Clear Sky Science · en
Elucidation of the process of delayed colonic perforation after endoscopic thermal injury in a rat model
Why this matters for everyday colon exams
Many people now have polyps or early tumors removed from the colon using tiny tools passed through a scope, avoiding major surgery. While this approach is generally safe, a rare but dangerous problem can occur days later: a small hole can open in the colon wall, causing life‑threatening infection. This study in rats asks a simple but crucial question: what actually happens inside the colon wall after it is heated by endoscopic instruments, and why do some of these injuries slowly turn into a tear?
A closer look at a delayed problem
The researchers focused on “delayed perforation,” a perforation that is not seen during the colonoscopy itself but appears afterward. Doctors suspect that excessive heat from electric tools can damage deeper layers of the colon, but the detailed chain of events has remained murky. To unravel this, the team created carefully controlled thermal injuries in rat colons using an endoscope, then examined the tissues at set time points up to 48 hours later. They also compared normal rats with rats whose gut bacteria had been drastically reduced by strong antibiotics.

From early burn to full‑thickness hole
Right after the heat injury, the inner lining of the colon turned pale and the muscle layer beneath it was already weakened and thinned. Over the first day, the injured area changed color on the outer surface, and the inner surface evolved into a deeper ulcer. By 24 hours, blood vessels in the lining and just below it became markedly widened and congested, creating a “zone of stasis” similar to what is seen around skin burns: tissue is badly stressed but not yet dead. By 36 hours, the gland‑like structures of the lining had largely disappeared, signaling widespread cell death, and by 48 hours the muscle layer had vanished at the burn site, leaving a complete hole. At that point, all examined rats in the main model had developed a delayed perforation.
How bacteria and immune cells push the damage deeper
The team then asked what drives this shift from a threatened but salvageable zone to irreversible breakdown. They tracked two key players: gut bacteria and the body’s first‑responder white blood cells. As the lining died back around 36 hours, bacteria such as Escherichia coli, normally confined to the gut cavity, began to appear deeper in the colon wall. At the same time, the number of immune cells marked by an enzyme called myeloperoxidase surged in the supporting layers. This combination of invading microbes and intense inflammation matched the expansion of the dead “zone of coagulation” seen in burn science, and preceded the final loss of the muscle layer.
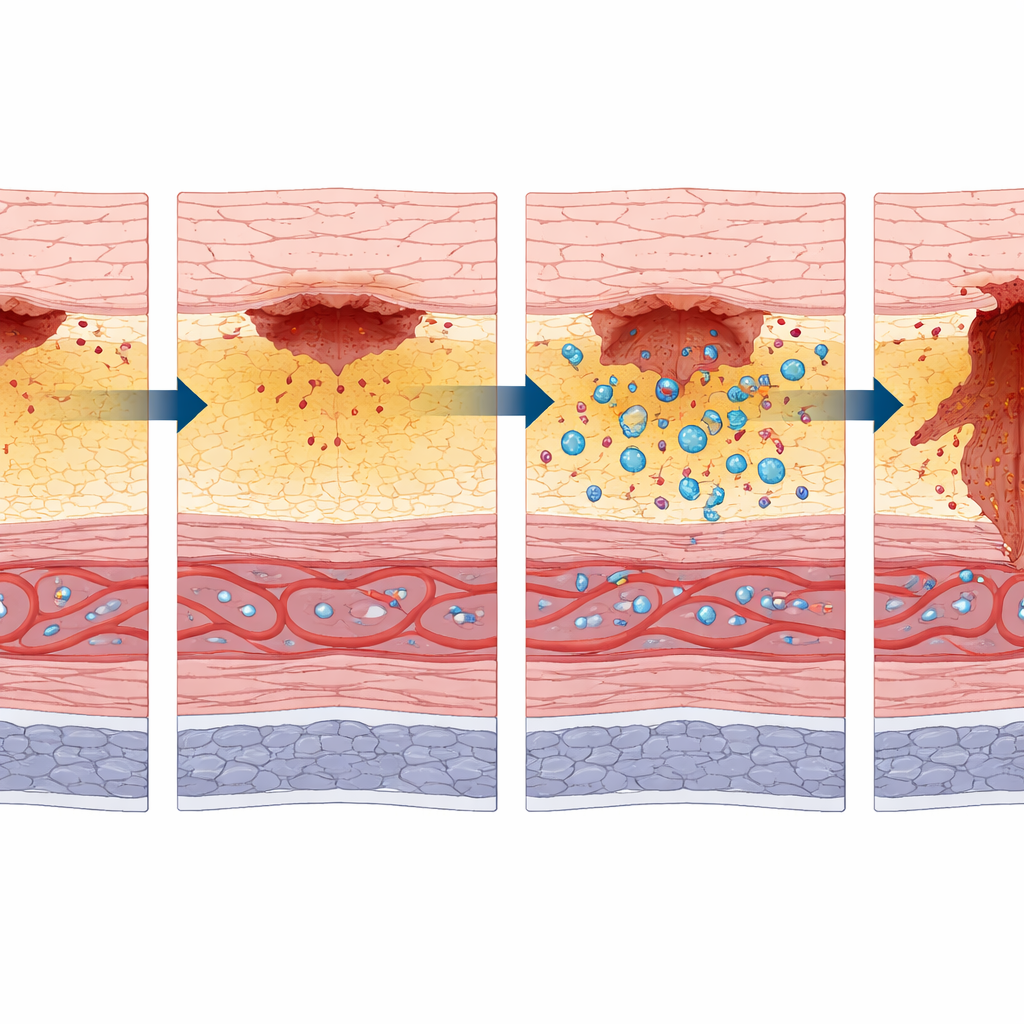
What happens when gut germs are mostly removed
To test how important bacteria are to this process, the scientists created “pseudo‑germ‑free” rats by giving them a cocktail of antibiotics for five days, sharply lowering bacterial enzyme activity in their stool. When these rats received the same thermal colon injury, they still developed ulcers and some muscle damage, but the results were strikingly different: only about one in nine developed a delayed perforation, compared with every single rat in the untreated control group. In the antibiotic‑treated group, the muscle layer was thin but still present, bacteria did not appear in the deeper wall, and far fewer immune cells crowded the outer surface. The rare treated rat that did perforate showed signs that reduced blood flow, not infection, may have been the main driver.
What this could mean for safer colon procedures
To a lay reader, the key message is that a heat injury in the colon does not instantly become a hole; instead, there is a vulnerable window during which stressed but living tissue can either recover or be pushed over the edge. This study shows that, at least in rats, invading gut bacteria and the resulting inflammation play a major role in tipping the balance toward perforation, in much the same way that infection worsens severe skin burns. While the rat colon differs from the human colon and the experimental burns were intentionally severe, the work suggests that future patient care might focus not only on careful control of heat during endoscopy, but also on ways to protect the damaged ulcer base from local infection—perhaps with targeted, burn‑style dressings—so that a temporary injury does not progress to a life‑threatening tear.
Citation: Sakae, T., Maeda, H., Sasaki, F. et al. Elucidation of the process of delayed colonic perforation after endoscopic thermal injury in a rat model. Sci Rep 16, 14538 (2026). https://doi.org/10.1038/s41598-026-45443-y
Keywords: colonoscopy complications, thermal injury, gut bacteria, intestinal perforation, endoscopic therapy