Clear Sky Science · en
Genomic convergence of multidrug resistance, virulence-associated loci, and phage defense systems in Klebsiella pneumoniae from pharmaceutical wastewater in Bangladesh
Why Wastewater Matters to Superbugs
Antibiotic resistance is often discussed in hospitals and clinics, but this study shows that the seeds of tomorrow’s “superbugs” can be sown far upstream—in the pipes of drug factories. The researchers investigated wastewater from antibiotic‑manufacturing plants in Bangladesh and discovered a strain of Klebsiella pneumoniae that brings together three worrying traits at once: strong resistance to many medicines, genetic features linked to severe disease, and an unusually powerful shield against viruses that infect bacteria. Understanding how such a strain emerges in the environment is crucial for protecting both local communities and global health.
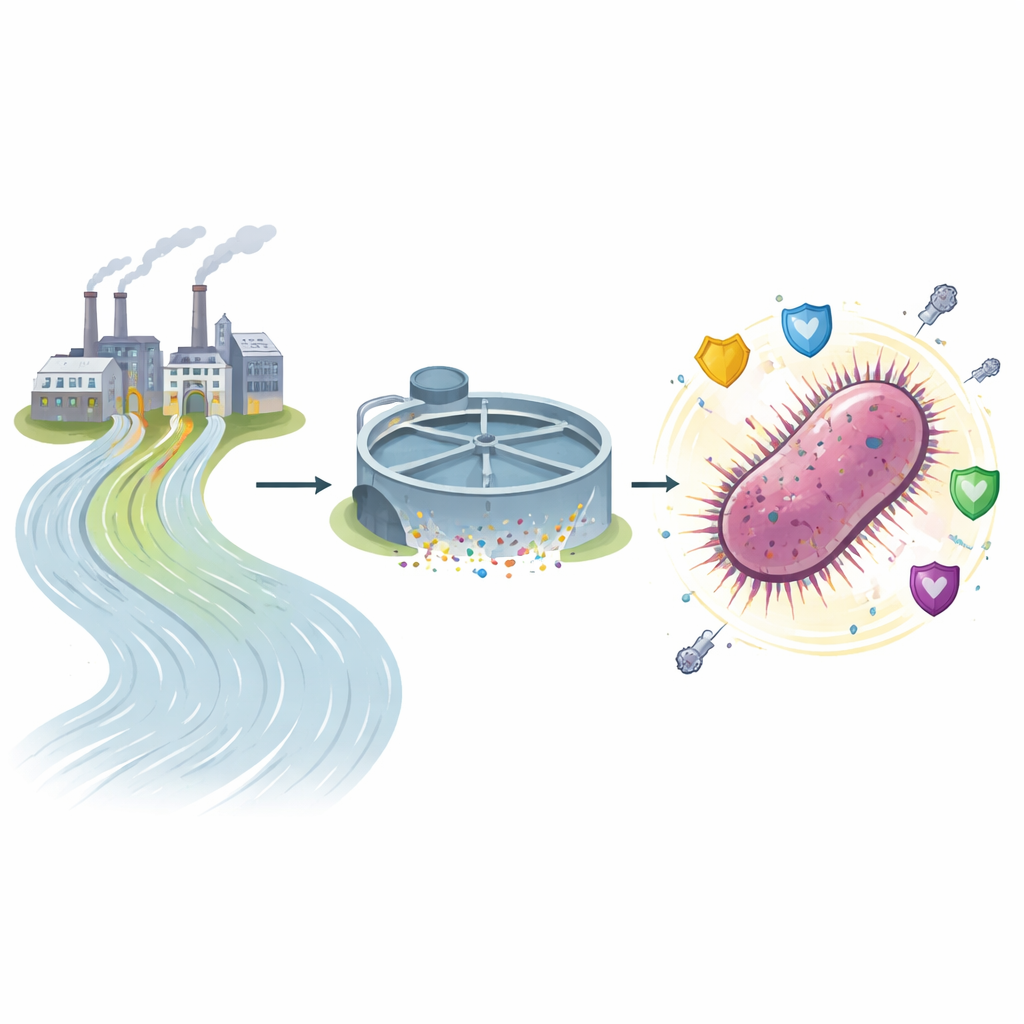
From Factory Pipes to Tough Bacteria
The team sampled both incoming and treated wastewater from five pharmaceutical factories near Dhaka. Standard treatment reduced organic pollution and removed detectable residues of two widely used antibiotics, ciprofloxacin and penicillin G. Yet the water still contained large numbers of living bacteria, many of them resistant to multiple drugs. Out of 150 isolates, resistance rates were high for common antibiotics, and about one‑fifth of the strains qualified as multidrug‑resistant. Klebsiella species were the most frequent among these hardy survivors, alongside other well‑known hospital troublemakers such as Acinetobacter baumannii and Escherichia coli.
Meeting a “Perfect Storm” Strain
One Klebsiella pneumoniae isolate, named JU‑BAEC‑01, stood out as the most drug‑resistant and was examined in depth using whole‑genome sequencing. The bacterium belongs to a distinct genetic lineage, different from typical outbreak strains, and carries a rich set of genes that help it thrive in harsh environments. It resists nearly all major antibiotic classes except two last‑resort drugs, carbapenems and colistin. This resistance comes largely from mobile DNA circles called plasmids, which act as swap‑able genetic toolkits. These plasmids carry a cocktail of genes that pump drugs out of the cell, chemically break them down, or alter the cell’s internal targets so antibiotics can no longer bind effectively.
Built‑In Tools for Causing Severe Disease
Beyond surviving antibiotics, JU‑BAEC‑01 also carries many genetic elements previously linked to especially invasive forms of Klebsiella that can cause liver abscesses and other severe infections in otherwise healthy people. The genome encodes powerful iron‑stealing systems that help the bacterium grow inside the body, structures such as fimbriae and sticky sugar coatings that enable it to cling to tissues and form hard‑to‑remove biofilms, and a molecular “spear gun” system used to attack host cells and rival microbes. Although this study did not test how dangerous the strain is in animals or humans, the presence of this combination of traits suggests a strong potential for serious disease if such bacteria enter clinical settings.
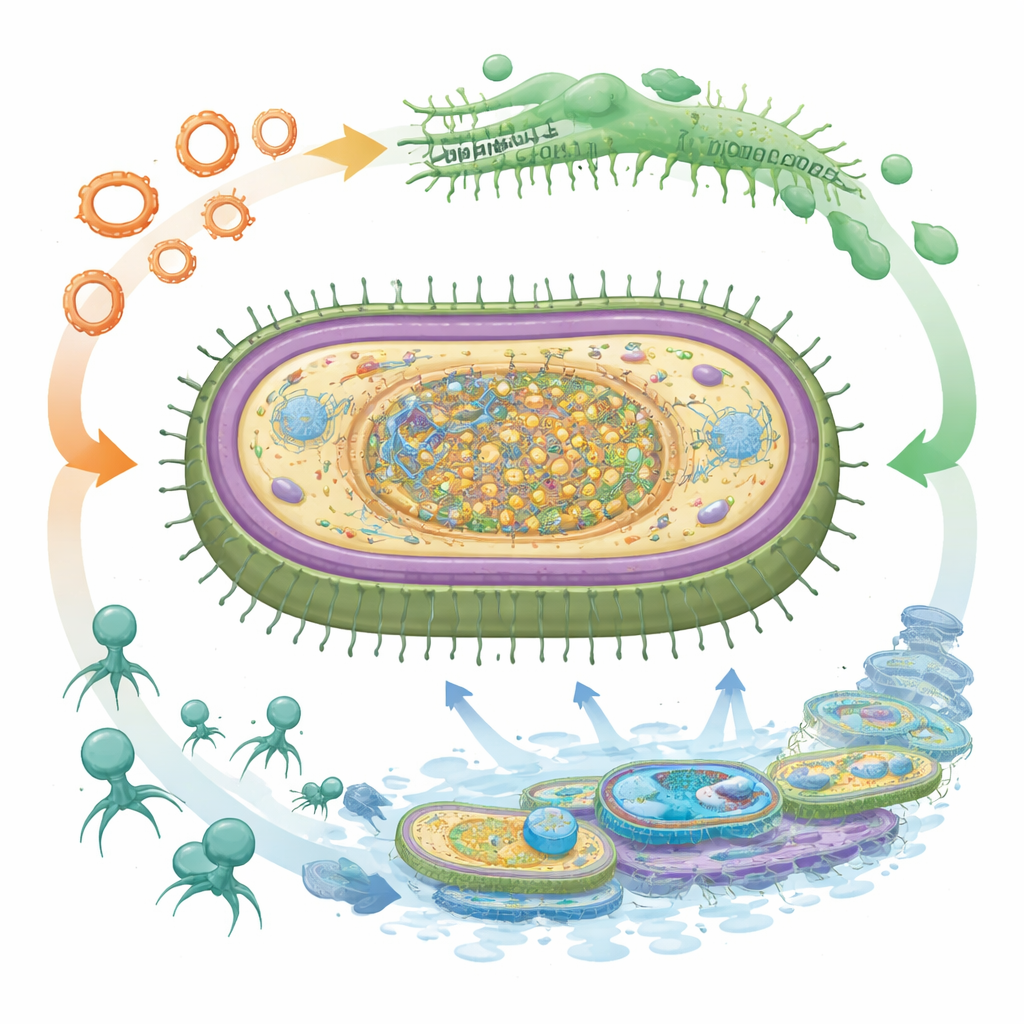
A Virus‑Proof Armor That Complicates Treatment
Perhaps the most striking discovery is the bacterium’s layered defenses against bacteriophages, the viruses that infect bacteria and are being explored as living drugs against resistant infections. JU‑BAEC‑01 carries several types of CRISPR‑Cas immune systems, which store genetic “mugshots” of past viral invaders and cut matching DNA on future encounters. It also has multiple restriction‑modification and BREX systems that recognize and block foreign genetic material, along with abortive infection systems that effectively sacrifice infected cells to halt virus spread. At the same time, viral DNA trapped in the genome encodes counter‑weapons that help phages dodge these defenses, revealing an ongoing arms race. This intricate shield may make phage‑based therapies harder to use against such strains, even as it allows the bacterium to keep acquiring helpful resistance and virulence genes.
What This Means for People and Policy
Taken together, the findings paint JU‑BAEC‑01 as a “perfect storm” bacterium that unites multidrug resistance, strong disease‑related traits, and an advanced anti‑virus toolkit—all in a strain recovered from treated industrial wastewater, not a hospital ward. While its real‑world virulence still needs experimental testing, its genetic profile warns that pharmaceutical effluents can act as breeding grounds and reservoirs for high‑risk lineages that may later reach patients. The study underscores the urgency of tightening controls on antibiotic pollution, improving wastewater treatment to remove both resistant bacteria and resistance genes, and maintaining genomic surveillance of industrial discharges. Doing so could help prevent today’s hidden environmental strains from becoming tomorrow’s untreatable infections.
Citation: Ahmed, M.F., Sarkar, M.M.H., Mehzabin, K. et al. Genomic convergence of multidrug resistance, virulence-associated loci, and phage defense systems in Klebsiella pneumoniae from pharmaceutical wastewater in Bangladesh. Sci Rep 16, 14554 (2026). https://doi.org/10.1038/s41598-026-45102-2
Keywords: antimicrobial resistance, pharmaceutical wastewater, Klebsiella pneumoniae, multidrug-resistant bacteria, bacteriophage defense