermentation.
Lingering effects during the alcohol-burning phase
Once cultures had shifted toward using ethanol, differences in gene activity between the two strains largely faded, but differences in metabolites became pronounced. In the mutant, several related molecules that carry and store cellular reducing power, including different forms of NAD, built up to higher levels, as did some amino acids such as glutamate and asparagine. Pathway analyses indicated broader changes in amino acid production, vitamin metabolism, and lipid-related pathways. Together, these findings suggest that although YGR067C’s gene-regulating activity is strongest when glucose is present, the metabolic consequences of its absence extend into the ethanol-using phase, reshaping how the cell balances energy production, building-block synthesis, and growth.
What this tells us about the gene and the bigger picture
By combining automated experiments with multi-layered measurements and several modeling strategies, the study arrives at a clear, intuitive take-home message for non-specialists: YGR067C helps switch on and fine-tune the yeast’s machinery for burning ethanol through respiration as sugar runs low. When the gene is missing, the cells lean more heavily on simple sugar fermentation, invest less in mitochondrial energy pathways, and show knock-on changes in key energy-carrying molecules and amino acid production, while growing somewhat faster under the tested conditions. Just as importantly, the work showcases a general roadmap for turning vague ideas about mysterious genes into concrete, testable predictions—an approach that could be scaled up to decode many of the remaining “unknown” genes in yeast and other organisms.
Citation: Bjurström, E.Y., Gower, A.H., Lasin, P. et al. Investigating uncharacterised genes in Saccharomyces cerevisiae using robot scientists. Sci Rep 16, 10999 (2026). https://doi.org/10.1038/s41598-026-46236-z
Keywords: yeast genetics, diauxic shift, metabolism, robotic laboratory, gene regulation