Clear Sky Science · en
Genomic insights into the spread and evolution of insecticide resistance variants in Anopheles gambiae s.l. from Burkina Faso
Why these mosquitoes matter for everyone
Across much of Africa, including Burkina Faso, the main shield against malaria is still insecticide-treated bed nets and indoor spraying. But the mosquitoes that spread malaria are evolving ways to survive these chemicals. This study uses powerful DNA tools to look inside the genomes of malaria mosquitoes across Burkina Faso, revealing how resistance is spreading and changing. Understanding this hidden arms race helps explain why some control tools are losing power—and how smarter, faster surveillance could keep lifesaving interventions working.
Looking across a country’s mosquito populations
The researchers collected malaria mosquitoes from eight villages spanning the main climate zones of Burkina Faso, from wetter southern regions to the drier Sahel in the north. They then used whole-genome sequencing to read the DNA of 665 mosquitoes belonging to three closely related species that are major malaria carriers. By focusing on genes targeted by insecticides, as well as genes involved in breaking down chemicals, they were able to map where different resistance variants occur, how common they are, and how they are combined inside individual mosquitoes.
Key resistance switches in nerve-cell targets
Many widely used insecticides, such as pyrethroids, act on a protein in mosquito nerve cells called a sodium channel. Small DNA changes, known as mutations, in the gene for this protein can blunt the effect of the insecticide and are often called “knockdown resistance” variants. The team found five such mutations—named L995F, L995S, V402L, I1527T and N1570Y—circulating at high levels in Burkina Faso’s mosquito populations. In one species, Anopheles gambiae sensu stricto, the L995F mutation was almost fixed, meaning nearly every mosquito carried it. In another species, Anopheles coluzzii, L995F was common but not dominant, while other mutations such as V402L and I1527T appeared together at high frequencies, especially in certain ecological zones.
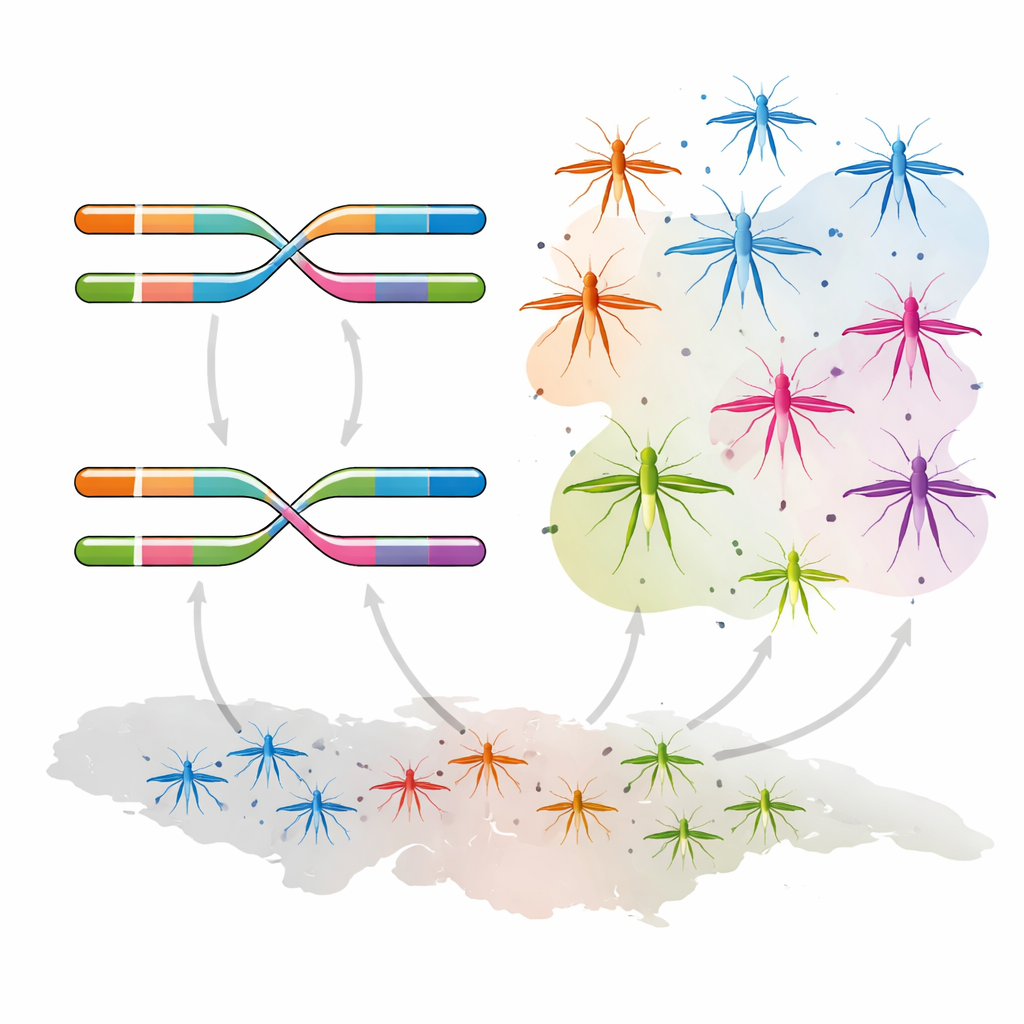
New genetic combinations and rapid mixing
When the team examined how these mutations were arranged on pairs of chromosomes in each mosquito, they uncovered six major “diplotype” groups—distinct combinations of resistance variants. Most of this complexity was concentrated in An. coluzzii, where new genotypes had emerged that combined different resistance mutations in novel ways. Strong statistical associations showed that one version of V402L tended to occur together with I1527T, suggesting they are being selected as a package, while an alternative version of V402L appears to evolve more independently. A network analysis of DNA haplotypes indicated that recombination and gene flow are shuffling these resistance packages between populations and species, with little sign of geographic barriers inside the country. In practical terms, once a powerful combination appears, it can spread quickly and silently.
Multiple backup systems for surviving insecticides
Target-site changes are only part of the story. The researchers also studied another gene, Ace1, which is targeted by organophosphate and carbamate insecticides used in indoor spraying. They detected both a well-known mutation (G280S) and copies of the Ace1 gene being duplicated, particularly in southern and agricultural areas where these chemicals are heavily used. On top of that, they found widespread changes in the number of copies of detoxification genes—families of enzymes that help mosquitoes break down insecticides. Many mosquitoes carried extra copies of certain cytochrome P450s, esterases and glutathione transferases, while others had lost particular genes entirely. These shifts in copy number were common across the country and differed between species, pointing to a rich and evolving toolkit of metabolic resistance.
What this means for malaria control
Altogether, the study shows that malaria mosquitoes in Burkina Faso are not relying on a single trick but on a growing mix of genetic defenses, which can blend and spread rapidly. Classic resistance mutations in nerve-cell targets, newer target-site variants, and changes in detoxification and other genes now coexist in the same populations, sometimes in the same mosquitoes. For non-specialists, the take-home message is that resistance is more widespread and genetically complex than routine field tests can reveal. The authors argue that ongoing genomic surveillance—regularly reading mosquito DNA at scale and feeding this information into decision-making—is essential to keep current tools effective, guide the choice and rotation of insecticides, and support the design of next-generation interventions that can stay ahead of mosquito evolution.
Citation: Kientega, M., Kaboré, H., Sawadogo, G. et al. Genomic insights into the spread and evolution of insecticide resistance variants in Anopheles gambiae s.l. from Burkina Faso. Sci Rep 16, 12459 (2026). https://doi.org/10.1038/s41598-026-45950-y
Keywords: malaria mosquitoes, insecticide resistance, genomic surveillance, Burkina Faso, vector control