Clear Sky Science · en
Exercise and time-restricted and/or dietary feeding jointly improve hepatic lipid homeostasis in diet-induced obese mice
Why everyday habits matter for your liver
Obesity is often discussed in terms of waistlines and bathroom scales, but it also quietly reshapes one of our most vital organs: the liver. When too much fat builds up there, people can develop a condition now called metabolic dysfunction-associated steatotic liver disease (MASLD), which raises the risk of diabetes, heart disease, and liver failure. This study in mice asks a practical question with clear relevance for everyday life: if you already have diet-induced obesity, how far can changes in what you eat, when you eat, and how much you move actually reverse liver damage and restore healthier fat handling inside liver cells?
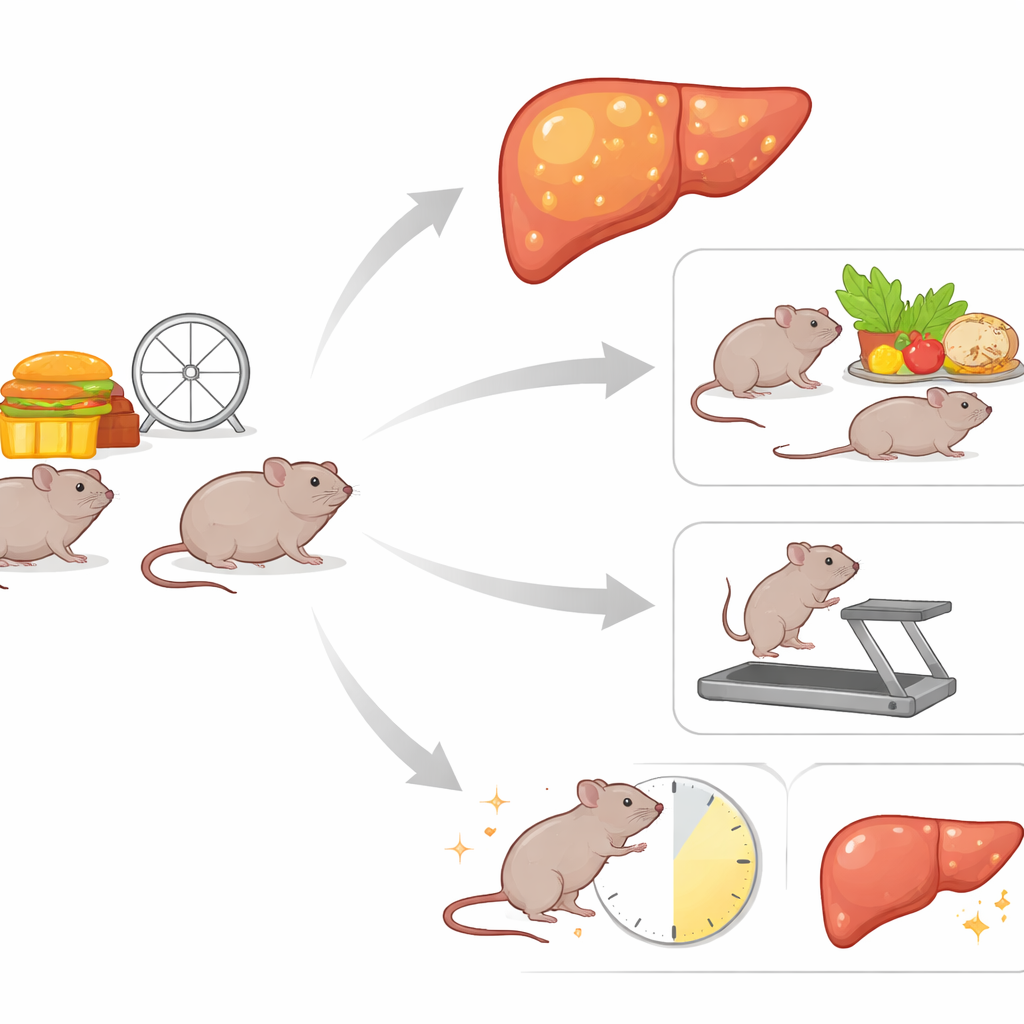
How the study was set up
Researchers first fed young female mice a very high-fat diet for six months, long enough to make them obese and to stress their livers. After this “induction” period, the animals were split into six groups for another six months. One group simply stayed on the high-fat diet. Others either continued on that diet but added treadmill running, added both treadmill running and time-restricted feeding (food available only during the active night phase), or switched to a low-fat diet with or without those same add-ons. This design allowed the team to compare the impact of diet change, exercise, and eating-window timing alone and in combination on body weight, blood markers of liver injury, detailed liver fat composition, and the activity of key genes that control how the liver makes and burns fat.
Weight loss, liver damage, and simple blood tests
Switching from a high-fat to a low-fat diet had the biggest effect on body weight: mice that changed diets ended the study weighing about half as much as those kept on high fat. Exercise or time-restricted feeding on their own did not cause large weight loss when the high-fat diet continued, but combining both produced a modest drop, hinting that aligning feeding with the animals’ natural daily rhythms helps metabolism even without changing what they eat. The same pattern showed up in basic liver blood tests. Mice that stayed on the high-fat diet had high levels of AST and ALT—enzymes that leak into the blood when liver cells are injured. Adding treadmill running and especially time-restricted feeding pushed these levels back toward normal, and diet change to low fat brought them down the most. Albumin, a protein made by the liver, stayed within typical ranges, suggesting the organ’s basic synthetic function remained intact while damage markers improved.
What changed inside the liver’s fat landscape
Beyond total fat, the team used lipidomics, a chemistry toolkit that can catalog many individual lipid types at once, to see how the liver’s “fat landscape” shifted. High-fat feeding distorted this landscape, boosting particular species such as certain ceramides and phospholipids that have been linked to insulin resistance and liver dysfunction. When mice switched to a low-fat diet—especially when combined with treadmill running—the balance of lipids shifted again. Levels of sphingomyelins and closely related molecules called dihydrosphingomyelins, as well as lyso- and phosphatidylcholines, rose in patterns that likely reflect a remodeling of cell membranes as the liver recovers. Importantly, many of these lipid species were negatively correlated with AST and ALT: when they increased, signs of liver injury decreased, suggesting that at least some of these changes are part of a healing response rather than a new kind of damage.
How the liver’s control switches responded
The researchers also measured activity of genes that act as control switches for fat handling—some that promote building new fat and cholesterol, and others that promote burning fat for energy. With diet change plus exercise and time-restricted feeding, genes that drive fat creation, such as those governed by sterol-regulatory and liver X receptors, tended to dial down. At the same time, genes tied to fat burning in mitochondria and peroxisomes showed a more nuanced response: in some high-fat groups with exercise, their activity was relatively high, likely reflecting the liver’s attempt to cope with ongoing fat overload, while in low-fat groups these genes could relax as the overall fat pressure eased. Together, the patterns point not to a single switch being flipped but to a broad retuning of how the liver balances fat storage, breakdown, and export.
What this means for people and future research
For a lay reader, the main message is straightforward: changing what you eat remains the most powerful way to reverse obesity-related liver stress, but exercise and a consistent daily eating window can add meaningful benefits—even if an unhealthy diet has not yet been fully corrected. In these mice, combining a lower-fat diet with regular treadmill running and nighttime-restricted feeding improved weight, reduced blood signs of liver damage, and reshaped the mix of fats and regulatory genes inside liver cells toward a healthier state. The work is exploratory and in animals, so it does not prescribe a specific regimen for people. Still, it highlights a hopeful principle: the liver is remarkably adaptable, and coordinated changes in diet, movement, and meal timing can help nudge it back toward balance after years of overload.
Citation: Guerra, N.P., Bräuer, A.U., Gräler, M.H. et al. Exercise and time-restricted and/or dietary feeding jointly improve hepatic lipid homeostasis in diet-induced obese mice. Sci Rep 16, 10508 (2026). https://doi.org/10.1038/s41598-026-45394-4
Keywords: fatty liver, time-restricted feeding, exercise, high-fat diet, lipid metabolism