Clear Sky Science · en
Integrating metabolomics and machine learning with in silico analysis to identify early biomarkers and molecular interactions in sepsis-associated acute kidney injury
Why spotting kidney danger early matters
In intensive care units, many patients with life-threatening infections (sepsis) suddenly develop severe kidney problems. Doctors currently rely on a blood waste product called creatinine to detect this damage, but creatinine rises only after the kidneys are already injured. This study asked a critical question: can hidden chemical signals in the blood reveal that sepsis-related kidney damage is coming a day in advance, giving doctors a chance to act earlier?
Looking for hidden chemical clues in blood
The researchers followed fifty adults in an intensive care unit who had sepsis. Some went on to develop acute kidney injury within two days, while others did not. Importantly, blood samples were taken soon after sepsis was diagnosed, before the kidneys had clearly failed. Instead of testing a few known markers, the team used a broad “chemical fingerprinting” method to scan for more than a thousand small molecules in the blood, known collectively as metabolites. These molecules reflect how the body is using and transforming nutrients and energy, and they can change quickly when organs are under stress.
From thousands of signals to a handful of key markers
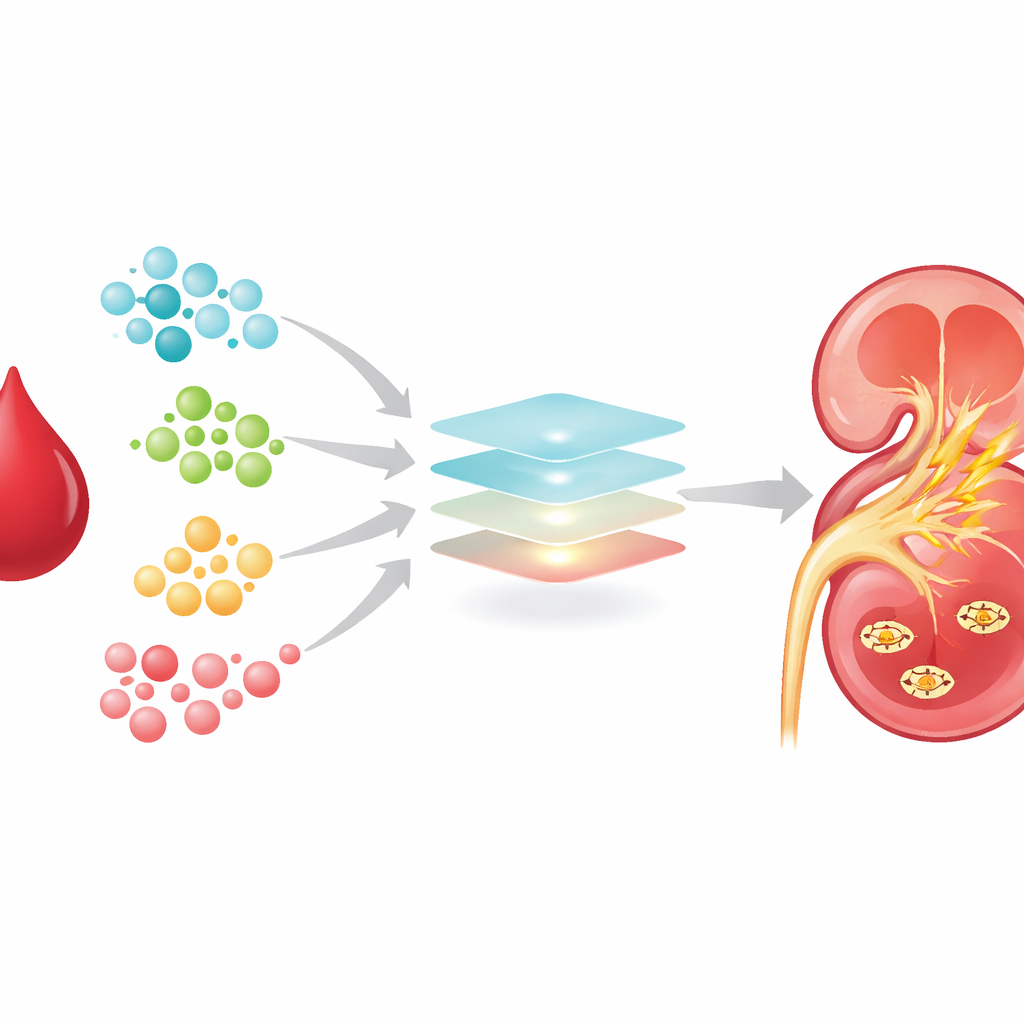
Computer analysis showed that patients who later developed kidney injury had a distinctly different blood chemistry pattern from those who did not. Out of 1,425 features, 634 specific metabolites could be reliably identified, and 150 of these differed strongly between the two patient groups. Many of the changes pointed to disturbed handling of amino acids and fats, especially pathways involving the amino acid phenylalanine and the vitamin-like molecule NAD+, which is essential for energy production in cells. To turn this sprawling data into something clinically useful, the researchers used machine learning methods designed to sift through many variables and pick out the most informative ones.
A five-molecule warning panel
Two independent computer algorithms converged on a small set of five metabolites that were especially powerful in distinguishing between patients who would and would not develop kidney injury. These were sebacic acid, a type of acylcarnitine, methyl acetate, threonic acid (a breakdown product of vitamin C), and a molecule called 1-RDN, which is closely tied to NAD+ production. All five were higher in patients headed toward kidney injury. When the team built a prediction model using only these five molecules and tested it in a rigorous “leave-one-out” fashion, the best-performing model correctly separated high- and low-risk patients with strong accuracy, outperforming traditional measures such as creatinine and common inflammatory markers.
How metabolism and kidney stress may be linked
Beyond prediction, the pattern of metabolites tells a story about what may be happening inside the kidneys during early sepsis. Elevated acylcarnitine and sebacic acid suggest that kidney cells are struggling to burn fats properly, leading to energy shortfalls. Rising levels of 1-RDN point to stressed attempts to rebuild NAD+, the fuel-handling cofactor that helps power mitochondria, the cell’s energy factories. The buildup of threonic acid hints that the body is drawing heavily on vitamin C defenses against oxidative damage. Methyl acetate, formed from acetone, may further worsen oxidative stress and drain energy. Taken together, these shifts paint a picture of kidneys caught in a loop of faulty fat burning, energy collapse, and oxidative injury even before conventional tests flag trouble.
A possible molecular bridge from chemistry to damage
To probe deeper, the team used computer docking simulations to see whether any of the key metabolites might physically interact with proteins known to be involved in kidney disease. They found that 1-RDN could bind tightly to phenylalanine hydroxylase, an enzyme that helps process the amino acid phenylalanine and is present not only in the liver but also in kidney tissue. This raises the possibility that shifts in NAD+-related chemistry could directly alter amino acid handling in the kidney itself, linking energy stress with changes in signaling molecules derived from phenylalanine. Although this idea still needs to be tested in laboratory and animal experiments, it offers a concrete starting point for exploring how metabolic disruptions might trigger structural kidney damage.
What this could mean for future care
This work suggests that a simple blood test measuring a small panel of metabolites could warn doctors, up to a day in advance, which sepsis patients are on the brink of acute kidney injury. While the current approach relies on sophisticated laboratory instruments and is not yet ready for bedside use, the identified molecules could be turned into faster, targeted assays in the future. If validated in larger, multi-center studies, such tests could allow clinicians to adjust drugs, fine-tune fluid and blood pressure management, and consider protective therapies before the kidneys reach a point of no return, potentially improving survival and recovery for some of the sickest patients.
Citation: Xu, W., Zhang, Z., Gu, F. et al. Integrating metabolomics and machine learning with in silico analysis to identify early biomarkers and molecular interactions in sepsis-associated acute kidney injury. Sci Rep 16, 10963 (2026). https://doi.org/10.1038/s41598-026-45255-0
Keywords: sepsis, acute kidney injury, metabolomics, biomarkers, machine learning