Clear Sky Science · en
The involvement of miRNAs in CYP450 enzymes and UDP-glucuronosyltransferases regulation in the human liver
Why this matters for medicines
When you swallow a pill, your body has to turn that chemical into something it can safely use or remove. Much of this work happens in the liver, which relies on a large set of helper proteins to break down drugs. This study explores how tiny RNA molecules, called microRNAs, may quietly dial these helpers up or down, potentially changing how different people respond to the same medicine.
Small regulators with a big impact
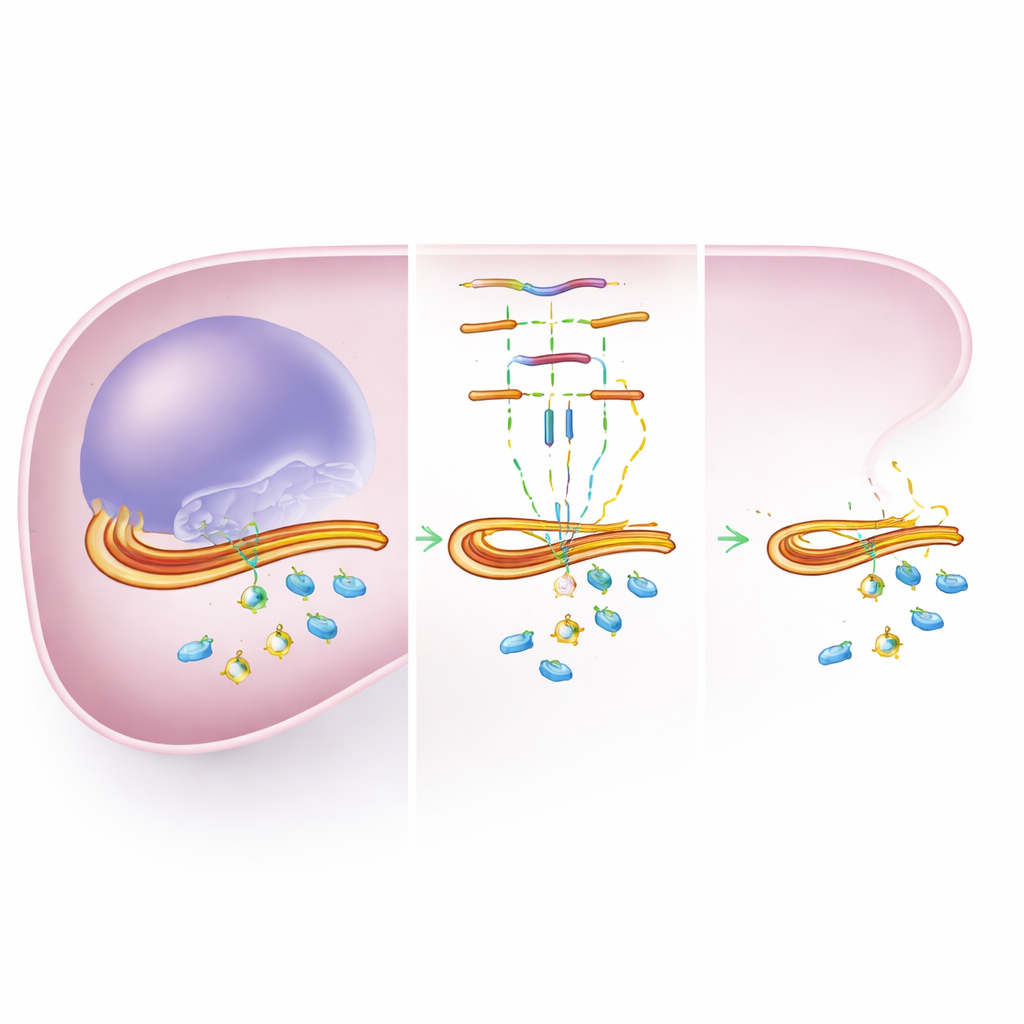
MicroRNAs are short pieces of genetic material that do not build proteins themselves but instead fine-tune how much protein other genes make. By attaching to the tail end of a gene’s message, they can slow or block the production of the corresponding protein. Because a single microRNA can touch many genes at once, and each gene can be influenced by several microRNAs, they form a dense control network that helps the liver adapt to stress, infection, and injury. The same network can also shift how quickly drugs are processed.
The liver’s drug-breaking toolkit
The liver relies heavily on two families of enzymes to handle medicines and other foreign chemicals. One family, known as CYP450 enzymes, performs the initial chemical changes that start to dismantle drug molecules. Another family, called UGT enzymes, then attaches a small sugar-like tag that makes these compounds easier to dissolve in water and flush out of the body. A handful of CYP and UGT members do most of the work for the medicines we use every day, so any change in their levels can strongly influence how much drug ends up in the bloodstream and for how long.
Hunting for the microRNA–enzyme links
The researchers first used several online databases to predict which microRNAs might bind to the genes behind key CYP and UGT enzymes. From nearly 500 possible pairings, they narrowed the list to 22 microRNAs that were actually detectable in human liver samples. These samples came from people with a range of serious liver diseases, plus a comparison group without such conditions. For each sample, they already had precise measurements of enzyme protein levels. By comparing microRNA amounts with enzyme amounts, they looked for patterns where higher microRNA levels matched lower enzyme levels, a hallmark of direct control.
What the patterns revealed
Several microRNAs showed such negative pairings with important enzymes. For example, certain microRNAs tended to go up when CYP2C8, CYP2C9, or CYP3A4—enzymes that handle many common drugs—went down. Similar relationships appeared for UGT enzymes involved in the second step of drug processing. To probe these relationships more deeply, the team focused on two particularly promising combinations: one microRNA (miR-655-3p) suspected of targeting CYP2C8, and another (miR-200a-3p) suspected of targeting UGT1A3. They introduced these microRNAs, along with the tail ends of the enzyme genes, into liver-like cells grown in the lab and measured how strongly a light-producing test signal was switched off.
Zooming in on a confirmed control switch
The cell experiments showed that miR-200a-3p clearly and strongly reduced the signal tied to UGT1A3, indicating that it can directly latch onto this gene’s message and curb its activity. In contrast, miR-655-3p only weakened the CYP2C8 signal slightly and not in a statistically convincing way, suggesting that if it influences this enzyme at all, it may do so only under certain conditions or together with other factors. The study also highlighted other intriguing microRNAs that tracked with changes in enzyme levels across different liver diseases, hinting at a broader web of control that still needs to be charted.
What this means for patients and drugs
Overall, the work supports the idea that microRNAs act as subtle dimmer switches for the liver’s drug-handling machinery. By helping to identify which microRNAs tune specific enzymes, the study lays groundwork for explaining why two people given the same dose of a medicine might experience different benefits or side effects—especially when liver disease is present. One clear example is miR-200a-3p, which can directly lower levels of a key UGT enzyme and may therefore slow the clearance of certain drugs. In the long run, understanding these tiny regulators could improve drug dosing, guide the choice of safer treatments for people with liver problems, and open the door to new therapies that adjust microRNA activity itself.
Citation: Szeląg-Pieniek, S., Perużyńska, M., Komaniecka, N. et al. The involvement of miRNAs in CYP450 enzymes and UDP-glucuronosyltransferases regulation in the human liver. Sci Rep 16, 14255 (2026). https://doi.org/10.1038/s41598-026-45113-z
Keywords: microRNAs, drug metabolism, liver enzymes, pharmacogenomics, epigenetic regulation